Abstract
Key message
Floral thermogenesis is an important reproductive strategy for attracting pollinators. We developed essential biological tools for studying floral thermogenesis using two species of thermogenic aroids, Symplocarpus renifolius and Alocasia odora.
Abstract
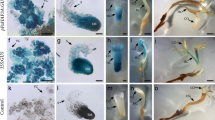
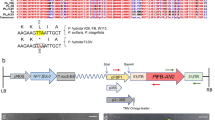
Aroids contain many species with intense heat-producing abilities in their inflorescences. Several genes have been proposed to be involved in thermogenesis of these species, but biological tools for gene functional analyses are lacking. In this study, we aimed to develop a protoplast-based transient expression (PTE) system for the study of thermogenic aroids. Initially, we focused on skunk cabbage (Symplocarpus renifolius) because of its ability to produce intense as well as durable heat. In this plant, leaf protoplasts were isolated from potted and shoot tip-cultured plants with high efficiency (ca. 1.0 × 105/g fresh weight), and more than half of these protoplasts were successfully transfected. Using this PTE system, we determined the protein localization of three mitochondrial energy-dissipating proteins, SrAOX, SrUCPA, and SrNDA1, fused to green fluorescent protein (GFP). These three GFP-fused proteins were localized in MitoTracker-stained mitochondria in leaf protoplasts, although the green fluorescent particles in protoplasts expressing SrUCPA-GFP were significantly enlarged. Finally, to assess whether the PTE system established in the leaves of S. renifolius is applicable for floral tissues of thermogenic aroids, inflorescences of S. renifolius and another thermogenic aroid (Alocasia odora) were used. Although protoplasts were successfully isolated from several tissues of the inflorescences, PTE systems worked well only for the protoplasts isolated from the female parts (slightly thermogenic or nonthermogenic) of A. odora inflorescences. Our developed system has a potential to be widely used in inflorescences as well as leaves in thermogenic aroids and therefore may be a useful biological tool for investigating floral thermogenesis.





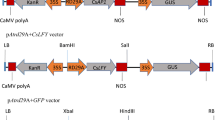
taken from the spathe. c Protoplasts isolated from the male (upper), sterile (middle), and female part (lower). The value in parenthesis indicates the isolation efficiency of protoplasts in each part. Few protoplasts were isolated from the appendix. d Bright-field (DIC) and fluorescent microphotographs (GFP) were collected on the Zeiss AxioImager A2 microscope equipped with appropriate filter sets
Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Aoki S, Toh S, Nakamichi N, Hayashi Y, Wang Y, Suzuki T, Tsuji H, Kinoshita T (2019) Regulation of stomatal opening and histone modification by photoperiod in Arabidopsis thaliana. Sci Rep 9:10054. https://doi.org/10.1038/s41598-019-46440-0
Azuma H, Kono M (2006) Estragole (4-allylanisole) is the primary compound in volatiles emitted from the male and female cones of Cycas revoluta. J Plant Res 119:671–676
Barreto P, Okura V, Pena IA, Maia R, Maia IG, Arruda P (2015) Overexpression of mitochondrial uncoupling protein 1 (UCP1) induces a hypoxic response in Nicotiana tabacum leaves. J Exp Bot 67:301–313
Barreto P, Yassitepe JECT, Wilson ZA, Arruda P (2017) Mitochondrial uncoupling protein 1 overexpression increases yield in Nicotiana tabacum under drought stress by improving source and sink metabolism. Front Plant Sci 8:1836. https://doi.org/10.3389/fpls.2017.01836
Bian F, Luo Y, Li L, Pang Y, Peng Y (2021) Inflorescence development, thermogenesis and flower-visiting insect activity in Alocasia odora. Flora 279:151818. https://doi.org/10.1016/j.flora.2021.151818
Burris KP, Dlugosz EM, Collins AG, Stewart CN Jr, Lenaghan SC (2016) Development of a rapid, low-cost protoplast transfection system for switchgrass (Panicum virgatum L.). Plant Cell Rep 35:693–704
Cao J, Yao D, Lin F, Jiang M (2014) PEG-mediated transient gene expression and silencing system in maize mesophyll protoplasts: a valuable tool for signal transduction study in maize. Acta Physiol Plant 36:1271–1281
Dahal K, Martyn GD, Alber NA, Vanlerberghe GC (2016) Coordinated regulation of photosynthetic and respiratory components is necessary to maintain chloroplast energy balance in varied growth conditions. J Exp Bot 68:657–671
Dahal K, Vanlerberghe GC (2018) Growth at elevated CO2 requires acclimation of the respiratory chain to support photosynthesis. Plant Physiol 178:82–100
Davey MR, Anthony P, Power JB, Lowe KC (2005) Plant protoplasts: status and biotechnological perspectives. Biotechnol Adv 23:131–171
Fiorani F, Umbach AL, Siedow JN (2005) The alternative oxidase of plant mitochondria is involved in the acclimation of shoot growth at low temperature. A study of Arabidopsis AOX1a transgenic plants. Plant Physiol 139:1795–1805
Fujikawa Y, Nakanishi T, Kawakami H, Yamasaki K, Sato MH, Tsuji H, Matsuoka M, Kato N (2014) Split luciferase complementation assay to detect regulated protein-protein interactions in rice protoplasts in a large-scale format. Rice 7:11. https://doi.org/10.1186/s12284-014-0011-8
Gibernau M, Barabe D, Moisson M, Trombe A (2005) Physical constraints on temperature difference in some thermogenic aroid inflorescences. Ann Bot 96:117–125
Grant NM, Miller RE, Watling JR, Robinson SA (2008) Synchronicity of thermogenic activity, alternative pathway respiratory flux, AOX protein content, and carbohydrates in receptacle tissues of sacred lotus during floral development. J Exp Bot 59:705–714
Hirata H, Ohnishi T, Ishida H, Tomida K, Sakai M, Hara M, Watanabe N (2012) Functional characterization of aromatic amino acid aminotransferase involved in 2-phenylethanol biosynthesis in isolated rose petal protoplasts. J Plant Physiol 169:444–451
Inaba T, Nagano Y, Sakakibara T, Sasaki Y (1999) Identification of a cis-regulatory element involved in phytochrome down-regulated expression of the pea small GTPase gene pra2. Plant Physiol 120:491–500
Ito-Inaba Y, Hida Y, Ichikawa M, Kato Y, Yamashita T (2008) Characterization of the plant uncoupling protein, SrUCPA, expressed in spadix mitochondria of the thermogenic skunk cabbage. J Exp Bot 59:995–1005
Ito-Inaba Y, Hida Y, Inaba T (2009) What is critical for plant thermogenesis? Differences in mitochondrial activity and protein expression between thermogenic and non-thermogenic skunk cabbages. Planta 231:121–130
Ito-Inaba Y, Masuko-Suzuki H, Maekawa H, Watanabe M, Inaba T (2016) Characterization of two PEBP genes, SrFT and SrMFT, in thermogenic skunk cabbage (Symplocarpus renifolius). Sci Rep 6:29440. https://doi.org/10.1038/srep29440
Ito-Inaba Y, Sato M, Sato MP, Kurayama Y, Yamamoto H, Ohata M, Ogura Y, Hayashi T, Toyooka K, Inaba T (2019) Alternative oxidase capacity of mitochondria in microsporophylls may function in cycad thermogenesis. Plant Physiol 180:743–756
Kakizaki Y, Moore AL, Ito K (2012) Different molecular bases underlie the mitochondrial respiratory activity in the homoeothermic spadices of Symplocarpus renifolius and the transiently thermogenic appendices of Arum maculatum. Biochem J 445:237–246
Kim J, Somers DE (2010) Rapid assessment of gene function in the circadian clock using artificial MicroRNA in Arabidopsis mesophyll protoplasts. Plant Physiol 154:611–621
Kitaura H, Mori M, Nishibori Y, Ohtani H (2008) A study on the mass propagation of skunk cabbages (Symplocarpus foetidus). Bull Shiga Prefect Agric Technol Promot Cent 47:43–50
Kite GC, Hetterscheid WLA (2017) Phylogenetic trends in the evolution of inflorescence odours in Amorphophallus. Phytochemistry 142:126–142
Knutson RM (1974) Heat production and temperature regulation in eastern skunk cabbage. Science 186:746–747
Kumano Y, Yamaoka R (2006) Synchronization between temporal variation in heat generation, floral scents and pollinator arrival in the beetle-pollinated tropical Araceae Homalomena propinqua. Plant Species Biol 21:173–183
Lin H-Y, Chen J-C, Fang S-C (2018) A protoplast transient expression system to enable molecular, cellular, and functional studies in Phalaenopsis orchids. Front Plant Sci. https://doi.org/10.3389/fpls.2018.00843
Liu J, Li Z, Wang Y, Xing D (2014) Overexpression of alternative oxidase1a alleviates mitochondria-dependent programmed cell death induced by aluminium phytotoxicity in Arabidopsis. J Exp Bot 65:4465–4478
Marotz-Clausen G, Jürschik S, Fuchs R, Schäffler I, Sulzer P, Gibernau M, Dötterl S (2018) Incomplete synchrony of inflorescence scent and temperature patterns in Arum maculatum L. (Araceae). Phytochemistry 154:77–84
Masani MY, Noll GA, Parveez GK, Sambanthamurthi R, Prüfer D (2014) Efficient transformation of oil palm protoplasts by PEG-mediated transfection and DNA microinjection. PLoS ONE 9:e96831. https://doi.org/10.1371/journal.pone.0096831
Meeuse BJD, Raskin I (1988) Sexual reproduction in the arum lily family, with emphasis on thermogenicity. Sex Plant Reprod 1:3–15
Miller RE, Grant NM, Giles L, Ribas-Carbo M, Berry JA, Watling JR, Robinson SA (2011) In the heat of the night—alternative pathway respiration drives thermogenesis in Philodendron bipinnatifidum. New Phytol 189:1013–1026
Moore AL, Shiba T, Young L, Harada S, Kita K, Ito K (2013) Unraveling the heater: new insights into the structure of the alternative oxidase. Ann Rev Plant Biol 64:637–663
Nagata T, Takebe I (1971) Plating of isolated tobacco mesophyll protoplasts on agar medium. Planta 99:12–20
Nanjareddy K, Arthikala M-K, Blanco L, Arellano ES, Lara M (2016) Protoplast isolation, transient transformation of leaf mesophyll protoplasts and improved Agrobacterium-mediated leaf disc infiltration of Phaseolus vulgaris: tools for rapid gene expression analysis. BMC Biotechnol 16:53. https://doi.org/10.1186/s12896-016-0283-8
Nie Z-L, Sun H, Li H, Wen J (2006) Intercontinental biogeography of subfamily Orontioideae (Symplocarpus, Lysichiton, and Orontium) of Araceae in eastern Asia and North America. Mol Phylogenetics Evol 40:155–165
Oguri S, Sakamaki K, Sakamoto H, Kubota K (2019) Compositional changes of the floral scent volatile emissions from Asian skunk cabbage (Symplocarpus renifolius, Araceae) over flowering sex phases. Phytochem Anal 30:139–147
Page MT, Parry MAJ, Carmo-Silva E (2019) A high-throughput transient expression system for rice. Plant Cell Environ 42:2057–2064
Ren R, Gao J, Lu C, Wei Y, Jin J, Wong S-M, Zhu G, Yang F (2020) Highly efficient protoplast isolation and transient expression system for functional characterization of flowering related genes in cymbidium orchids. Int J Mol Sci 21:2264
Salzman S, Crook D, Crall JD, Hopkins R, Pierce NE (2020) An ancient push-pull pollination mechanism in cycads. Sci Adv 6:eaay6169. https://doi.org/10.1126/sciadv.aay6169
Sasamoto H, Ashihara H (2014) Effect of nicotinic acid, nicotinamide and trigonelline on the proliferation of lettuce cells derived from protoplasts. Phytochem Lett 7:38–41
Seymour RS (2010) Scaling of heat production by thermogenic flowers: limits to floral size and maximum rate of respiration. Plant Cell Environ 33:1474–1485
Seymour RS, Blaylock AJ (1999) Switching off the heater: influence of ambient temperature on thermoregulation by eastern skunk cabbage Symplocarpus foetidus. J Exp Bot 50:1525–1532
Seymour RS, Matthews PG (2006) The role of thermogenesis in the pollination biology of the Amazon waterlily Victoria amazonica. Ann Bot 98:1129–1135
Shen Y, Meng D, McGrouther K, Zhang J, Cheng L (2017) Efficient isolation of Magnolia protoplasts and the application to subcellular localization of MdeHSF1. Plant Methods 13:44. https://doi.org/10.1186/s13007-017-0193-3
Shi T, Toda JM, Takano TK, Yafuso M, Suwito A, Wong YS, Shang SQ, Gao JJ (2019) A review of taxonomy and flower-breeding ecology of the Colocasiomyia toshiokai species group (Diptera: Drosophilidae), with description of a new species from Indonesia. Eur J Entomol 116:341–361
Shirasu M, Fujioka K, Kakishima S, Nagai S, Tomizawa Y, Tsukaya H, Murata J, Manome Y, Touhara K (2010) Chemical identity of a rotting animal-like odor emitted from the inflorescence of the Titan arum (Amorphophallus titanum). Biosci Biotech Biochem 74:2550–2554
Smith AMO, Ratcliffe RG, Sweetlove LJ (2004) Activation and function of mitochondrial uncoupling protein in plants. J Biol Chem 279:51944–51952
Song J, Sorensen EL, Liang GH (1990) Direct embryogenesis from single mesophyll protoplasts in alfalfa (Medicago sativa L.). Plant Cell Rep 9:21–25
Stensmyr MC, Urru I, Collu I, Celander M, Hansson BS, Angioy AM (2002) Pollination: rotting smell of dead-horse arum florets. Nature 420:625–626
Sweetlove LJ, Lytovchenko A, Morgan M, Nunes-Nesi A, Taylor NL, Baxter CJ, Eickmeier I, Fernie AR (2006) Mitochondrial uncoupling protein is required for efficient photosynthesis. Proc Natl Acad Sci USA 103:19587–19592
Sweetman C, Waterman CD, Rainbird BM, Smith PMC, Jenkins CD, Day DA, Soole KL (2019) AtNDB2 Is the main external NADH dehydrogenase in mitochondria and is important for tolerance to environmental stress. Plant Physiol 181:774–788
Tang W (1987) Heat production in cycad cones. Botl Gaz 148:165–174
Terry I, Moore CJ, Walter GH, Forster PI, Roemer RB, Donaldson JD, Machin PJ (2004) Association of cone thermogenesis and volatiles with pollinator specificity in Macrozamia cycads. Plant Syst Evol 243:233–247
Terry I, Walter GH, Moore C, Roemer R, Hull C (2007) Odor-mediated push-pull pollination in cycads. Science 318:70. https://doi.org/10.1126/science.1145147
Uemura S, Ohkawara K, Kudo G, Wada N, Higashi S (1993) Heat-production and cross-pollination of the Asian skunk cabbage Symplocarpus renifolius. Am J Bot 80:635–640
Wagner AM, Krab K, Wagner MJ, Moore AL (2008) Regulation of thermogenesis in flowering Araceae: the role of the alternative oxidase. Biochim Biophys Acta Bioenerg 1777:993–1000
Watling JR, Robinson SA, Seymour RS (2006) Contribution of the alternative pathway to respiration during thermogenesis in flowers of the sacred lotus. Plant Physiol 140:1367–1373
Wehner N, Hartmann L, Ehlert A, Böttner S, Oñate-Sánchez L, Dröge-Laser W (2011) High-throughput protoplast transactivation (PTA) system for the analysis of Arabidopsis transcription factor function. Plant J 68:560–569
Yafuso M (1993) Thermogenesis of Alocasia odora (Araceae) and the role of Colocasiomyia flies (Diptera: Drosophilidae) as cross-pollinators. Environ Entomol 22:601–606
Yoo S-D, Cho Y-H, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2:1565–1572
Zhai Z, Sooksa-nguan T, Vatamaniuk OK (2009) Establishing RNA interference as a reverse-genetic approach for gene functional analysis in protoplasts. Plant Physiol 149:642–652
Zhang Y, Su J, Duan S, Ao Y, Dai J, Liu J, Wang P, Li Y, Liu B, Feng D, Wang J, Wang H (2011) A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods 7:30. https://doi.org/10.1186/1746-4811-7-30
Zhang X, Wang L, He C, Luo H (2016) An efficient transient mesophyll protoplast system for investigation of the innate immunity responses in the rubber tree (Hevea brasiliensis). Plant Cell Tissue Organ Cult 126:281–290
Acknowledgements
We thank Yoko Katayama (University of Miyazaki) for their assistance in the cultivation of skunk cabbage plants. This work was supported by a Grant-in-Aid for Scientific Research (grant no. 20H02917 to Y.I.-I., and grant nos. 18H02169 and 20K21282 to T. I.), by the Takeda Science Foundation, and by the Sasakawa Scientific Research Grant from The Japan Science Society.
Funding
This work was supported by a Grant-in-Aid for Scientific Research (grant no. 20H02917 to Y.I.-I., and grant nos. 18H02169 and 20K21282 to T. I.), by the Takeda Science Foundation, and by the Sasakawa Scientific Research Grant from The Japan Science Society.
Author information
Authors and Affiliations
Contributions
HM, MO, KM, DK, TI, and YII conducted the experiments. YII and TI analyzed the data. YII, MPS, and TT contributed to the experimental design. YII wrote the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Communicated by Kinya Toriyama.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Maekawa, H., Otsubo, M., Sato, M.P. et al. Establishing an efficient protoplast transient expression system for investigation of floral thermogenesis in aroids. Plant Cell Rep 41, 263–275 (2022). https://doi.org/10.1007/s00299-021-02806-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-021-02806-1