Abstract.
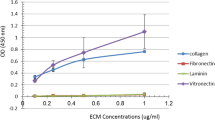
In this study, 13 bifidobacterial strains were tested for their ability to adhere to immobilized extracellular matrix (ECM) proteins. Only two Bifidobacterium adolescentis strains adhered to immobilized type I and type V collagens, but not to laminin, fibronectin, and type III and IV collagens. The adhesion of B. adolescentis BB-119 to type V collagen was inhibited by type I and V collagens and gelatin, and was diminished after protease treatment of the cells. Periodate treatment of immobilized collagen and the presence of galactose inhibited the adhesion of strain BB-119 to type V collagen. Two cell surface proteins with molecular masses of 36 kDa and 52 kDa from strain BB-119 were found to bind to horseradish peroxidase-conjugated type V collagen by ligand blotting. We concluded that B. adolescentis BB-119 binds to type V collagen at galactose chains as target via these two cell surface proteins by their lectin-like activity.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 15 October 1996 / Accepted: 20 November 1996
Rights and permissions
About this article
Cite this article
Mukai, T., Toba, T. & Ohori, H. Collagen Binding of Bifidobacterium adolescentis . Curr Microbiol 34, 326–331 (1997). https://doi.org/10.1007/s002849900190
Issue Date:
DOI: https://doi.org/10.1007/s002849900190