Abstract
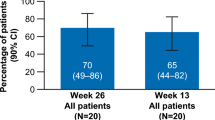
Eltrombopag has shown a promising effect on patients with refractory/relapsed aplastic anemia (AA). However, data in Asian patients are limited due to the short launching time. Data from relapsed/refractory AA patients treated with eltrombopag in Peking Union Medical College Hospital from December 2017 to May 2019 were analyzed retrospectively. Totally 41 patients including 37 non-severe AA and 4 severe AA were analyzed with a median age of 47 (13~81) years old. The dosage of eltrombopag varies from 25 mg every other day (qod) to 100 mg every day (qd) (median 75 mg qd) with the duration of 13 (3~23) months. The overall response rates were 51.2%, 58.5%, and 55.2% at 3-month, 6-month, and 1-year follow-up. Seventy-five percent of patients achieved the best response below the dosage of 75 mg qd at 3 (1–6) months. Two patients stopped eltrombopag after achieving complete remission and remained stable. Two patients relapsed and evolved to myelodysplastic syndrome. Adverse events included gastrointestinal discomfort, hepatic toxicity, and skin reactions, which were mild and controllable. Eltrombopag is effective for Chinese relapsed/refractory AA patients with a relatively lower dose and acceptable side effects.

Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to personal data protection reasons but are available from the corresponding author on reasonable request.
Change history
02 November 2020
The article ���Eltrombopag is effective in patients with relapse/refractory aplastic anemia���report from a single center in China���.
References
Killick SB, Bown N, Cavenagh J, Dokal I, Foukaneli T, Hill A, Hillmen P, Ireland R, Kulasekararaj A, Mufti G, Snowden JA, Samarasinghe S, Wood A, Marsh JC (2016) Guidelines for the diagnosis and management of adult aplastic anaemia. Br J Haematol 172(2):187–207. https://doi.org/10.1111/bjh.13853
Gill H, Wong RSM, Kwong YL (2017) From chronic immune thrombocytopenia to severe aplastic anemia: recent insights into the evolution of eltrombopag. Ther Adv Hematol 8(5):159–174. https://doi.org/10.1177/2040620717693573
Fattizzo B, Levati G, Cassin R, Barcellini W (2019) Eltrombopag in immune thrombocytopenia, aplastic anemia, and myelodysplastic syndrome: from megakaryopoiesis to immunomodulation. Drugs 79(12):1305–1319. https://doi.org/10.1007/s40265-019-01159-0
Desmond R, Townsley DM, Dumitriu B, Olnes MJ, Scheinberg P, Bevans M, Parikh AR, Broder K, Calvo KR, Wu CO, Young NS, Dunbar CE (2014) Eltrombopag restores trilineage hematopoiesis in refractory severe aplastic anemia that can be sustained on discontinuation of drug. Blood 123(12):1818–1825. https://doi.org/10.1182/blood-2013-10-534743
Scheinberg P (2018) Activity of eltrombopag in severe aplastic anemia. Blood Adv 2(21):3054–3062. https://doi.org/10.1182/bloodadvances.2018020248
Zhao Z, Sun Q, Sokoll LJ, Streiff M, Cheng Z, Grasmeder S, Townsley DM, Young NS, Dunbar CE, Winkler T (2018) Eltrombopag mobilizes iron in patients with aplastic anemia. Blood 131(21):2399–2402. https://doi.org/10.1182/blood-2018-01-826784
Olnes MJ, Scheinberg P, Calvo KR, Desmond R, Tang Y, Dumitriu B, Parikh AR, Soto S, Biancotto A, Feng X, Lozier J, Wu CO, Young NS, Dunbar CE (2012) Eltrombopag and improved hematopoiesis in refractory aplastic anemia. N Engl J Med 367(1):11–19. https://doi.org/10.1056/NEJMoa1200931
Geng W, Kearney S, Nelson S (2018) Upfront eltrombopag monotherapy induces stable hematologic remission in pediatric patients with nonsevere idiopathic aplastic anemia. Pediatr Blood Cancer 65(10):e27290. https://doi.org/10.1002/pbc.27290
Fattizzo B, Kulasekararaj AG, Hill A, Benson-Quarm N, Griffin M, Munir T, Arnold L, Riley K, Ireland R, De Lavallade H, Potter V, Consonni D, Hillmen P, Mufti GJ, Barcellini W, Marsh JCW (2019) Clinical and morphological predictors of outcome in older aplastic anemia patients treated with eltrombopag. Haematologica 104(11):e494–e496. https://doi.org/10.3324/haematol.2019.216374
Cheng H, Wang X, Zhou D, Cao J, Larochelle A, Xu KL (2019) Eltrombopag combined with cyclosporine may have an effect on very severe aplastic anemia. Ann Hematol 98(8):2009–2011. https://doi.org/10.1007/s00277-019-03658-2
Desmond R, Townsley DM, Dunbar C, Young NS (2015) Eltrombopag in aplastic anemia. Semin Hematol 52(1):31–37. https://doi.org/10.1053/j.seminhematol.2014.10.002
Ecsedi M, Lengline E, Knol-Bout C, Bosman P, Eikema DJ, Afanasyev B, Maschan A, Dreger P, Halkes CJM, Drexler B, Cortelezzi A, Drenou B, Patriarca A, Bruno B, Onofrillo D, Lanino E, Pulanic D, Serventi-Seiwerth R, Garnier A, Ljungman P, Bonifazi F, Giammarco S, Tournilhac O, Pioltelli P, Rovo A, Risitano AM, de Latour RP, Dufour C, Passweg J (2019) Use of eltrombopag in aplastic anemia in Europe. Ann Hematol 98(6):1341–1350. https://doi.org/10.1007/s00277-019-03652-8
Hwang YY, Gill H, Chan TSY, Leung GMK, Cheung CYM, Kwong YL (2018) Eltrombopag in the management of aplastic anaemia: real-world experience in a non-trial setting. Hematology (Amsterdam, Netherlands) 23(7):399–404. https://doi.org/10.1080/10245332.2017.1422306
Konishi A, Nakaya A, Fujita S, Satake A, Nakanishi T, Azuma Y, Tsubokura Y, Saito R, Hotta M, Yoshimura H, Ishii K, Ito T, Nomura S (2019) Evaluation of eltrombopag in patients with aplastic anemia in real-world experience. Leuk Res Rep 11:11–13. https://doi.org/10.1016/j.lrr.2019.03.002
Yamazaki H, Ohta K, Iida H, Imada K, Obara N, Tokumine Y, Tomiyama Y, Usuki K, Imajo K, Miyamura K, Sasaki O, Fanghong Z, Hattori T, Tajima T, Matsuda A, Nakao S (2019) Hematologic recovery induced by eltrombopag in Japanese patients with aplastic anemia refractory or intolerant to immunosuppressive therapy. Int J Hematol 110(2):187–196. https://doi.org/10.1007/s12185-019-02683-1
Camitta BM, Rappeport JM, Parkman R, Nathan DG (1975) Selection of patients for bone marrow transplantation in severe aplastic anemia. Blood 45(3):355–363
Marsh JC, Ball SE, Cavenagh J, Darbyshire P, Dokal I, Gordon-Smith EC, Keidan J, Laurie A, Martin A, Mercieca J, Killick SB, Stewart R, Yin JA (2009) Guidelines for the diagnosis and management of aplastic anaemia. Br J Haematol 147(1):43–70. https://doi.org/10.1111/j.1365-2141.2009.07842.x
Townsley DM, Scheinberg P, Winkler T, Desmond R, Dumitriu B, Rios O, Weinstein B, Valdez J, Lotter J, Feng X, Desierto M, Leuva H, Bevans M, Wu C, Larochelle A, Calvo KR, Dunbar CE, Young NS (2017) Eltrombopag added to standard immunosuppression for aplastic anemia. N Engl J Med 376(16):1540–1550. https://doi.org/10.1056/NEJMoa1613878
Matthys G, Park JW, McGuire S, Wire MB, Bowen C, Williams D, Jenkins J, Peng B (2011) Clinical pharmacokinetics, platelet response, and safety of eltrombopag at supratherapeutic doses of up to 200 mg once daily in healthy volunteers. J Clin Pharmacol 51(3):301–308. https://doi.org/10.1177/0091270010368677
Gibiansky E, Zhang J, Williams D, Wang Z, Ouellet D (2011) Population pharmacokinetics of eltrombopag in healthy subjects and patients with chronic idiopathic thrombocytopenic purpura. J Clin Pharmacol 51(6):842–856
Alexander WS, Roberts AW, Nicola NA, Li R, Metcalf D (1996) Deficiencies in progenitor cells of multiple hematopoietic lineages and defective megakaryocytopoiesis in mice lacking the thrombopoietic receptor c-Mpl. Blood 87(6):2162–2170
Qian H, Buza-Vidas N, Hyland CD, Jensen CT, Antonchuk J, Mansson R, Thoren LA, Ekblom M, Alexander WS, Jacobsen SEW Critical role of thrombopoietin in maintaining adult quiescent hematopoietic stem cells. Cell Stem Cell 1(6):671–684
Groarke EM, Patel BA, Diamond C, Gutierrez-Rodrigues F, Young NS (2019) Outcomes in pediatric patients with severe aplastic anemia treated with standard immunosuppression and eltrombopag. Blood 134(Supplement_1):454–454
Ho C, Visintini S (2018) CADTH rapid response reports. In: Eltrombopag for the treatment of aplastic anemia: a review of clinical and cost-effectiveness. Canadian Agency for Drugs and Technologies in Health Copyright (c) 2018 Canadian Agency for Drugs and Technologies in Health., Ottawa (ON)
Hong Y, Li X, Wan B, Li N, Chen Y (2019) Efficacy and safety of eltrombopag for aplastic anemia: a systematic review and meta-analysis. Clin Drug Investig 39(2):141–156. https://doi.org/10.1007/s40261-018-0725-2
Lum SH, Grainger JD (2016) Eltrombopag for the treatment of aplastic anemia: current perspectives. Drug Des Dev Ther 10:2833–2843. https://doi.org/10.2147/dddt.s95715
Cooper JN, Young NS (2017) Clonality in context: hematopoietic clones in their marrow environment. Blood 130(22):2363–2372. https://doi.org/10.1182/blood-2017-07-794362
Acknowledgments
We thank all patients who consented to disclose their medical records and answered our review calls. Their cooperation and helpful comments throw light upon our data collection and statistical analysis.
Code availability
Not applicable.
Funding
This study was supported by grants from Beijing Natural Science Foundation (7192168), the Chinese Academy of Medical Sciences (CAMS) innovation fund for medical sciences (2016-I2M-3-004), and the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2019XK 320047).
Author information
Authors and Affiliations
Contributions
J.R. and B.H. designed the study and wrote the manuscript. J.R and W.Z. collected and analyzed the clinical data. M.C. and C.Y. helped to analyze the date and provide critical suggestions. All authors reviewed the manuscript finally. J.R., W.Z., M.C., C.Y., and H.B. approved the final submission of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
This study was approved by the ethics committee of Peking Union Medical College Hospital and was conducted in accordance with the Declaration of Helsinki.
Consent to participate
Written informed consent to participate was signed by all individual participants included in the study before data collected.
Consent for publication
Written informed consent for the publication of identifying images or other personal or clinical details was obtained from all of the participants in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised: This article was originally published with an Open Access but due to the authors final decision not to opt for Open Choice this correction was created.
Rights and permissions
About this article
Cite this article
Ruan, J., Zuo, W., Chen, M. et al. Eltrombopag is effective in patients with relapse/refractory aplastic anemia—report from a single center in China. Ann Hematol 99, 2755–2761 (2020). https://doi.org/10.1007/s00277-020-04266-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-020-04266-1