Abstract
Background
The association of chemotherapy-induced myelosuppression with tumor response and overall survival remained controversial. The study was conducted to investigate the association between them in small cell lung cancer (SCLC).
Methods
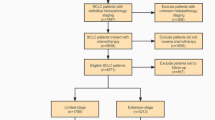
204 eligible patients with SCLC were respectively included and categorized into three groups (no, mild, and severe myelosuppression) based on myelosuppression degree after the first chemotherapy. Curative efficacy of 2-cycle chemotherapy was evaluated by the objective response rate (ORR) and disease control rate (DCR). Univariate and multivariate logistic regression analyses were conducted to investigate their association. Receiver operator characteristic (ROC) curves, net reclassification index (NRI), and integrated discrimination improvement (IDI) were used to assess the predictive ability of myelosuppression.
Results
In the fully-adjusted model, mild (OR, 4.61; 95% CI, 1.35 to 18.27; P = 0.020) and severe (OR, 7.22; 95% CI, 1.30 to 72.44; P = 0.046) myelosuppression were positively associated with DCR. However, only mild myelosuppression was significantly associated with ORR (OR, 2.78; 95% CI, 1.30 to 6.14; P = 0.010). Although we observed evidence of increased ORR in severe myelosuppression, the difference was not statistically significant. Furthermore, based on the results of the ROC curve, NRI and IDI, chemotherapy-induced myelosuppression cannot be used as a accurate and independent predictor for curative efficacy, but it can improve overall prediction accuracy.
Conclusion
Chemotherapy-induced myelosuppression was significantly associated with curative efficacy of 2-cycle chemotherapy in SCLC, which could help predict treatment efficacy and guide chemotherapy dosage.


Similar content being viewed by others
Data availability
The raw data supporting the findings of this study are available upon reasonable request by contact with the corresponding author.
Abbreviations
- SCLC:
-
Small cell lung cancer
- ECOG PS:
-
Cooperative Oncology Group Performance Status
- NCCN:
-
National Comprehensive Cancer Network
- ORR:
-
Objective response rate
- DCR:
-
Disease control rate
- RECIST:
-
Response Evaluation Criteria in Solid Tumors
- CR:
-
Complete response
- PR:
-
Partial response
- SD:
-
Stable disease
- PD:
-
Progressive disease
- OR:
-
Odds ratio
- CI:
-
Confidence interval
- ROC:
-
Receiver operator characteristic
- AUC:
-
Area under the curve
- NRI:
-
Net reclassification index
- IDI:
-
Integrated discrimination improvement
- DNA:
-
Deoxyribonucleic acid
References
Sung H, Ferlay J, Siegel RL et al (2021) Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/caac.21660
Siegel RL, Miller KD, Fuchs HE, Jemal A (2021) Cancer statistics, 2021. CA Cancer J Clin 71:7–33. https://doi.org/10.3322/caac.21654
Sun A, Durocher-Allen LD, Ellis PM et al (2019) Initial management of small-cell lung cancer (limited- and extensive-stage) and the role of thoracic radiotherapy and first-line chemotherapy: a systematic review. Curr Oncol 26:e372–e384. https://doi.org/10.3747/co.26.4481
Jiang S, Huang L, Zhen H et al (2021) Carboplatin versus cisplatin in combination with etoposide in the first-line treatment of small cell lung cancer: a pooled analysis. BMC Cancer 21:1308. https://doi.org/10.1186/s12885-021-09034-6
Rossi A, Di Maio M, Chiodini P et al (2012) Carboplatin- or cisplatin-based chemotherapy in first-line treatment of small-cell lung cancer: the COCIS meta-analysis of individual patient data. J Clin Oncol 30:1692–1698. https://doi.org/10.1200/JCO.2011.40.4905
Käsmann L, Eze C, Taugner J et al (2020) Chemoradioimmunotherapy of inoperable stage III non-small cell lung cancer: immunological rationale and current clinical trials establishing a novel multimodal strategy. Radiat Oncol 15:167. https://doi.org/10.1186/s13014-020-01595-3
Denduluri N, Patt DA, Wang Y et al (2015) Dose delays, dose reductions, and relative dose intensity in patients with cancer who received adjuvant or neoadjuvant chemotherapy in community oncology practices. J Natl Compr Cancer Netw 13:1383–1393. https://doi.org/10.6004/jnccn.2015.0166
Weycker D, Hatfield M, Grossman A et al (2019) Risk and consequences of chemotherapy-induced thrombocytopenia in US clinical practice. BMC Cancer 19:151. https://doi.org/10.1186/s12885-019-5354-5
Lim D, Kim HK, Jeong J-H et al (2020) Lipocalin2 induced by bacterial flagellin protects mice against cyclophosphamide mediated neutropenic sepsis. Microorganisms 8:646. https://doi.org/10.3390/microorganisms8050646
Shitara K, Matsuo K, Takahari D et al (2009) Neutropaenia as a prognostic factor in metastatic colorectal cancer patients undergoing chemotherapy with first-line FOLFOX. Eur J Cancer 45:1757–1763. https://doi.org/10.1016/j.ejca.2009.01.019
Rocconi RP, Matthews KS, Kemper MK et al (2008) Chemotherapy-related myelosuppression as a marker of survival in epithelial ovarian cancer patients. Gynecol Oncol 108:336–341. https://doi.org/10.1016/j.ygyno.2007.10.012
Yamanaka T, Matsumoto S, Teramukai S et al (2007) Predictive value of chemotherapy-induced neutropenia for the efficacy of oral fluoropyrimidine S-1 in advanced gastric carcinoma. Br J Cancer 97:37–42. https://doi.org/10.1038/sj.bjc.6603831
Banerji U, Ashley S, Coward J et al (2006) The association of chemotherapy induced neutropenia on treatment outcomes in small cell lung cancer. Lung Cancer 54:371–377. https://doi.org/10.1016/j.lungcan.2006.08.001
Yashiki C, Hirose T, Sugiyama T et al (2010) Chemotherapy-induced myelosuppression and treatment efficacy in limited-stage disease small cell lung cancer. Gan To Kagaku Ryoho 37:245–249
Zhu K, Jiang M, Xu Y et al (2021) Curative efficacy might be an early predictor of prognosis in patients with small cell lung cancer treated with 2 cycles of platinum-based first-line chemotherapy. J Thorac Dis 13:1205–1214. https://doi.org/10.21037/jtd-21-216
Ganti AKP, Loo BW, Bassetti M et al (2021) Small cell lung cancer, version 2.2022, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw 19:1441–1464. https://doi.org/10.6004/jnccn.2021.0058
Miller AB, Hoogstraten B, Staquet M, Winkler A (1981) Reporting results of cancer treatment. Cancer 47:207–214. https://doi.org/10.1002/1097-0142(19810101)47:1%3c207::aid-cncr2820470134%3e3.0.co;2-6
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247. https://doi.org/10.1016/j.ejca.2008.10.026
Blazek K, van Zwieten A, Saglimbene V, Teixeira-Pinto A (2021) A practical guide to multiple imputation of missing data in nephrology. Kidney Int 99:68–74. https://doi.org/10.1016/j.kint.2020.07.035
Liu X, Zhang W, Yin W et al (2017) The prognostic value of the serum neuron specific enolase and lactate dehydrogenase in small cell lung cancer patients receiving first-line platinum-based chemotherapy. Medicine (Baltimore) 96:e8258. https://doi.org/10.1097/MD.0000000000008258
Steyerberg EW, Vickers AJ, Cook NR et al (2010) Assessing the performance of prediction models: a framework for traditional and novel measures. Epidemiology 21:128–138. https://doi.org/10.1097/EDE.0b013e3181c30fb2
Minami S, Ogata Y, Ihara S et al (2016) Retrospective analysis of outcomes and prognostic factors of chemotherapy for small-cell lung cancer. Lung Cancer (Auckl) 7:35–44. https://doi.org/10.2147/LCTT.S100184
Moser SS, Bar J, Kan I et al (2021) Real world analysis of small cell lung cancer patients: prognostic factors and treatment outcomes. Curr Oncol 28:317–331. https://doi.org/10.3390/curroncol28010036
Johnstone TC, Suntharalingam K, Lippard SJ (2016) The next generation of platinum drugs: targeted Pt(II) agents, nanoparticle delivery, and Pt(IV) prodrugs. Chem Rev 116:3436–3486. https://doi.org/10.1021/acs.chemrev.5b00597
Amable L (2016) Cisplatin resistance and opportunities for precision medicine. Pharmacol Res 106:27–36. https://doi.org/10.1016/j.phrs.2016.01.001
Montecucco A, Zanetta F, Biamonti G (2015) Molecular mechanisms of etoposide. EXCLI J 14:95–108. https://doi.org/10.17179/excli2015-561
Oun R, Moussa YE, Wheate NJ (2018) The side effects of platinum-based chemotherapy drugs: a review for chemists. Dalton Trans 47:6645–6653. https://doi.org/10.1039/c8dt00838h
Jiang C, Hu X, Wang L et al (2015) Excessive proliferation and impaired function of primitive hematopoietic cells in bone marrow due to senescence post chemotherapy in a T cell acute lymphoblastic leukemia model. J Transl Med 13:234. https://doi.org/10.1186/s12967-015-0543-8
Han J, Xia J, Zhang L et al (2019) Studies of the effects and mechanisms of ginsenoside Re and Rk3 on myelosuppression induced by cyclophosphamide. J Ginseng Res 43:618–624. https://doi.org/10.1016/j.jgr.2018.07.009
Săftescu S, Popovici D, Oprean C et al (2020) Endurance of erythrocyte series in chemotherapy. Exp Ther Med 20:214. https://doi.org/10.3892/etm.2020.9344
Hande KR (1992) Etoposide pharmacology. Semin Oncol 19:3–9
Donahue A, McCune JS, Faucette S et al (2001) Measured versus estimated glomerular filtration rate in the Calvert equation: influence on carboplatin dosing. Cancer Chemother Pharmacol 47:373–379. https://doi.org/10.1007/s002800000260
Wang L-Y, Cui J-J, Liu J-Y et al (2018) Gene-gene and gene-environment interaction data for platinum-based chemotherapy in non-small cell lung cancer. Sci Data 5:180284. https://doi.org/10.1038/sdata.2018.284
Wang Y, Yin J-Y, Li X-P et al (2014) The association of transporter genes polymorphisms and lung cancer chemotherapy response. PLoS ONE 9:e91967. https://doi.org/10.1371/journal.pone.0091967
Pencina MJ, D’Agostino RB, Demler OV (2012) Novel metrics for evaluating improvement in discrimination: net reclassification and integrated discrimination improvement for normal variables and nested models. Stat Med 31:101–113. https://doi.org/10.1002/sim.4348
Funding
This work was sponsored by the General Project of Jiangsu Provincial Health Commission (No. H2019029), the Jiangsu Province Six One Project (No. LGY2018054), the “Six talent peaks” high-level talents level B (No. WSN-015), and the 333 High-level personnel Training Program.
Author information
Authors and Affiliations
Contributions
XW: Conceptualization, Methodology, Software, Writing—Original Draft; SG: Software, Validation, Writing—Review and Editing; JW: Resources, Validation, Writing—Review and Editing; LZ: Data Curation, Writing—Review and Editing; XQ: Conceptualization, Writing—Review and Editing, Project administration, Project administration.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships.
Studies involving animal subjects
No animal studies are presented in this manuscript.
Studies involving human subjects
The Ethics Committee of The First Affiliated Hospital of Nanjing Medical University reviewed and approved the cross-sectional study (2023-SR-365). The work described has been carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans.
Inclusion of identifiable human data
No potentially identifiable human images or data is presented in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., Gu, S., Wen, J. et al. Association between chemotherapy-induced myelosuppression and curative efficacy of 2-cycle chemotherapy in small cell lung cancer. Cancer Chemother Pharmacol 93, 151–159 (2024). https://doi.org/10.1007/s00280-023-04608-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-023-04608-6