Abstract
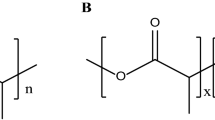
A copolymer of limonene with 2-(4-methoxyphenylamino)-2-oxoethyl methacrylate (LIM-co-MPAEMA) which is thought to be a non-toxic and used generally as a dietary supplement was synthesized and characterized for the first time both experimentally and theoretically. The structure of synthesized limonene copolymer was chemically characterized by Fourier transform infrared and nuclear magnetic resonant (1H NMR) spectroscopic techniques and compared with the theoretical calculated results. Charge transfer that is important in the formation of chemically bonded adducts causing cancer is quantitatively calculated. It was found that this polymer can be used as a biomaterial.












Similar content being viewed by others
References
Odian G (2004) Principles of polymerization. Wiley, Hoboken
Kerton FM, Marriott R (2013) Alternative solvents for green chemistry. In: Clark JH, Kraus G, Stankiewicz A (eds) Renewable solvents, vol 20. Royal Society of Chemistry, Cambridge, p 97
Kim YW, Kim MJ, Chung BY, Bang DY, Lim SK, Choi SM, Lim DS, Cho MC, Yoon K, Kim HS, Kim KB, Kim YS, Kwack SJ, Lee B (2013) Safety evaluation and risk assessment of D-limonene. J Toxicol Environ Health B 16:17
Filipsson AF, Bard J, Karlsson S (1998) Concise international chemical assessment document 5: limonene. World Health Organization, Geneva, p 1
Bauer K, Garbe D (2006) Surburg H. In: Bauer K (ed) Common fragrance and flavor materials: preparation, properties and uses. Wiley, Weinheim, p 330
Breitmaier E (2006) Terpenes: importance, general structure, and biosynthesis. Wiley, Weinheim, p 1
Gu Y, Jerome F (2013) Bio-based solvents: an emerging generation of fluids for the design of eco-efficient processes in catalysis and organic chemistry. Chem Soc Rev 42:9550
Mohammad A (ed) (2012) Green solvents. I, properties and application in chemistry. Springer, New York (eds: Inamuddin)
Modena M, Bates RB, Marvel CS (1965) Some low molecular weight polymers of D-limonene and related terpenes obtained by Ziegler-type catalyst. J Polym Sci A 3:949–960
Roberts WJ, Day AR (1950) Study of the polymerization of a-pinene and b-pinene with Friedal–Craft catalyst. J Am Chem Soc 72:1226–1230
Doiuchi T, Yamaguchi H, Minoura Y (1981) Cyclocopolymerization of d-limonene with maleic anhydride. Eur Polym J 17(9):961
Sharma S, Srivastava A (2004) Synthesis and characterization of copolymers of limonene with styrene initiated by azobisisobutyronitrile. Eur Polym J 9:2235–2240
Sharma S, Srivastava A (2003) Radical copolymerization of limonene with acrylonitrile: kinetics and mechanism. Polym Plast Technol Eng 3:485–502
Sharma S, Srivastava A (2003) Alternating copolymers of limonene with methyl methacrylate: kinetics and mechanism. J Appl Polym Sci 6:593–603
Zhang Y, Dube AM (2015) Copolymerization of 2-ethylhexyl acrylate and dlimonene, copolymerization of 2-ethylhexyl acrylate and D-limonene. Polym Plast Technol Eng 54(5):499–505
Kindermann N, Cristofol A, Kleij AW (2017) Access to biorenewable polycarbonates with unusual glass-transition temperature (T g) modulation. ACS Catal 7(6):3860–3863
Nendza M, Volmer J, Klein W, Kalcher N, Devillers J (eds) (1990) Risk assessment based on QSAR estimates. Kluwer Academic Publishers, Dordrecht, pp 213–240
Schultz TW (1997) Tetrahymena pyriformis population growth impairment endpointa surrogate for fish lethality. Toxicol Methods 7:289–309
Dimitrov SD, Mekenyan OG, Schultz TW (2000) Interspecies modeling of narcotics toxicity to aquatic animals. Bull Environ Contam Toxicol 65:399–406
Chattaraj PK, Nath S, Maiti B (2003) Reactivity descriptors. In: Tollenaere J, Bultinck P, Winter HD, Langenaeker W (eds) Computational medicinal chemistry for drug discovery, chap 11. Marcel Dekker, New York, pp 295–322
Parthasarathi R, Padmanabhan J, Subramanian V, Maiti B, Chattaraj PK (2003) Molecular structure, reactivity, and toxicity of the complete series of chlorinated benzenes. J Phys Chem A 107:13046
Parthasarathi R, Padmanabhan J, Subramanian V, Maiti B, Chattaraj PK (2004) Toxicity analysis of 33′44′5-pentachloro biphenyl through chemical reactivity and selectivity profiles. Curr Sci 86:535–542
Padmanabhan J, Parthasarathi R, Sarkar U, Subramanian V, Chattaraj PK (2004) Effect of solvation on the condensed Fukui function and the generalized philicity index. Chem Phys Lett 383(1–2):122–128
Roy RK, De Proft F, Geerlings P (1998) Site of protonation in aniline and substituted anilines in the gas phase: a study via the local hard and soft acids and bases concept. J Phys Chem A 102:7035
Mendez F, Tamariz J, Geerlings P (1998) 1, 3-dipolar cycloaddition reactions: a DFT and HSAB principle theoretical model. J Phys Chem A 102:6292
Langenaeker W, De Proft F, Geerlings P (1998) Ab initio and density functional theory study of the geometry and reactivity of benzyne, 3-fluorobenzyne, 4-fluorobenzyne, and 4, 5-didehydropyrimidine. J Phys Chem A 102:5944
Roy RK, Krishnamurthy S, Geerlings P, Pal S (1998) Local softness and hardness based reactivity descriptors for predicting intra-and intermolecular reactivity sequences: carbonyl compounds. J Phys Chem A 102:3746
Chatterjee A, Iwasakı T, Ebina T (1999) Reactivity index scale for interaction of heteroatomic molecules with zeolite framework. J Phys Chem A 103:2489
Perez P, Toro-Labbe A, Contreras R (1999) HSAB analysis of charge transfer in the gas-phase acid–base equilibria of alkyl-substituted alcohols. J Phys Chem A 103:11246
Jaque P, Toro-Labbe A (2000) Theoretical study of the double proton transfer in the CHX–XH⊙⊙⊙ CHX–XH (X = O, S) complexes. J Phys Chem A 104:995
Perez P, Toro-Labbe A, Contreras R (2000) Global and local analysis of the gas-phase acidity of haloacetic acids. J Phys Chem A 104:5882
Gutıerrez-Oliva S, Jaque P, Toro-Labbe A (2000) Using Sanderson’s principle to estimate global electronic properties and bond energies of hydrogen-bonded complexes. J Phys Chem A 104:8955
Parthasarathi R, Padmanabhan J, Sarkar U, Maiti B, Subramanian V, Chattaraj PK (2003) Toxicity analysis of benzidine through chemical reactivity and selectivity profiles: a DFT approach. Internet Electron J Mol Des 2:798–813
Roy DR, Parthasarathi R, Maiti B, Subramanian V, Chattaraj PK (2005) Electrophilicity as a possible descriptor for toxicity prediction. Bioorg Med Chem 13:3405–3412
Kloosterboer JG (1988) Network formation by chain crosslinking photopolymerization and its applications in electronics. Adv Polym Sci 84:1
Matyjaszewski K, Gnanou Y, Leibler L (2007) Macromolecular engineering: precise synthesis, materials properties, applications. Wiley, Weinheim
Ljubic TS, Pahovnik D, Žigon M, Žagar E (2012) Photochemically active systems and probes for polymer research. Sci World J 2012:1
Acikbas Y, Cankaya N, Capan R, Erdogan M, Soykan C (2016) Swelling behaviour of the 2-(4-methoxyphenylamino)-2-oxoethyl methacrylate monomer LB thin film exposed to various organic vapours by quartz crystal microbalance technique. J Macromol Sci A Pure Appl Chem 53(1):18–25
Gülbaş HE, Çankaya N (2017) D-Limonen İle 2-(4-Metoksifenilamino)-2-Oksoetil Metakrilat (MPAEMA) Kopolimerinin (Limonen-co-MPAEMA) Sentezi ve Karakterizasyonu. In: 2nd international congress on engineering architecture and design, oral presentation, Kocaeli-Turkey, pp 749–750
Kohn W, Sham LJ (1965) Self-consistent equations including exchange and correlation effects. Phys Rev 140:A1133–A1138
Becke AD (1988) Density-functional exchange-energy approximation with correct asymptotic behavior. Phys Rev A 38:3098–3100
Vosko SH, Vilk L, Nusair M (1980) Accurate spin-dependent electron liquid correlation energies for local spin density calculations: a critical analysis. Can J Phys 58:1200–1211
Lee C, Yang W, Parr RG (1988) Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789
Frisch MJ et al (2009) Gaussian 09, revision A.1. Gaussian, Inc., Wallingford
Jamroz MH (2013) Vibrational energy distribution analysis (VEDA): scopes and limitations. Spectrochim Acta A Mol Biomol Spectrosc 114:220–230
Parr RG, Pearson RG (1983) Absolute hardness: companion parameter to absolute electronegativity. J Am Chem Soc 105:7512–7516
Ditchfield R (1972) Molecular orbital theory of magnetic shielding and magnetic susceptibility. J Chem Phys 56:5688–5691
Wolinski K, Hinton JF, Pulay P (1990) Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations. J Am Chem Soc 112:8251–8260
O’Boyle NM, Tenderholt AL, Langner KM (2008) Cclib: a library for package‐independent computational chemistry algorithms. J Comput Chem 29:839–845
Abraham CS, Prasana JC, Muthu S (2017) Quantum mechanical, spectroscopic and docking studies of 2-amino-3-bromo-5-nitropyridine by density functional method. Spectrochim Acta A 181:153–163
Socrates G (2001) Infrared and Raman characteristics group frequencies, tables and charts, 3rd edn. Wiley, Chichester
Sundaraganesan N, Illakiamani S, Meganathan C, Joshua BD (2007) Vibrational spectroscopy investigation using ab initio and density functional theory analysis on the structure of 3-aminobenzotrifluoride. Spectrochim Acta A 67:214–224
Fukui K (1982) Role of frontier orbitals in chemical reactions. Science 218:747–754
Tanış E, Babur Şaş E, Kurban M, Kurt M (2017) The structural, electronic and spectroscopic properties of 4FPBAPE molecule: experimental and theoretical study. J Mol Struct 1154:301–318
Hughbanks T, Hoffmann R (1983) Chains of trans-edge-sharing molybdenum octahedra: metal–metal bonding in extended systems. J Am Chem Soc 105:3528–3537
Małecki JG (2010) Synthesis, crystal, molecular and electronic structures of thiocyanate ruthenium complexes with pyridine and its derivatives as ligands. Polyhedron 29:1973–1979
Chen M, Waghmare UV, Friend CM, Kaxiras E (1998) A density functional study of clean and hydrogen-covered α-MoO3(010):α-MoO3(010): electronic structure and surface relaxation. J Chem Phys 109:6854–6860
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cankaya, N., Tanış, E., Gülbaş, H.E. et al. A new synthesis of limonene copolymer: experimental and theoretical analysis. Polym. Bull. 76, 3297–3327 (2019). https://doi.org/10.1007/s00289-018-2543-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-018-2543-3