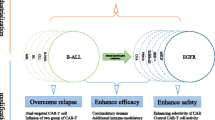
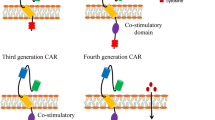
Abstract
Although CAR-T cell therapy has been particularly successful as a treatment for B cell malignancies, effectively treating acute myeloid leukemia with CAR remains a greater challenge. Multiple preclinical studies and clinical trials are underway, including on AML-related surface markers that CAR-T cells can target, such as CD123, CD33, NKG2D, CLL1, CD7, FLT3, Lewis Y and CD70, all of which provide opportunities for developing CAR-T therapies with improved specificity and efficacy. We also explored specific strategies for CAR-T cell treatment of AML, including immune checkpoints, suicide genes, dual targeting, genomic tools and the potential for universal CAR. In addition, CAR-T cell therapy for AML still has certain risks and challenges, including cytokine release syndrome (CRS) and haematotoxicity. Despite these challenges, as a new targeting method for AML treatment, CAR-T cell therapy still has great prospects. Ongoing research aims to further optimize this treatment mode.

Similar content being viewed by others
Data availablity
Data openly available in a public repository.
References
Vishwasrao P, Li G, Boucher JC, Smith DL, Hui SK (2022) Emerging CAR T cell strategies for the treatment of AML. Cancers 14(5). https://doi.org/10.3390/cancers14051241
Schultz LM, Eaton A, Baggott C, Rossoff J, Prabhu S, Keating AK, Krupski C, Pacenta H, Philips CL, Talano JA, Moskop A, Baumeister SHC, Myers GD, Karras NA, Brown PA, Qayed M, Hermiston M, Satwani P, Wilcox R et al (2023) Outcomes after nonresponse and relapse post-tisagenlecleucel in children, adolescents, and young adults with B-cell acute lymphoblastic leukemia. J Clin Oncol 41(2):354–363. https://doi.org/10.1200/jco.22.01076
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, Braunschweig I, Oluwole OO, Siddiqi T, Lin Y, Timmerman JM, Stiff PJ, Friedberg JW, Flinn IW, Goy A, Hill BT, Smith MR, Deol A, Farooq U et al (2017) Axicabtagene Ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med 377(26):2531–2544. https://doi.org/10.1056/NEJMoa1707447
Frey NV (2022) Approval of brexucabtagene autoleucel for adults with relapsed and refractory acute lymphocytic leukemia. Blood 140(1):11–15. https://doi.org/10.1182/blood.2021014892
Kamdar M, Solomon SR, Arnason J, Johnston PB, Glass B, Bachanova V, Ibrahimi S, Mielke S, Mutsaers P, Hernandez-Ilizaliturri F, Izutsu K, Morschhauser F, Lunning M, Maloney DG, Crotta A, Montheard S, Previtali A, Stepan L, Ogasawara K et al (2022) Lisocabtagene maraleucel versus standard of care with salvage chemotherapy followed by autologous stem cell transplantation as second-line treatment in patients with relapsed or refractory large B-cell lymphoma (TRANSFORM): results from an interim analysis of an open-label, randomised, phase 3 trial. Lancet (London, England) 399(10343):2294–2308. https://doi.org/10.1016/s0140-6736(22)00662-6
Hansen DK, Sidana S, Peres LC, Colin Leitzinger C, Shune L, Shrewsbury A, Gonzalez R, Sborov DW, Wagner C, Dima D, Hashmi H, Kocoglu MH, Atrash S, Simmons G, Kalariya N, Ferreri C, Afrough A, Kansagra A, Voorhees P et al (2023) Idecabtagene vicleucel for relapsed/refractory multiple myeloma: real-world experience from the Myeloma CAR T Consortium. J Clin Oncol 41(11):2087–2097. https://doi.org/10.1200/jco.22.01365
Martin T, Usmani SZ, Berdeja JG, Agha M, Cohen AD, Hari P, Avigan D, Deol A, Htut M, Lesokhin A, Munshi NC, O'Donnell E, Stewart AK, Schecter JM, Goldberg JD, Jackson CC, Yeh TM, Banerjee A, Allred A et al (2023) Ciltacabtagene autoleucel, an anti-B-cell maturation antigen chimeric antigen receptor T-cell therapy, for relapsed/refractory multiple myeloma: CARTITUDE-1 2-year follow-up. J Clin Oncol 41(6):1265–1274. https://doi.org/10.1200/jco.22.00842
Perna F, Berman SH, Soni RK, Mansilla-Soto J, Eyquem J, Hamieh M, Hendrickson RC, Brennan CW, Sadelain M (2017) Integrating proteomics and transcriptomics for systematic combinatorial chimeric antigen receptor therapy of AML. Cancer Cell 32(4):506–519.e505. https://doi.org/10.1016/j.ccell.2017.09.004
Wang QS, Wang Y, Lv HY, Han QW, Fan H, Guo B, Wang LL, Han WD (2015) Treatment of CD33-directed chimeric antigen receptor-modified T cells in one patient with relapsed and refractory acute myeloid leukemia. Mol Ther : J Am Soc Gene Ther 23(1):184–191. https://doi.org/10.1038/mt.2014.164
Tambaro FP, Singh H, Jones E, Rytting M, Mahadeo KM, Thompson P, Daver N, DiNardo C, Kadia T, Garcia-Manero G, Chan T, Shah RR, Wierda WG (2021) Autologous CD33-CAR-T cells for treatment of relapsed/refractory acute myelogenous leukemia. Leukemia 35(11):3282–3286. https://doi.org/10.1038/s41375-021-01232-2
Jordan CT, Upchurch D, Szilvassy SJ, Guzman ML, Howard DS, Pettigrew AL, Meyerrose T, Rossi R, Grimes B, Rizzieri DA, Luger SM, Phillips GL (2000) The interleukin-3 receptor alpha chain is a unique marker for human acute myelogenous leukemia stem cells. Leukemia 14(10):1777–1784. https://doi.org/10.1038/sj.leu.2401903
Jin L, Lee EM, Ramshaw HS, Busfield SJ, Peoppl AG, Wilkinson L, Guthridge MA, Thomas D, Barry EF, Boyd A, Gearing DP, Vairo G, Lopez AF, Dick JE, Lock RB (2009) Monoclonal antibody-mediated targeting of CD123, IL-3 receptor alpha chain, eliminates human acute myeloid leukemic stem cells. Cell Stem Cell 5(1):31–42. https://doi.org/10.1016/j.stem.2009.04.018
Rongvaux A, Takizawa H, Strowig T, Willinger T, Eynon EE, Flavell RA, Manz MG (2013) Human hemato-lymphoid system mice: current use and future potential for medicine. Annu Rev Immunol 31:635–674. https://doi.org/10.1146/annurev-immunol-032712-095921
Uy GL, Aldoss I, Foster MC, Sayre PH, Wieduwilt MJ, Advani AS, Godwin JE, Arellano ML, Sweet KL, Emadi A, Ravandi F, Erba HP, Byrne M, Michaelis L, Topp MS, Vey N, Ciceri F, Carrabba MG, Paolini S et al (2021) Flotetuzumab as salvage immunotherapy for refractory acute myeloid leukemia. Blood 137(6):751–762. https://doi.org/10.1182/blood.2020007732
Mardiros A, Dos Santos C, McDonald T, Brown CE, Wang X, Budde LE, Hoffman L, Aguilar B, Chang WC, Bretzlaff W, Chang B, Jonnalagadda M, Starr R, Ostberg JR, Jensen MC, Bhatia R, Forman SJ (2013) T cells expressing CD123-specific chimeric antigen receptors exhibit specific cytolytic effector functions and antitumor effects against human acute myeloid leukemia. Blood 122(18):3138–3148. https://doi.org/10.1182/blood-2012-12-474056
Gill S, Tasian SK, Ruella M, Shestova O, Li Y, Porter DL, Carroll M, Danet-Desnoyers G, Scholler J, Grupp SA, June CH, Kalos M (2014) Preclinical targeting of human acute myeloid leukemia and myeloablation using chimeric antigen receptor-modified T cells. Blood 123(15):2343–2354. https://doi.org/10.1182/blood-2013-09-529537
Laszlo GS, Estey EH, Walter RB (2014) The past and future of CD33 as therapeutic target in acute myeloid leukemia. Blood Rev 28(4):143–153. https://doi.org/10.1016/j.blre.2014.04.001
Walter RB, Gooley TA, van der Velden VH, Loken MR, van Dongen JJ, Flowers DA, Bernstein ID, Appelbaum FR (2007) CD33 expression and P-glycoprotein-mediated drug efflux inversely correlate and predict clinical outcome in patients with acute myeloid leukemia treated with gemtuzumab ozogamicin monotherapy. Blood 109(10):4168–4170. https://doi.org/10.1182/blood-2006-09-047399
Castaigne S, Pautas C, Terré C, Raffoux E, Bordessoule D, Bastie JN, Legrand O, Thomas X, Turlure P, Reman O, de Revel T, Gastaud L, de Gunzburg N, Contentin N, Henry E, Marolleau JP, Aljijakli A, Rousselot P, Fenaux P et al (2012) Effect of gemtuzumab ozogamicin on survival of adult patients with de-novo acute myeloid leukaemia (ALFA-0701): a randomised, open-label, phase 3 study. Lancet (London, England) 379(9825):1508–1516. https://doi.org/10.1016/s0140-6736(12)60485-1
Dutour A, Marin V, Pizzitola I, Valsesia-Wittmann S, Lee D, Yvon E, Finney H, Lawson A, Brenner M, Biondi A, Biagi E, Rousseau R (2012) In vitro and in vivo antitumor effect of anti-CD33 chimeric receptor-expressing EBV-CTL against CD33 acute myeloid leukemia. Adv Hematol 2012:683065. https://doi.org/10.1155/2012/683065
Marin V, Pizzitola I, Agostoni V, Attianese GM, Finney H, Lawson A, Pule M, Rousseau R, Biondi A, Biagi E (2010) Cytokine-induced killer cells for cell therapy of acute myeloid leukemia: improvement of their immune activity by expression of CD33-specific chimeric receptors. Haematologica 95(12):2144–2152. https://doi.org/10.3324/haematol.2010.026310
Barber A, Zhang T, DeMars LR, Conejo-Garcia J, Roby KF, Sentman CL (2007) Chimeric NKG2D receptor-bearing T cells as immunotherapy for ovarian cancer. Cancer Res 67(10):5003–5008. https://doi.org/10.1158/0008-5472.Can-06-4047
Salih HR, Antropius H, Gieseke F, Lutz SZ, Kanz L, Rammensee HG, Steinle A (2003) Functional expression and release of ligands for the activating immunoreceptor NKG2D in leukemia. Blood 102(4):1389–1396. https://doi.org/10.1182/blood-2003-01-0019
Spear P, Barber A, Rynda-Apple A, Sentman CL (2013) NKG2D CAR T-cell therapy inhibits the growth of NKG2D ligand heterogeneous tumors. Immunol Cell Biol 91(6):435–440. https://doi.org/10.1038/icb.2013.17
Spear P, Wu MR, Sentman ML, Sentman CL (2013) NKG2D ligands as therapeutic targets. Cancer Immun 13:8
Driouk L, Gicobi JK, Kamihara Y, Rutherford K, Dranoff G, Ritz J, Baumeister SHC (2020) Chimeric antigen receptor T cells targeting NKG2D-ligands show robust efficacy against acute myeloid leukemia and T-cell acute lymphoblastic leukemia. Front Immunol 11:580328. https://doi.org/10.3389/fimmu.2020.580328
Baumeister SH, Murad J, Werner L, Daley H, Trebeden-Negre H, Gicobi JK, Schmucker A, Reder J, Sentman CL, Gilham DE, Lehmann FF, Galinsky I, DiPietro H, Cummings K, Munshi NC, Stone RM, Neuberg DS, Soiffer R, Dranoff G et al (2019) Phase I trial of autologous CAR T cells targeting NKG2D ligands in patients with AML/MDS and multiple myeloma. Cancer Immunol Res 7(1):100–112. https://doi.org/10.1158/2326-6066.Cir-18-0307
Tashiro H, Sauer T, Shum T, Parikh K, Mamonkin M, Omer B, Rouce RH, Lulla P, Rooney CM, Gottschalk S, Brenner MK (2017) Treatment of acute myeloid leukemia with T cells expressing chimeric antigen receptors directed to C-type lectin-like molecule 1. Mol Ther :J Am Soc Gene Ther 25(9):2202–2213. https://doi.org/10.1016/j.ymthe.2017.05.024
Wang J, Chen S, Xiao W, Li W, Wang L, Yang S, Wang W, Xu L, Liao S, Liu W, Wang Y, Liu N, Zhang J, Xia X, Kang T, Chen G, Cai X, Yang H, Zhang X et al (2018) CAR-T cells targeting CLL-1 as an approach to treat acute myeloid leukemia. J Hematol Oncol 11(1):7. https://doi.org/10.1186/s13045-017-0553-5
Liu F, Cao Y, Pinz K, Ma Y, Wada M, Chen K, Ma G, Shen J, Tse CO, Su Y, Xiong Y, He G, Li Y, Ma Y (2018) First-in-Human CLL1-CD33 Compound CAR T cell therapy induces complete remission in patients with refractory acute myeloid leukemia: update on phase 1 clinical trial. Blood 132(Supplement 1):901–901. https://doi.org/10.1182/blood-2018-99-110579/JBlood
Lin G, Zhang Y, Yu L, Wu D (2021) Cytotoxic effect of CLL-1 CAR-T cell immunotherapy with PD-1 silencing on relapsed/refractory acute myeloid leukemia. Mol Med Rep 23:(3). https://doi.org/10.3892/mmr.2021.11847
Zhang H, Wang P, Li Z, He Y, Gan W, Jiang H (2021) Anti-CLL1 chimeric antigen receptor T-cell therapy in children with relapsed/refractory acute myeloid leukemia. Clin Cancer Res an official J Am Assoc Cancer Res 27(13):3549–3555. https://doi.org/10.1158/1078-0432.Ccr-20-4543
Tiftik N, Bolaman Z, Batun S, Ayyildiz O, Isikdogan A, Kadikoylu G, Muftuoglu E (2004) The importance of CD7 and CD56 antigens in acute leukaemias. Int J Clin Pract 58(2):149–152. https://doi.org/10.1111/j.1368-5031.2004.0018.x
Gomes-Silva D, Atilla E, Atilla PA, Mo F, Tashiro H, Srinivasan M, Lulla P, Rouce RH, Cabral JMS, Ramos CA, Brenner MK, Mamonkin M (2019) CD7 CAR T cells for the therapy of acute myeloid leukemia. Mol Ther : the J Am Soc Gene Ther 27(1):272–280. https://doi.org/10.1016/j.ymthe.2018.10.001
Levis M, Small D (2003) FLT3: ITDoes matter in leukemia. Leukemia 17(9):1738–1752. https://doi.org/10.1038/sj.leu.2403099
Perl AE, Martinelli G, Cortes JE, Neubauer A, Berman E, Paolini S, Montesinos P, Baer MR, Larson RA, Ustun C, Fabbiano F, Erba HP, Di Stasi A, Stuart R, Olin R, Kasner M, Ciceri F, Chou WC, Podoltsev N et al (2019) Gilteritinib or chemotherapy for relapsed or refractory FLT3-mutated AML. N Engl J Med 381(18):1728–1740. https://doi.org/10.1056/NEJMoa1902688
Stone RM, Mandrekar SJ, Sanford BL, Laumann K, Geyer S, Bloomfield CD, Thiede C, Prior TW, Döhner K, Marcucci G, Lo-Coco F, Klisovic RB, Wei A, Sierra J, Sanz MA, Brandwein JM, de Witte T, Niederwieser D, Appelbaum FR et al (2017) Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med 377(5):454–464. https://doi.org/10.1056/NEJMoa1614359
Cortes JE, Khaled S, Martinelli G, Perl AE, Ganguly S, Russell N, Krämer A, Dombret H, Hogge D, Jonas BA, Leung AY, Mehta P, Montesinos P, Radsak M, Sica S, Arunachalam M, Holmes M, Kobayashi K, Namuyinga R et al (2019) Quizartinib versus salvage chemotherapy in relapsed or refractory FLT3-ITD acute myeloid leukaemia (QuANTUM-R): a multicentre, randomised, controlled, open-label, phase 3 trial. Lancet Oncol 20(7):984–997. https://doi.org/10.1016/s1470-2045(19)30150-0
Chen L, Mao H, Zhang J, Chu J, Devine S, Caligiuri MA, Yu J (2017) Targeting FLT3 by chimeric antigen receptor T cells for the treatment of acute myeloid leukemia. Leukemia 31(8):1830–1834. https://doi.org/10.1038/leu.2017.147
Wang Y, Xu Y, Li S, Liu J, Xing Y, Xing H, Tian Z, Tang K, Rao Q, Wang M, Wang J (2018) Targeting FLT3 in acute myeloid leukemia using ligand-based chimeric antigen receptor-engineered T cells. J Hematol Oncol 11(1):60. https://doi.org/10.1186/s13045-018-0603-7
Riether C, Pabst T, Höpner S, Bacher U, Hinterbrandner M, Banz Y, Müller R, Manz MG, Gharib WH, Francisco D, Bruggmann R, van Rompaey L, Moshir M, Delahaye T, Gandini D, Erzeel E, Hultberg A, Fung S, de Haard H et al (2020) Targeting CD70 with cusatuzumab eliminates acute myeloid leukemia stem cells in patients treated with hypomethylating agents. Nat Med 26(9):1459–1467. https://doi.org/10.1038/s41591-020-0910-8
Riether C, Schürch CM, Bührer ED, Hinterbrandner M, Huguenin AL, Hoepner S, Zlobec I, Pabst T, Radpour R, Ochsenbein AF (2017) CD70/CD27 signaling promotes blast stemness and is a viable therapeutic target in acute myeloid leukemia. J Exp Med 214(2):359–380. https://doi.org/10.1084/jem.20152008
Sauer T, Parikh K, Sharma S, Omer B, Sedloev D, Chen Q, Angenendt L, Schliemann C, Schmitt M, Müller-Tidow C, Gottschalk S, Rooney CM (2021) CD70-specific CAR T cells have potent activity against acute myeloid leukemia without HSC toxicity. Blood 138(4):318–330. https://doi.org/10.1182/blood.2020008221
Westwood JA, Murray WK, Trivett M, Haynes NM, Solomon B, Mileshkin L, Ball D, Michael M, Burman A, Mayura-Guru P, Trapani JA, Peinert S, Hönemann D, Miles Prince H, Scott AM, Smyth MJ, Darcy PK, Kershaw MH (2009) The Lewis-Y carbohydrate antigen is expressed by many human tumors and can serve as a target for genetically redirected T cells despite the presence of soluble antigen in serum. J Immunother (Hagerstown, Md : 1997) 32(3):292–301. https://doi.org/10.1097/CJI.0b013e31819b7c8e
Peinert S, Prince HM, Guru PM, Kershaw MH, Smyth MJ, Trapani JA, Gambell P, Harrison S, Scott AM, Smyth FE, Darcy PK, Tainton K, Neeson P, Ritchie DS, Hönemann D (2010) Gene-modified T cells as immunotherapy for multiple myeloma and acute myeloid leukemia expressing the Lewis Y antigen. Gene Ther 17(5):678–686. https://doi.org/10.1038/gt.2010.21
Neeson P, Shin A, Tainton KM, Guru P, Prince HM, Harrison SJ, Peinert S, Smyth MJ, Trapani JA, Kershaw MH, Darcy PK, Ritchie DS (2010) Ex vivo culture of chimeric antigen receptor T cells generates functional CD8+ T cells with effector and central memory-like phenotype. Gene Ther 17(9):1105–1116. https://doi.org/10.1038/gt.2010.59
Li S, Siriwon N, Zhang X, Yang S, Jin T, He F, Kim YJ, Mac J, Lu Z, Wang S, Han X, Wang P (2017) Enhanced cancer immunotherapy by chimeric antigen receptor-modified T cells engineered to secrete checkpoint inhibitors. Clin Cancer Res an official J Am Assoc Cancer Res 23(22):6982–6992. https://doi.org/10.1158/1078-0432.Ccr-17-0867
Rafiq S, Yeku OO, Jackson HJ, Purdon TJ, van Leeuwen DG, Drakes DJ, Song M, Miele MM, Li Z, Wang P, Yan S, Xiang J, Ma X, Seshan VE, Hendrickson RC, Liu C, Brentjens RJ (2018) Targeted delivery of a PD-1-blocking scFv by CAR-T cells enhances anti-tumor efficacy in vivo. Nat Biotechnol 36(9):847–856. https://doi.org/10.1038/nbt.4195
Chong EA, Melenhorst JJ, Lacey SF, Ambrose DE, Gonzalez V, Levine BL, June CH, Schuster SJ (2017) PD-1 blockade modulates chimeric antigen receptor (CAR)-modified T cells: refueling the CAR. Blood 129(8):1039–1041. https://doi.org/10.1182/blood-2016-09-738245
Cassaday RD, Garcia KA, Fromm JR, Percival MM, Turtle CJ, Nghiem PT, Stevenson PA, Estey EH (2020) Phase 2 study of pembrolizumab for measurable residual disease in adults with acute lymphoblastic leukemia. Blood Adv 4(14):3239–3245. https://doi.org/10.1182/bloodadvances.2020002403
Stadtmauer EA, Fraietta JA, Davis MM, Cohen AD, Weber KL, Lancaster E, Mangan PA, Kulikovskaya I, Gupta M, Chen F, Tian L, Gonzalez VE, Xu J, Jung IY, Melenhorst JJ, Plesa G, Shea J, Matlawski T, Cervini A et al (2020) CRISPR-engineered T cells in patients with refractory cancer. Science (New York, NY) 367 (6481). https://doi.org/10.1126/science.aba7365
Minagawa K, Jamil MO, Al-Obaidi M, Pereboeva L, Salzman D, Erba HP, Lamb LS, Bhatia R, Mineishi S, Di Stasi A (2017) Correction: in vitro pre-clinical validation of suicide gene modified anti-CD33 redirected chimeric antigen receptor T-cells for acute myeloid leukemia. PLoS One 12(2):e0172640. https://doi.org/10.1371/journal.pone.0172640
Wang XY, Bian MR, Lin GQ, Yu L, Zhang YM, Wu DP (2023) Tandem bispecific CD123/CLL-1 CAR-T cells exhibit specific cytolytic effector functions against human acute myeloid leukaemia. Eur J Haematol. https://doi.org/10.1111/ejh.14104
Majzner RG, Mackall CL (2018) Tumor antigen escape from CAR T-cell therapy. Cancer Discov 8(10):1219–1226. https://doi.org/10.1158/2159-8290.Cd-18-0442
Schneider D, Xiong Y, Wu D, Nӧlle V, Schmitz S, Haso W, Kaiser A, Dropulic B, Orentas RJ (2017) A tandem CD19/CD20 CAR lentiviral vector drives on-target and off-target antigen modulation in leukemia cell lines. J Immunother Cancer 5:42. https://doi.org/10.1186/s40425-017-0246-1
Zah E, Lin MY, Silva-Benedict A, Jensen MC, Chen YY (2016) T cells expressing CD19/CD20 bispecific chimeric antigen receptors prevent antigen escape by malignant B cells. Cancer Immunol Res 4(6):498–508. https://doi.org/10.1158/2326-6066.Cir-15-0231
Petrov JC, Wada M, Pinz KG, Yan LE, Chen KH, Shuai X, Liu H, Chen X, Leung LH, Salman H, Hagag N, Liu F, Jiang X, Ma Y (2018) Compound CAR T-cells as a double-pronged approach for treating acute myeloid leukemia. Leukemia 32(6):1317–1326. https://doi.org/10.1038/s41375-018-0075-3
Bielamowicz K, Fousek K, Byrd TT, Samaha H, Mukherjee M, Aware N, Wu MF, Orange JS, Sumazin P, Man TK, Joseph SK, Hegde M, Ahmed N (2018) Trivalent CAR T cells overcome interpatient antigenic variability in glioblastoma. Neuro-oncology 20(4):506–518. https://doi.org/10.1093/neuonc/nox182
Huang L, Wang N, Li C, Cao Y, Xiao Y, Xiao M, Zhang Y, Zhang T, Zhou J (2017) Sequential infusion of anti-CD22 and anti-CD19 chimeric antigen receptor T cells for adult patients with refractory/relapsed B-cell acute lymphoblastic leukemia. Blood 130(Supplement 1):846–846. https://doi.org/10.1182/blood.V130.Suppl_1.846.846/JBlood
Bachmann M (2019) The UniCAR system: A modular CAR T cell approach to improve the safety of CAR T cells. Immunol Lett 211:13–22. https://doi.org/10.1016/j.imlet.2019.05.003
Meyer JE, Loff S, Dietrich J, Spehr J, Jurado Jiménez G, von Bonin M, Ehninger G, Cartellieri M, Ehninger A (2021) Evaluation of switch-mediated costimulation in trans on universal CAR-T cells (UniCAR) targeting CD123-positive AML. Oncoimmunology 10(1):1945804. https://doi.org/10.1080/2162402x.2021.1945804
Wermke M, Kraus S, Ehninger A, Bargou RC, Goebeler ME, Middeke JM, Kreissig C, von Bonin M, Koedam J, Pehl M, Bornhäuser M, Einsele H, Ehninger G, Cartellieri M (2021) Proof of concept for a rapidly switchable universal CAR-T platform with UniCAR-T-CD123 in relapsed/refractory AML. Blood 137(22):3145–3148. https://doi.org/10.1182/blood.2020009759
Peroni E, Randi ML, Rosato A, Cagnin S (2023) Acute myeloid leukemia: from NGS, through scRNA-seq, to CAR-T. dissect cancer heterogeneity and tailor the treatment. J Exp Clin Cancer Res CR 42(1):259. https://doi.org/10.1186/s13046-023-02841-8
Rupp LJ, Schumann K, Roybal KT, Gate RE, Ye CJ, Lim WA, Marson A (2017) CRISPR/Cas9-mediated PD-1 disruption enhances anti-tumor efficacy of human chimeric antigen receptor T cells. Sci Rep 7(1):737. https://doi.org/10.1038/s41598-017-00462-8
Dimitri A, Herbst F, Fraietta JA (2022) Engineering the next-generation of CAR T-cells with CRISPR-Cas9 gene editing. Mol Cancer 21(1):78. https://doi.org/10.1186/s12943-022-01559-z
Jiang W, He Y, He W, Wu G, Zhou X, Sheng Q, Zhong W, Lu Y, Ding Y, Lu Q, Ye F, Hua H (2020) Exhausted CD8+T cells in the tumor immune microenvironment: new pathways to therapy. Front Immunol 11:622509. https://doi.org/10.3389/fimmu.2020.622509
Sterner RM, Cox MJ, Sakemura R, Kenderian SS (2019) Using CRISPR/Cas9 to Knock Out GM-CSF in CAR-T Cells. J Visual Exp : JoVE 149. https://doi.org/10.3791/59629
Eyquem J, Mansilla-Soto J, Giavridis T, van der Stegen SJ, Hamieh M, Cunanan KM, Odak A, Gönen M, Sadelain M (2017) Targeting a CAR to the TRAC locus with CRISPR/Cas9 enhances tumour rejection. Nature 543(7643):113–117. https://doi.org/10.1038/nature21405
Oluwole OO, Davila ML (2016) At the bedside: clinical review of chimeric antigen receptor (CAR) T cell therapy for B cell malignancies. J Leukoc Biol 100(6):1265–1272. https://doi.org/10.1189/jlb.5BT1115-524R
Park JH, Geyer MB, Brentjens RJ (2016) CD19-targeted CAR T-cell therapeutics for hematologic malignancies: interpreting clinical outcomes to date. Blood 127(26):3312–3320. https://doi.org/10.1182/blood-2016-02-629063
Fiorenza S, Turtle CJ (2021) CAR-T cell therapy for acute myeloid leukemia: preclinical rationale, current clinical progress, and barriers to success. BioDrugs : Clin Immunother , Biopharm Gene Ther 35(3):281–302. https://doi.org/10.1007/s40259-021-00477-8
Jain T, Knezevic A, Pennisi M, Chen Y, Ruiz JD, Purdon TJ, Devlin SM, Smith M, Shah GL, Halton E, Diamonte C, Scordo M, Sauter CS, Mead E, Santomasso BD, Palomba ML, Batlevi CW, Maloy MA, Giralt S et al (2020) Hematopoietic recovery in patients receiving chimeric antigen receptor T-cell therapy for hematologic malignancies. Blood Adv 4(15):3776–3787. https://doi.org/10.1182/bloodadvances.2020002509
Rejeski K, Perez A, Iacoboni G, Penack O, Bücklein V, Jentzsch L, Mougiakakos D, Johnson G, Arciola B, Carpio C, Blumenberg V, Hoster E, Bullinger L, Locke FL, von Bergwelt-Baildon M, Mackensen A, Bethge W, Barba P, Jain MD, Subklewe M (2022) The CAR-HEMATOTOX risk-stratifies patients for severe infections and disease progression after CD19 CAR-T in R/R LBCL. J Immunother Cancer 10(5). https://doi.org/10.1136/jitc-2021-004475
Bordignon C, Bonini C, Verzeletti S, Nobili N, Maggioni D, Traversari C, Giavazzi R, Servida P, Zappone E, Benazzi E et al (1995) Transfer of the HSV-tk gene into donor peripheral blood lymphocytes for in vivo modulation of donor anti-tumor immunity after allogeneic bone marrow transplantation. Hum Gene Ther 6(6):813–819. https://doi.org/10.1089/hum.1995.6.6-813
Tey SK, Dotti G, Rooney CM, Heslop HE, Brenner MK (2007) Inducible caspase 9 suicide gene to improve the safety of allodepleted T cells after haploidentical stem cell transplantation. Biol Blood Marrow Transpl :J Am Soc Blood Marrow Transpl 13(8):913–924. https://doi.org/10.1016/j.bbmt.2007.04.005
Cummins KD, Gill S (2019) Will CAR T cell therapy have a role in AML? Promises and pitfalls. Semin Hematol 56(2):155–163. https://doi.org/10.1053/j.seminhematol.2018.08.008
Casirati G, Cosentino A, Mucci A, Salah Mahmoud M, Ugarte Zabala I, Zeng J, Ficarro SB, Klatt D, Brendel C, Rambaldi A, Ritz J, Marto JA, Pellin D, Bauer DE, Armstrong SA, Genovese P (2023) Epitope editing enables targeted immunotherapy of acute myeloid leukaemia. Nature 621(7978):404–414. https://doi.org/10.1038/s41586-023-06496-5
Morris EC, Neelapu SS, Giavridis T, Sadelain M (2022) Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy. Nat Rev Immunol 22(2):85–96. https://doi.org/10.1038/s41577-021-00547-6
Kochenderfer JN, Dudley ME, Feldman SA, Wilson WH, Spaner DE, Maric I, Stetler-Stevenson M, Phan GQ, Hughes MS, Sherry RM, Yang JC, Kammula US, Devillier L, Carpenter R, Nathan DA, Morgan RA, Laurencot C, Rosenberg SA (2012) B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood 119(12):2709–2720. https://doi.org/10.1182/blood-2011-10-384388
Cobb DA, Lee DW (2021) Cytokine release syndrome biology and management. Cancer J (Sudbury, Mass) 27(2):119–125. https://doi.org/10.1097/ppo.0000000000000515
Grupp SA, Kalos M, Barrett D, Aplenc R, Porter DL, Rheingold SR, Teachey DT, Chew A, Hauck B, Wright JF, Milone MC, Levine BL, June CH (2013) Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med 368(16):1509–1518. https://doi.org/10.1056/NEJMoa1215134
Topp MS, van Meerten T, Houot R, Minnema MC, Bouabdallah K, Lugtenburg PJ, Thieblemont C, Wermke M, Song KW, Avivi I, Kuruvilla J, Dührsen U, Zheng Y, Vardhanabhuti S, Dong J, Bot A, Rossi JM, Plaks V, Sherman M et al (2021) Earlier corticosteroid use for adverse event management in patients receiving axicabtagene ciloleucel for large B-cell lymphoma. Br J Haematol 195(3):388–398. https://doi.org/10.1111/bjh.17673
Funding
This work was supported by the grants from the National Natural Science Foundation of China (Grant No. 81970138, 82270165), National Key R&D Program of China (2022YFC2502703), Jiangsu Province Natural Science Foundation of China (Grant No. BK20221235), Translational Research Grant of NCRCH (Grant No. 2020ZKMB05), Jiangsu Province “333” Project, Social Development Project of the Science and Technology Department of Jiangsu (Grant No. BE2021649).
Author information
Authors and Affiliations
Contributions
The review article was collaboratively conceptualized and designed by all three authors, W. X., Z. Y., and X. S. The overall framework of the paper was envisioned and designed by W. X., Z. Y., and X. S. W. X. conducted the literature search and collection. Z. Y., and X. S. provided expert theoretical support. The manuscript was written by W. X.
Corresponding authors
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The author Xiangyu Wang contributed equally to this work.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., Zhang, Y. & Xue, S. Recent progress in chimeric antigen receptor therapy for acute myeloid leukemia. Ann Hematol 103, 1843–1857 (2024). https://doi.org/10.1007/s00277-023-05601-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-023-05601-y