Abstract
An increased level of apoptotic material and B cell activation leading to autoantibody production are hallmarks of systemic lupus erythematoses (SLE). Increased FAS expression, apoptosis, and CD154-mediated signaling, enabling T–B cell interaction are involved in the pathogenesis of SLE. This study addresses the expression profile of CD154 and FAS in the peripheral blood of patients with SLE, rheumatoid arthritis (RA) and normal healthy control donors. Surface markers on peripheral blood T and B cells from patients and healthy control donors were assessed using flow cytometry. The expression of CD154 and FAS were significantly increased in T and B cells of SLE patients as compared to healthy control donors and RA patients. In SLE and RA patients, FAS expression strongly correlated with CD154 expression on T cells, which was not found in healthy control donors. FAS expression was also associated with the occurrence of anti-DNA antibodies. We demonstrate high CD154 and FAS expression as a characteristic feature of SLE. This pattern may reflect simultaneous activation of apoptosis and activation of B–T cell interaction in SLE.
Similar content being viewed by others
Introduction
Systemic lupus erythematoses (SLE) is a systemic autoimmune disease characterized by plasma B cells secreting nuclear autoantibodies [11], especially the pathognomonic anti-double-stranded DNA autoantibodies (anti-dsDNA). B and T cell interaction is required for the induction of humoral immunity. T cells can induce B cell proliferation and differentiation via the ligation of CD154 by CD40 as a costimulatory molecule. The interaction of T and B cells in SLE is of major importance for the pathogenesis of the disease. CD154 hyperexpression in patients with active SLE was shown on T cells, B cells, and monocytes and correlated with disease activity [4]. Interestingly, not only membrane bound, but also soluble CD154 levels correlate with disease activity and enhanced titers of anti-dsDNA in patients with SLE, indicating a central role for the pathogenesis of CD154 [9]. Underlining the importance of the CD154–CD40 interaction in SLE, treatment with a monoclonal Abs to CD154 significantly decreased disease activity in SLE patients [3]. However, this treatment is associated with thromboembolic events [13] as a severe side effect leading to the hypothesis that CD154 cross-linking has effects aside co-stimulation.
An accumulation of apoptotic remnants in the tissue is observed in a subgroup of patients with SLE. FAS (CD95) and FAS–ligand interaction are associated with the induction of apoptosis [7]. The stimulation of autoreactive B cells with anti-CD40 leads to an increase in FAS-mediated apoptosis in vitro [8]. In parallel, elevated plasma levels of sCD154 in patients with SLE increase CD95 expression on B cells [16]. B cells, stimulated via CD40, are highly susceptible to FAS-mediated apoptosis [8], when exposed to FAS-ligand expressing T cells [14]. Consequently, we hypothesized that the CD154–CD40 mediated interaction and the upregulation of FAS may occur simultaneously in SLE. Therefore, we measured the expression of FAS, CD40, and CD154 on T and B lymphocytes in a cohort of 52 patients with SLE, 32 patients with rheumatoid arthritis (RA), and 40 normal healthy control donors. The expression of these surface markers was correlated with each other and with the clinical course of SLE.
Materials and methods
Patient data
The patient population comprises 52 SLE and 32 RA patients. All patients were treated with disease modifying anti-rheumatic drugs (DMARDs) and/or corticosteroids. At the time of first analysis, all patients had an established disease. Disease activity differed, and was assessed by serum levels of C-reactive protein (CRP), erythrocyte sedimentation rates [10], and SLE disease activity index (SLEDAI) score for each patient and the use of medication was recorded (Table 1). Forty age and sex-matched healthy donors served as controls. The study was approved by the University of Erlangen–Nuremberg Institutional Review Board, and written informed consent was obtained from all individuals before entering the study.
Immune phenotyping
Whole blood was stained for 30 min with saturating amounts of directly labeled mAbs. After automated lyses of the red blood cells (Q-Prep, Beckman Coulter, Cambridge, MA), the leucocytes were fixed with 1% paraformaldehyde and analyzed using flow cytometry (EPICS, Beckman Coulter, Cambridge, MA). The following phycoerythrin (PE), fluorescein-isothiocyanate (FITC), or CyChrome (Cy5) conjugated monoclonal antibodies were used: anti-CD4 (FITC/PE), anti-CD19 (FITC/PE/Cy5) (PE), anti-CD40 (FITC), anti-CD154 (FITC), and anti-CD95 (PE) (BD Pharmingen, Heidelberg, Germany). Unlabeled cells were used as controls for determining background and autofluorescence.
Statistical analysis
Differences in data distribution were analyzed by two-tailed Student t tests. Correlation analysis was performed following the Pearson test. Comparison of two correlation analysis was assessed by the MetaAnalysis program 5.3 of Ralf Schwarzer; http://web.fu-berlin.de/gesund/gesu_engl/meta_e.htm.
Results
Expression of CD154 in SLE patients in comparison to RA and healthy control donors
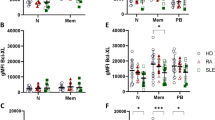
Since ligation of the costimulatory molecule CD40 on B cells by its ligand CD154 on T cells plays a central role for B cell activation, we assessed the expression of both molecules using flow cytometry. The individual percentages of CD154 expressing B cells (CD19pos, Fig. 1a) and T helper cells (CD4pos, Fig. 1b) were significantly increased in patients with SLE compared with healthy control donors and patients with RA (B cells) or only compared with healthy control donors (T helper cells). In detail, 0.6 ± 0.7% of B cells from healthy control donors were positive for CD154, which was comparable to RA patients (0.6 ± 0.8%). In contrast, CD154 expression was significantly more frequent (2.1 ± 3.2%) in patients suffering from SLE. Similarly, CD154 expression was also increased in T helper cells of patients with SLE (1.8 ± 3.1%) as compared to healthy control donors (0.6 ± 0.8%). The data suggest that expression of the costimulatory molecule CD154 is increased in T and B cells of SLE patients. Thus, an increased expression of CD154 in B cells also discriminates SLE patients from other autoimmune diseases such as RA [12].
CD154 and CD40 expression in SLE: T and B cells were isolated from full blood by gradient centrifugation. The percentage of a CD154pos B and b CD4+ T cells and c CD40pos B cells was assessed using flow cytometry. All data are represented as dots for every individual patient and healthy control donor. Percentages of cells expressing certain surface markers are demonstrated for patients with RA and SLE represented on the first and second and such found on healthy control donors (NHD) on the right column, representatively. Statistical differences were calculated employing the student’s t test
Expression of CD40 in SLE patients in comparison to RA and healthy control donors
In parallel, we assessed CD40, the ligand of CD154 in peripheral blood cells of patients with SLE and RA as well as healthy control donors. CD40 expression was found in the majority of B cells although there was a trend to a lower expression in SLE patients (71.9 ± 22.9%) than in healthy control donors (85.4 ± 14.1%) and RA patients (79.2 ± 18.6%) (Fig. 1c). Thus, in contrast to the findings made for CD154 expression on SLE patient’s B cells no discriminative propensity for CD40 expression could be found among RA patients, healthy donors and SLE patients.
Expression of FAS in SLE patients in comparison to RA and healthy control donors
An accumulation of apoptotic material is observed in various tissues of patients with SLE. Since CD154 leads to an augmented expression of CD95, the expression of this pro-apoptotic molecule was also analyzed. FAS expression was increased in B cells (Fig. 2a) and T cells (Fig. 2b) from patients with SLE in comparison to healthy control donors. In detail, 38 ± 20% of B cells and 66 ± 14% of T cells expressed FAS in SLE patients. In healthy control donors only 19 ± 7% of B cells and 51 ± 9% of T cells expressed FAS. Importantly also RA patients displayed a lower expression of FAS in B cells (25 ± 14%), whereas its expression in T cells (63 ± 13%) was comparable to the expression in T cells of SLE patients.
CD95 expression in SLE: T and B cells were isolated from full blood by gradient centrifugation. Percentages of CD95pos cells among a CD4pos T and b CD19pos T cells were assessed with flow cytometry. All data are represented as dots for every individual patient in two columns. Percentages of cells expressing CD95 are demonstrated for patients with RA, SLE, and healthy control donors (NHD). Statistical differences were calculated employing the Student’s t test. c Patients with SLE were separated into two groups: negative (left) and positive (right) for anti-DNA antibodies. Percentages of CD95pos T cells are demonstrated for both groups as mean ± standard deviation. Statistical differences were calculated employing the Student’s t test. Correlation analysis of CD154 and CD95 expression on T cells of SLE patients d and healthy control donors e Every dot represents the individual expressional percentage found within one patient for CD154 and CD95 on T cells. The correlation line is depicted as a line with the 95% confidence interval within the graph
Correlation of CD154 and FAS expression
To delineate an association between CD154 and CD95 expression correlation analysis was performed. There was a significant correlation (r = 0.75; P = 0.001, Fig. 2d) between the percentages of CD154pos and CD95pos T cells in patients with SLE. To delineate whether this balance is similar a correlation analysis was also performed in healthy control donors. However, the correlation was much less pronounced (r = 0.35, P = 0.0252, Fig. 2e). In RA patients, the correlation of CD154pos and CD95pos T cells was similar as observed in SLE patients (r = 0.72, P = 0.0001, data not shown). Importantly, the correlations calculated for SLE and RA patients differed significantly (P = 0.003) from the correlation calculated in healthy control donors. Thus, CD154 and CD95 expression strongly correlate in T cells of patients with SLE and RA.
Since FAS is associated with the induction of apoptosis and an increase in apoptotic material is a hallmark of SLE, we next addressed whether increased FAS expression in T cells is associated with increased SLE disease activity. Indeed, patients with SLE and high FAS expression in T cells also display an increased amount of anti-DNA antibodies (Fig. 2c).
Discussion
In this study, an increased expression of CD154 and FAS was denoted in peripheral blood T and B cells of patients with SLE as compared to healthy control donors. Moreover, the expression of CD154 in B cells and FAS in T and B cells of SLE was higher than the expression of both molecules in lymphocytes of RA patients. Moreover, there was tight correlation observed between CD154 and FAS expression in SLE and RA. These findings lead us to the hypothesis of an altered balance between CD154 and FAS on T cells in autoimmunity. FAS expression was also associated with the occurrence of anti-DNA antibodies a good marker of disease activity in SLE.
In coherence with various other publications, we also found an increased expression of CD154 on T cells [4] and B cells [6] in patients with SLE. In contrast to the published data [4], the increased expression of CD154 did not correlate with disease activity as assessed by SLEDAI, but corelated to the occurrence of anti-DNA antibodies levels, which is a surrogate marker for disease activity in SLE patients. Thus, we assume that CD154 plays an important role in patients with SLE that might lead to an increase in clinical disease activity as shown for the correlation of CD95pos with CD154 on T cells on one hand and the occurrence of anti-DNA antibodies on the other hand.
Treatment with neutralizing antibody against CD154 [13] leads to the occurrence of thromboembolic events. Because anti-phospholipid antibodies are associated with vascular events in SLE and of monoclonal antibodies to CD40 was linked to the anti-phospholipid antibodies in vivo [2], we analyzed whether increased levels of CD40 of CD154 expressing cells correlate with anti-phospholipid antibodies in our cohort. No association could be found (data not shown).
Autoantibodies against FAS–ligand can be found in 30% of the patients with SLE. It has been suggested that this leads to an inhibition of FAS/FAS-L-mediated apoptosis [15]. In contrast, we demonstrated that the percentage of FAS expressing cells is increased on lymphocytes of patients with SLE. Whether this increase in FAS expression is a reaction on autoreactive anti-FAS antibodies remains unclear. However, the level of apoptotic material in patients with SLE is increased. This may be due to a clearance defect of apoptotic material in patients with SLE [5]. Alternatively, it could also be associated with an increased induction of apoptosis [12], potentially mediated by increased expression of FAS. Since the expression level of FAS correlates with CD154 and, in parallel, the disease activity, an increase in disease activity may lead to an increase in apoptosis. In coherence, an increased expression of CD154 leads to an increased expression of FAS–ligand and induced, subsequently, apoptosis in biliary epithelial cells [1].
In summary, we showed that the expression of CD154 and FAS on SLE patients’ T and B cells was increased and correlated with each other in tight correlation. FAS expression was also associated with the occurrence of anti-DNA antibodies a good marker of disease activity in SLE.
References
Afford SC, Ahmed-Choudhury J, Randhawa S, Russell C, Youster J, Crosby HA, Eliopoulos A, Hubscher SG, Young LS, Adams DH (2001) CD40 activation-induced, Fas-dependent apoptosis and NF-kappaB/AP-1 signaling in human intrahepatic biliary epithelial cells. FASEB J 15:2345–2354. doi:10.1096/fj.01-0088com
Aleksandrova EN, Novikov AA, Popkova TV, Reshetniak TM, Novikova DS, Kliukvina NG, Il’ina AE, Mach ES, Volkov AV, Nasonov EL (2006) Soluble CD40 ligand in systemic lupus erythematosus and antiphospholipid syndrome. Ter Arkh 78:35–39
Boumpas DT, Furie R, Manzi S, Illei GG, Wallace DJ, Balow JE, Vaishnaw A (2003) A short course of BG9588 (anti-CD40 ligand antibody) improves serologic activity and decreases hematuria in patients with proliferative lupus glomerulonephritis. Arthritis Rheum 48:719–727. doi:10.1002/art.10856
Desai-Mehta A, Lu L, Ramsey-Goldman R, Datta SK (1996) Hyperexpression of CD40 ligand by B and T cells in human lupus and its role in pathogenic autoantibody production. J Clin Invest 97:2063–2073. doi:10.1172/JCI118643
Gaipl US, Voll RE, Sheriff A, Franz S, Kalden JR, Herrmann M (2005) Impaired clearance of dying cells in systemic lupus erythematosus. Autoimmun Rev 4:189–194. doi:10.1016/j.autrev.2004.10.007
Grammer AC, Bergman MC, Miura Y, Fujita K, Davis LS, Lipsky PE (1995) The CD40 ligand expressed by human B cells costimulates B cell responses. J Immunol 154:4996–5010
Haas JP, Grunke M, Frank C, Kolowos W, Dirnecker D, Leipold G, Hieronymus T, Lorenz HM, Herrmann M (1998) Increased spontaneous in vitro apoptosis in double negative T cells of humans with a fas/apo-1 mutation. Cell Death Differ 5:751–757. doi:10.1038/sj.cdd.4400426
Hirose S, Yan K, Abe M, Jiang Y, Hamano Y, Tsurui H, Shirai T (1997) Precursor B cells for autoantibody production in genomically Fas-intact autoimmune disease are not subject to Fas-mediated immune elimination. Proc Natl Acad Sci USA 94:9291–9295. doi:10.1073/pnas.94.17.9291
Kato K, Santana-Sahagun E, Rassenti LZ, Weisman MH, Tamura N, Kobayashi S, Hashimoto H, Kipps TJ (1999) The soluble CD40 ligand sCD154 in systemic lupus erythematosus. J Clin Invest 104:947–955. doi:10.1172/JCI7014
Lederer JA, Perez VL, DesRoches L, Kim SM, Abbas AK, Lichtman AH (1996) Cytokine transcriptional events during helper T cell subset differentiation. J Exp Med 184:397–406. doi:10.1084/jem.184.2.397
Lipsky PE (2001) Systemic lupus erythematosus: an autoimmune disease of B cell hyperactivity. Nat Immunol 2:764–766. doi:10.1038/ni0901-764
Lorenz HM, Grunke M, Hieronymus T, Herrmann M, Kuhnel A, Manger B, Kalden JR (1997) In vitro apoptosis and expression of apoptosis-related molecules in lymphocytes from patients with systemic lupus erythematosus and other autoimmune diseases. Arthritis Rheum 40:306–317. doi:10.1002/art.1780400216
Nakamura M, Tanaka Y, Satoh T, Kawai M, Hirakata M, Kaburaki J, Kawakami Y, Ikeda Y, Kuwana M (2006) Autoantibody to CD40 ligand in systemic lupus erythematosus: association with thrombocytopenia but not thromboembolism. Rheumatology (Oxford) 45:150–156. doi:10.1093/rheumatology/kei118
Schattner EJ, Elkon KB, Yoo DH, Tumang J, Krammer PH, Crow MK, Friedman SM (1995) CD40 ligation induces Apo-1/Fas expression on human B lymphocytes and facilitates apoptosis through the Apo-1/Fas pathway. J Exp Med 182:1557–1565. doi:10.1084/jem.182.5.1557
Suzuki N, Ichino M, Mihara S, Kaneko S, Sakane T (1998) Inhibition of Fas/Fas ligand-mediated apoptotic cell death of lymphocytes in vitro by circulating anti-Fas ligand autoantibodies in patients with systemic lupus erythematosus. Arthritis Rheum 41:344–353. doi:10.1002/1529-0131(199802)41:2<344::AID-ART19>3.0.CO;2-J
Vakkalanka RK, Woo C, Kirou KA, Koshy M, Berger D, Crow MK (1999) Elevated levels and functional capacity of soluble CD40 ligand in systemic lupus erythematosus sera. Arthritis Rheum 42:871–881. doi:10.1002/1529-0131(199905)42:5<871::AID-ANR5>3.0.CO;2-J
Acknowledgments
This work was supported by “Deutsche Forschungsgemeinschaft” SFB 643 (project B5), by the Interdisciplinary Centre for Clinical Research (IZKF) (project number A4 and N2) at the University Hospital of the University of Erlangen–Nuremberg, by the European Commissions [E.U. (QLK3-CT-2002-02017_APOCLEAR)], by the Lupus Erythemathodes Selbsthilfegemeinschaft e.V., by the “Responsif GmbH” Erlangen, by the “Doktor Robert Pfleger” Foundation, Bamberg, by the ELAN-Fond of the university of Erlangen Nuremberg (Pro.-Nr. 53410026), A grand dedicated to Maria Elena Manea and Doru Deica by the Alexander von Humboldt Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
M. E. Manea and R. B. Mueller contributed equally to the work.
Rights and permissions
About this article
Cite this article
Manea, M.E., Mueller, R.B., Dejica, D. et al. Increased expression of CD154 and FAS in SLE patients’ lymphocytes. Rheumatol Int 30, 181–185 (2009). https://doi.org/10.1007/s00296-009-0933-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-009-0933-4