Abstract
Purpose
To investigate the efficacy and toxicity of the docetaxel and capecitabine combination in patients with previously treated, unresectable adenocarcinoma of the pancreas.
Patients and Methods
Patients with pancreatic adenocarcinoma, pre-treated with gemcitabine-based chemotherapy, were treated with capecitabine (800 mg/m2 orally, twice a day for 14 days) and docetaxel (75 mg/m2 i.v, on day1), every 3 weeks. The primary end-point was overall response rate (RR).
Results
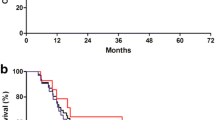
Thirty-one patients were enrolled in the study; 93.6% of them had a performance status (PS) of 0–1 and 96.8% had stage IV disease. Patients received a median of 4 cycles/patient, and the main reason for treatment discontinuation was disease progression. Partial response was observed in three (9.7%) patients, stable disease in seven (22.6%) (disease control rate: 32.3%, 95% CI: 15.80–48.71%) and disease progression in 21 (67.6%). The median progression-free survival (PFS) was 2.4 months (95% CI: 1.6–3.13) and the median overall survival (OS) was 6.3 months (95% CI: 3.38–9.23); the estimated 1-year survival rate was 14.7%. Grade III/IV neutropenia occurred in 10 (32.2%) patients and febrile neutropenia in one patient. Other severe non-hematologic toxicities were mild and manageable. After 2 chemotherapy cycles, pain control occurred in 20% of patients and stabilization of body weight in 40%.
Conclusion
The combination of docetaxel/capecitabine may confer good disease control associated with improvement of quality of life as second-line chemotherapy in patients with metastatic pancreatic cancer.


Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ (2009) Cancer statistics, 2009. CA Cancer J Clin. 59:225–249
Saif MW (2007) Pancreatic cancer: is this bleak landscape finally changing? Highlights from the ‘43rd ASCO Annual Meeting’. Chicago 8:365–373
Poplin E, Feng Y, Berlin J et al (2009) Phase III, randomized study of gemcitabine and oxaliplatin versus gemcitabine (fixed-dose rate infusion) compared with gemcitabine (30-minute infusion) in patients with pancreatic Carcinoma E6201: a trial of the eastern cooperative oncology group. J Clin Oncol 27:3778–3785
Louvet C, Labianca R, Hammel P et al (2005) Gemcitabine in combination with oxaliplatin compared with gemcitabine alone in locally advanced or metastatic pancreatic cancer: results of a GERCOR and GISCAD phase III trial. J Clin Oncol 23:3509–3516
Heinemann V, Quietzsch D, Gieseler FM et al (2006) Randomized phase III trial of gemcitabine plus cisplatin compared with gemcitabine alone in advanced pancreatic cancer. J Clin Oncol 24:3946–3952
Herrmann C, Abel U, Stremmel W, Jaeger D, Herrmann T (2007) Short time to progression under first-line chemotherapy is a negative prognostic factor for time to progression and residual survival under second-line chemotherapy in advanced pancreatic cancer. Oncology 73:335–339
Boeck S, Wilkowski R, Bruns CJ et al (2007) Oral capecitabine in gemcitabine-pretreated patients with advanced pancreatic cancer. Oncology 73:221–227
Cartwright TH, Cohn A, Varkey JA et al (2002) Phase II study of oral capecitabine in patients with advanced or metastatic pancreatic cancer. J Clin Oncol 20:160–164
Boeck S, Hoehler T, Seipelt G et al (2008) Capecitabine plus oxaliplatin (CapOx) versus capecitabine plus gemcitabine (CapGem) versus gemcitabine plus oxaliplatin (mGemOx): final results of a multicenter randomized phase II trial in advanced pancreatic cancer. Ann Oncol 19:340–347
Xiong HQ, Varadhachary GR, Blais JC, Hess KR, Abbruzzese JL, Wolff RA (2008) Phase 2 trial of oxaliplatin plus capecitabine (XELOX) as second-line therapy for patients with advanced pancreatic cancer. Cancer 113:2046–2052
Rougier P, Adenis A, Ducreux M et al (2000) A phase II study: docetaxel as first-line chemotherapy for advanced pancreatic adenocarcinoma. Eur J Cancer 36:1016–1025
Pronk LC, Vasey P, Sparreboom A et al (2000) A phase I and pharmacokinetic study of the combination of capecitabine and docetaxel in patients with advanced solid tumours. Br J Cancer 83:22–29
Rosati G, Bilancia D, Germano D et al (2007) Reduced dose intensity of docetaxel plus capecitabine as second-line palliative chemotherapy in patients with metastatic gastric cancer: a phase II study. Ann Oncol 18(Suppl 6):128–132
O’Shaughnessy J, Miles D, Vukelja S et al (2002) Superior survival with capecitabine plus docetaxel combination therapy in anthracycline-pretreated patients with advanced breast cancer: phase III trial results. J Clin Oncol 20:2812–2823
Blaya M LG, Roman E Jr et al (2007) Phase II trial of capecitabine and docetaxel as second line therapy for locally advanced and metastatic pancreatic cancer. J Clin Oncol ASCO Annual Meeting Proceedings (Post-Meeting Edition) 25(18S): 15029
van Persijn van Meerten EL, Gelderblom H, Bloem JL (2009) RECIST revised: implications for the radiologist. A review article on the modified RECIST guideline. European Radiology (in press)
Cancer Therapy Evaluation Program (1999) Common Toxicity Criteria. Version 2.0. DCTD, NCI, NIH, DHHS
Lutz MP, Van Cutsem E, Wagener T et al (2005) Docetaxel plus gemcitabine or docetaxel plus cisplatin in advanced pancreatic carcinoma: randomized phase II study 40984 of the European Organisation for Research and Treatment of Cancer Gastrointestinal Group. J Clin Oncol 23:9250–9256
Kulke MH, Blaszkowsky LS, Ryan DP et al (2007) Capecitabine plus erlotinib in gemcitabine-refractory advanced pancreatic cancer. J Clin Oncol 25:4787–4792
National Comprehensive Cancer Network I (2009) Practice Guidelines in Oncology. Pancreatic Adenocarcinoma. v.1
KC TsavarisN, Skopelitis H, Gouveris P et al (2005) Second line treatment with Oxaliplatin, leucovorin and 5-fluiriuracil in gemcitabine-pretreated advanced pancreatic cancer: a phase II study. Invest New Drugs 23:369–375
Gebbia V, Maiello E, Giuliani F, Borsellino N et al (2007) Second-line chemotherapy in advanced pancreatic carcinoma: a multicenter survey of the Gruppo Oncologico Italia Meridionale on the activity and safety of the FOLFOX4 regimen in clinical practice. Ann Oncology 18(Suppl 6):124–127
Mitry E, Ducreux M, Ould-Kaci M, Boige V et al (2006) Oxaliplatin combined with 5FU in second line treatment of advanced pancreatic adenocarcinoma. Results of a phase II trial. Gastroenterol Clin Biol 30:357–363
Burris HA 3rd, Rivkin S, Reynolds R et al (2005) Phase II trial of oral rubitecan in previously treated pancreatic cancer patients. The Oncologist 10:183–190
Boeck S, Weigang-Kohler K, Fuchs M et al (2007) Second-line chemotherapy with pemetrexed after gemcitabine failure in patients with advanced pancreatic cancer: a multicenter phase II trial. Ann Oncol 18:745–751
Oettle H, Arnold D, Esser M, Huhn D, Riess H (2000) Paclitaxel as weekly second-line therapy in patients with advanced pancreatic carcinoma. Anti-cancer Drugs 11:635–638
Morizane C, Okusaka T, Furuse J et al (2009) A phase II study of S-1 in gemcitabine-refractory metastatic pancreatic cancer. Cancer Chemother Pharmacol 63:313–319
Demols A, Peeters M, Polus M et al (2006) Gemcitabine and oxaliplatin (GEMOX) in gemcitabine refractory advanced pancreatic adenocarcinoma: a phase II study. Br J Cancer 94:481–485
Gasent Blesa J, Alberola Candel V, Giner Marco V et al (2009) E. Phase II trial of second-line chemotherapy in metastatic cancer of the pancreas with the combination of oxaliplatin (Ox) and capecitabine (Cp). J Clin Oncol 27 (abstract 15561)
Cantore M, Rabbi C, Fiorentini G et al (2004) Combined irinotecan and oxaliplatin in patients with advanced pre-treated pancreatic cancer. Oncology 67:93–97
Mazzer M, Zanon E, Foltran L et al (2009) Second-line pemetrexed-oxaliplatin for advanced pancreatic adenocarcinoma. J Clin Oncol 27 (abstract 15597)
Reni M, Pasetto L, Aprile G et al (2006) Raltitrexed-eloxatin salvage chemotherapy in gemcitabine-resistant metastatic pancreatic cancer. Br J Cancer 94:785–791
Pelzer U, Kubica K, Stieler J et al (2008) A randomized trial in patients with gemcitabine refractory pancreatic cancer. Final results of the CONKO 003 study. ASCO Meeting Abstracts no 4508
Stathopoulos GP, Boulikas T, Vougiouka M, Rigatos SK, Stathopoulos JG (2006) Liposomal cisplatin combined with gemcitabine in pretreated advanced pancreatic cancer patients: a phase I-II study. Oncol Rep 15:1201–1204
Kozuch P, Grossbard ML, Barzdins A et al (2001) Irinotecan combined with gemcitabine, 5-fluorouracil, leucovorin, and cisplatin (G-FLIP) is an effective and noncrossresistant treatment for chemotherapy refractory metastatic pancreatic cancer. The Oncologist 6:488–495
Fine RL, Fogelman DR, Schreibman SM et al (2008) The gemcitabine, docetaxel, and capecitabine (GTX) regimen for metastatic pancreatic cancer: a retrospective analysis. Cancer Chemother Pharmacol 61:167–175
Acknowledgments
This work was partly supported by a research grant from the Cretan Association for Biomedical Research (CABR).
Conflict of interest statement
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Katopodis, O., Polyzos, A., Kentepozidis, N. et al. Second-line chemotherapy with Capecitabine (Xeloda) and Docetaxel (Taxotere) in previously treated, unresectable adenocarcinoma of pancreas: the final results of a phase II trial. Cancer Chemother Pharmacol 67, 361–368 (2011). https://doi.org/10.1007/s00280-010-1329-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-010-1329-6