Abstract
Introduction
Immune checkpoint inhibitors (ICIs) represent a cornerstone for the treatment of many advanced tumors. When 65 is considered as a cut-off age, ICIs are equally effective in younger and older patients. However, the efficacy of ICIs among patients aged ≥ 75 remains uncertain, since those patients were generally under-represented in clinical trials. We performed a pooled analysis of major randomized trials including data of outcome in very older population.
Material and methods
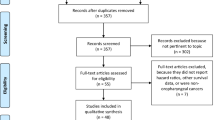
We searched PubMed, Embase, and the Cochrane Library for randomized controlled trials published from the inception of each database to November 22th, 2019. We only included (1) randomized studies comparing ICIs alone or in combinations with no ICIs, (2) studies reporting data of patients older than 75 years, (3) studies for solid tumors, and (4) studies with HR and 95% confidence interval (CI) available for OS based on 75 years as cut-off age. All data were expressed as the combination of HR and 95% CI, and P < 0.05 was considered to be statistically significant.
Results
A total of n = 8 publications for a total of n = 12 randomized studies were aggregated in the quantitative analysis. Overall, the pooled analysis showed a borderline significant OS benefit for ICIs compared to no ICIs arms (HR = 0.84, 95% CI 0.7–1; P = 0.05) in particular in first-line trials with HR = 0.77 (95%CI 0.61–0.96; P = 0.02).
Conclusion
We conclude that ICIs may be offered in patients older than 75 years, providing a complete geriatric and clinical evaluation is performed in all subjects before starting therapy.

Similar content being viewed by others
References
Elias R, Karantanos T, Sira E, Hartshorn KL (2017) Immunotherapy comes of age: immune aging and checkpoint inhibitors. J Geriatr Oncol. https://doi.org/10.1016/j.jgo.2017.02.001
Nishijima TF, Muss HB, Shachar SS, Moschos SJ (2016) Comparison of efficacy of immune checkpoint inhibitors (ICIs) between younger and older patients: a systematic review and meta-analysis. Cancer Treat Rev. https://doi.org/10.1016/j.ctrv.2016.02.006
Brahmer J, Reckamp KL, Baas P et al (2015) Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med 373(2):123–135. https://doi.org/10.1056/NEJMoa1504627
Robert C, Long GV, Brady B et al (2015) Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med 372(4):320–330. https://doi.org/10.1056/NEJMoa1412082
Govindan R, Szczesna A, Ahn MJ et al (2017) Phase III trial of ipilimumab combined with paclitaxel and carboplatin in advanced squamous non–small-cell lung cancer. J Clin Oncol 35(30):3449–3457. https://doi.org/10.1200/JCO.2016.71.7629
Nosaki K, Saka H, Hosomi Y et al (2019) Safety and efficacy of pembrolizumab monotherapy in elderly patients with PD-L1–positive advanced non–small-cell lung cancer: pooled analysis from the KEYNOTE-010, KEYNOTE-024, and KEYNOTE-042 studies. Lung Cancer 135:188–195. https://doi.org/10.1016/j.lungcan.2019.07.004
Marur S, Singh H, Mishra-Kalyani P et al (2018) FDA analyses of survival in older adults with metastatic non–small cell lung cancer in controlled trials of PD-1/PD-L1 blocking antibodies. Semin Oncol 45(4):220–225. https://doi.org/10.1053/j.seminoncol.2018.08.007
Reck M, Luft A, Szczesna A et al (2016) Phase III randomized trial of ipilimumab plus etoposide and platinum versus placebo plus etoposide and platinum in extensive-stage small-cell lung cancer. J Clin Oncol 34(31):3740–3748. https://doi.org/10.1200/JCO.2016.67.6601
Motzer RJ, Escudier B, McDermott DF et al (2015) Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med. https://doi.org/10.1056/NEJMoa1510665
Hellmann MD, Paz-Ares L, Bernabe Caro R et al (2019) Nivolumab plus Ipilimumab in advanced non–small-cell lung cancer. N Engl J Med. https://doi.org/10.1056/nejmoa1910231
Kanesvaran R, Le SO, Motzer R et al (2018) Elderly patients with metastatic renal cell carcinoma: position paper from the International Society of Geriatric Oncology. Lancet Oncol. https://doi.org/10.1016/S1470-2045(18)30125-6
Pallis AG, Gridelli C, Wedding U et al (2014) Management of elderly patients with NSCLC; updated expert’s opinion paper: EORTC elderly task force, lung cancer group and international society for geriatric oncology. Ann Oncol. https://doi.org/10.1093/annonc/mdu022
Corre R, Greillier L, Le Caër H et al (2016) Use of a comprehensive geriatric assessment for the management of elderly patients with advanced non-small cell lung cancer: the Phase III randomized ESOGIA-GFPC-GECP 08-02 study. J Clin Oncol. https://doi.org/10.1200/JCO.2015.63.5839
Sbrana A, Antognoli R, Pasqualetti G et al (2019) Effectiveness of Multi-Prognostic Index in older patients with advanced malignancies treated with immunotherapy. J Geriatr Oncol. https://doi.org/10.1016/j.jgo.2019.09.010
Author information
Authors and Affiliations
Contributions
All authors made substantial contributions to the conception or design of the work; the acquisition, analysis, and interpretation of data.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Melissa Bersanelli received research funding by Roche, Pfizer, Seqirus, AstraZeneca, Bristol-Myers Squibb, Novartis and Sanofi; she also received honoraria for advisory role and as speaker at scientific events by Bristol-Myers Squibb, Novartis and Pfizer. None to declare for all the other authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Petrelli, F., Inno, A., Ghidini, A. et al. Efficacy of immune checkpoint inhibitors in elderly patients aged ≥ 75 years. Cancer Immunol Immunother 70, 1777–1780 (2021). https://doi.org/10.1007/s00262-020-02779-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02779-2