Abstract
Background and objective
Activation of the immune checkpoints and expression of chemokines and chemokine receptors have been reported to promote HCC progression. This study aimed to assess the differential expression of Tim-3, PD-1, and CCR5 on peripheral blood lymphocytes from patients with HCV-related HCC and correlate their expression with the treatment outcomes.
Patients and methods
The study incorporated 40 patients with chronic HCV-related HCC and 40 healthy controls. Patients were radiologically assessed for hepatic focal lesions and portal vein thrombosis. Response to HCC treatment and overall survival (OS) outcomes were determined. The expression of Tim-3, PD-1, and CCR5 among CD19+, CD4+, and CD8+ lymphocytes was assessed by flow cytometry.
Results
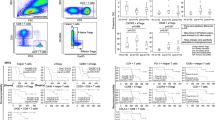
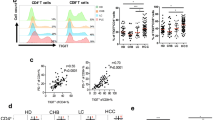
Higher frequencies of CD4+ and CD8+ cells expressing each of Tim-3 and PD-1 and PD-1+CD19+ cells were observed in the HCV-related HCC patients in comparison with controls. The highest expression of Tim-3 and PD-1 was by the CD8+ cells. Strong relations were detected among PD-1+CD19+, PD-1+CD4+ and PD-1+CD8+ cells. Elevated levels of PD-1+ lymphocytes were significantly associated with poor treatment response and shorter OS.
Conclusion
Modulation of the expression of immune checkpoints as Tim-3 and PD-1, and of CCR5 on T cells is somehow related to HCC. CD8+ T cells expressing PD-1 were the most relevant to HCC prognosis (OS and treatment response) and could represent a promising target for immune therapy against HCC. Future studies need to focus on exploring PD-1+ B cells and Tim-3+CD4+ cells, which seem to play a significant role in the pathogenesis of HCC.


Similar content being viewed by others
Abbreviations
- APC:
-
Allophycocyanin
- CCL5-CCR5:
-
Chemokine ligand 5-chemokine receptor 5
- DAA:
-
Direct-acting antiviral drugs
- FITC:
-
Fluoroisothiocyanate
- FLR:
-
Future liver remnant
- HCC:
-
Hepatocellular carcinoma
- OS:
-
Overall survival
- PBMCs:
-
Peripheral blood mononuclear cells
- PBS:
-
Phosphate buffer saline
- PD-1:
-
Programmed cell death protein-1
- PE:
-
Phycoerythrin
- Per-CP:
-
Peridinium-chlorophyll-protein
- PS:
-
Performance status
- RFA:
-
Radiofrequency ablation
- TACE:
-
Trans-arterial chemo-embolization
- Tim-3:
-
T-cell immunoglobulin and mucin-domain containing-3
References
Rayan A, Zahran A, El-Badawy O, Soliman A (2019) P-056 Differential expression of CCR5, TIM3 and PD-1 on peripheral blood T and B lymphocytes from hepatitis C patients with hepatocellular carcinoma and their impact on treatment outcomes. Ann Oncol 30((Supplement_4)):mdz155.055
Nordenstedt H, White DL, El-Serag HB (2010) The changing pattern of epidemiology in hepatocellular carcinoma. Dig Liver Dis 42(Suppl 3):S206–214. https://doi.org/10.1016/s1590-8658(10)60507-5
Laursen L (2014) A preventable cancer. Nature 516(7529):S2–3. https://doi.org/10.1038/516S2a
Zahran AM, Abdel-Rahim MH, Refaat A, Sayed M, Othman MM, Khalak LMR, Hetta HF (2020) Circulating hematopoietic stem cells, endothelial progenitor cells and cancer stem cells in hepatocellular carcinoma patients: contribution to diagnosis and prognosis. Acta Oncol 59(1):33–39
Chaturvedi VK, Singh A, Dubey SK, Hetta HF, John J, Singh M (2019) Molecular mechanistic insight of hepatitis B virus mediated hepatocellular carcinoma. Microb Pathog 128:184–194
Anzola M (2004) Hepatocellular carcinoma: role of hepatitis B and hepatitis C viruses proteins in hepatocarcinogenesis. J Viral Hepatitis 11(5):383–393. https://doi.org/10.1111/j.1365-2893.2004.00521.x
Hetta HF, Mekky MA, Khalil NK, Mohamed WA, El-Feky MA, Ahmed SH, Daef EA, Medhat A, Nassar MI, Sherman KE (2016) Extra-hepatic infection of hepatitis C virus in the colon tissue and its relationship with hepatitis C virus pathogenesis. J Med Microbiol 65(8):703
Zahran AM, Nafady-Hego H, Mansor SG, Abbas WA, Abdel-Malek MO, Mekky MA, Hetta HF (2019) Increased frequency and FOXP3 expression of human CD8+ CD25High+ T lymphocytes and its relation to CD4 regulatory T cells in patients with hepatocellular carcinoma. Hum Immunol 80:510–516
Hassan EA, Ahmed EH, Nafee AM, El-Gafary N, Hetta HF, El-Mokhtar MA (2019) Regulatory T cells, IL10 and IL6 in HCV related hepatocellular carcinoma after transarterial chemoembolization (TACE). Egypt J Immunol 26:69–78
Zahran AM, Ashmawy AM, Rayan A, Elkady A, Elsherbiny NM, Hetta HF (2018) Frequency and implications of natural killer and natural killer T cells in hepatocellular carcinoma. Egypt J Immunol 25:45
Elsegood CL, Tirnitz-Parker JE, Olynyk JK, Yeoh GC (2017) Immune checkpoint inhibition: prospects for prevention and therapy of hepatocellular carcinoma. Clin Transl Immunol 6(11):e161. https://doi.org/10.1038/cti.2017.47
Sarvaria A, Madrigal JA, Saudemont A (2017) B cell regulation in cancer and anti-tumor immunity. Cell Mol Immunol 14(8):662
Hetta H, Elkady A, Tohamy T, Badary M (2016) Regulatory B cells: key players in hepatocellular carcinoma progression. Gastroenterol Hepatol Open Access 5(2):00136
Fremd C, Schuetz F, Sohn C, Beckhove P, Domschke C (2013) B cell-regulated immune responses in tumor models and cancer patients. Oncoimmunology 2(7):e25443. https://doi.org/10.4161/onci.25443
Inoue S, Leitner WW, Golding B, Scott D (2006) Inhibitory effects of B cells on antitumor immunity. Can Res 66(15):7741–7747. https://doi.org/10.1158/0008-5472.can-05-3766
Schioppa T, Moore R, Thompson RG, Rosser EC, Kulbe H, Nedospasov S, Mauri C, Coussens LM, Balkwill FR (2011) B regulatory cells and the tumor-promoting actions of TNF-alpha during squamous carcinogenesis. Proc Natl Acad Sci USA 108(26):10662–10667. https://doi.org/10.1073/pnas.1100994108
DiLillo DJ, Yanaba K, Tedder TF (2010) B cells are required for optimal CD4+ and CD8+ T cell tumor immunity: therapeutic B cell depletion enhances B16 melanoma growth in mice. J Immunol (Baltimore, Md : 1950) 184(7):4006–4016. https://doi.org/10.4049/jimmunol.0903009
Li Q, Teitz-Tennenbaum S, Donald EJ, Li M, Chang AE (2009) In vivo sensitized and in vitro activated B cells mediate tumor regression in cancer adoptive immunotherapy. J Immunol (Baltimore, Md : 1950) 183(5):3195–3203. https://doi.org/10.4049/jimmunol.0803773
Raufi A, Tirona MT (2017) Prospect of the use of checkpoint inhibitors in hepatocellular cancer treatments. Cancer Manag Res 9:19–27. https://doi.org/10.2147/cmar.s111673
Hetta HF, Zahran AM, Mansor SG, Abdel-Malek MO, Mekky MA, Abbas WA (2019) Frequency and implications of myeloid-derived suppressor cells and lymphocyte subsets in Egyptian patients with hepatitis C virus-related hepatocellular carcinoma. J Med Virol 91(7):1319–1328
Zahran AM, Zahran ZAM, El-Badawy O, Abdel-Rahim MH, Ali WA, Rayan A, El-Masry MA, Abozaid MA, Hetta HF (2019) Prognostic impact of toll-like receptors 2 and 4 expression on monocytes in Egyptian patients with hepatocellular carcinoma. Immunol Res 67(2–3):157–165. https://doi.org/10.1007/s12026-019-09075-x
Hetta HF, Khairy H, Ismail S (2017) Circulating IL17A and IFN-gamma serum levels in cirrhotic hepatitis C virus infected patients with autoimmune thyroiditis. Int J Curr Microbiol Appl Sci 6(3):1972–1983
Liang CM, Chen L, Hu H, Ma HY, Gao LL, Qin J, Zhong CP (2015) Chemokines and their receptors play important roles in the development of hepatocellular carcinoma. World J Hepatol 7(10):1390–1402. https://doi.org/10.4254/wjh.v7.i10.1390
Nahon P, Sutton A, Rufat P, Simon C, Trinchet JC, Gattegno L, Beaugrand M, Charnaux N (2008) Chemokine system polymorphisms, survival and hepatocellular carcinoma occurrence in patients with hepatitis C virus-related cirrhosis. World J Gastroenterol 14(5):713–719
Das M, Zhu C, Kuchroo VK (2017) Tim-3 and its role in regulating anti-tumor immunity. Immunol Rev 276(1):97–111. https://doi.org/10.1111/imr.12520
Hato T, Goyal L, Greten TF, Duda DG, Zhu AX (2014) Immune checkpoint blockade in hepatocellular carcinoma: current progress and future directions. Hepatology (Baltimore, MD) 60(5):1776–1782. https://doi.org/10.1002/hep.27246
Keir ME, Butte MJ, Freeman GJ, Sharpe AH (2008) PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol 26:677–704. https://doi.org/10.1146/annurev.immunol.26.021607.090331
Chen J, Jiang CC, Jin L, Zhang XD (2016) Regulation of PD-L1: a novel role of pro-survival signalling in cancer. Ann Oncol 27(3):409–416. https://doi.org/10.1093/annonc/mdv615
Carter L, Fouser LA, Jussif J, Fitz L, Deng B, Wood CR, Collins M, Honjo T, Freeman GJ, Carreno BM (2002) PD-1:PD-L inhibitory pathway affects both CD4(+) and CD8(+) T cells and is overcome by IL-2. Eur J Immunol 32(3):634–643. https://doi.org/10.1002/1521-4141(200203)32:3%3c634:aid-immu634%3e3.0.co;2-9
Freeman GJ, Long AJ, Iwai Y, Bourque K, Chernova T, Nishimura H, Fitz LJ, Malenkovich N, Okazaki T, Byrne MC, Horton HF, Fouser L, Carter L, Ling V, Bowman MR, Carreno BM, Collins M, Wood CR, Honjo T (2000) Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med 192(7):1027–1034. https://doi.org/10.1084/jem.192.7.1027
Blank C, Gajewski TF, Mackensen A (2005) Interaction of PD-L1 on tumor cells with PD-1 on tumor-specific T cells as a mechanism of immune evasion: implications for tumor immunotherapy. Cancer Immunol Immunother 54(4):307–314. https://doi.org/10.1007/s00262-004-0593-x
Liu Y (2018) Immunomodulatory effects of Tim-3 and PD-1 on chronic hepatitis B virus infection. Infect Int 7(1):6–11
Missale G, Cariani E (2018) Time for hepatocellular carcinoma immunotherapy: insights for successful clinical applications in this challenging tumor. Hepatoma Res 4:22. https://doi.org/10.20517/2394-5079.2018.72
Golden-Mason L, Palmer BE, Kassam N, Townshend-Bulson L, Livingston S, McMahon BJ, Castelblanco N, Kuchroo V, Gretch DR, Rosen HR (2009) Negative immune regulator Tim-3 is overexpressed on T cells in hepatitis C virus infection and its blockade rescues dysfunctional CD4+ and CD8+ T cells. J Virol 83(18):9122–9130
Wu W, Shi Y, Li J, Chen F, Chen Z, Zheng M (2011) Tim-3 expression on peripheral T cell subsets correlates with disease progression in hepatitis B infection. Virol J 8(1):113
Schmidt J, Blum HE, Thimme R (2013) T-cell responses in hepatitis B and C virus infection: similarities and differences. Emerg Microbes Infect 2(3):e15. https://doi.org/10.1038/emi.2013.14
Li H, Wu K, Tao K, Chen L, Zheng Q, Lu X, Liu J, Shi L, Liu C, Wang G, Zou W (2012) Tim-3/galectin-9 signaling pathway mediates T-cell dysfunction and predicts poor prognosis in patients with hepatitis B virus-associated hepatocellular carcinoma. Hepatology (Baltimore, MD) 56(4):1342–1351. https://doi.org/10.1002/hep.25777
Shi F, Shi M, Zeng Z, Qi RZ, Liu ZW, Zhang JY, Yang YP, Tien P, Wang FS (2011) PD-1 and PD-L1 upregulation promotes CD8(+) T-cell apoptosis and postoperative recurrence in hepatocellular carcinoma patients. Int J Cancer 128(4):887–896. https://doi.org/10.1002/ijc.25397
He G, Zhang H, Zhou J, Wang B, Chen Y, Kong Y, Xie X, Wang X, Fei R, Wei L (2015) Peritumoural neutrophils negatively regulate adaptive immunity via the PD-L1/PD-1 signalling pathway in hepatocellular carcinoma. J Exp Clin Cancer Res 34(1):141
Pugh RN, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R (1973) Transection of the oesophagus for bleeding oesophageal varices. Br J Surg 60(8):646–649
Tsou P, Katayama H, Ostrin EJ, Hanash SM (2016) The emerging role of B cells in tumor immunity. Can Res 76(19):5597–5601. https://doi.org/10.1158/0008-5472.can-16-0431
Nelson BH (2010) CD20+ B cells: the other tumor-infiltrating lymphocytes. J Immunol 185(9):4977–4982
Xiao X, Lao XM, Chen MM, Liu RX, Wei Y, Ouyang FZ, Chen DP, Zhao XY, Zhao Q, Li XF, Liu CL, Zheng L, Kuang DM (2016) PD-1hi Identifies a novel regulatory B-cell population in human hepatoma that promotes disease progression. Cancer Discov 6(5):546–559. https://doi.org/10.1158/2159-8290.cd-15-1408
Fukada K, Sobao Y, Tomiyama H, Oka S, Takiguchi M (2002) Functional expression of the chemokine receptor CCR5 on virus epitope-specific memory and effector CD8+ T cells. J Immunol 168(5):2225–2232
Luther SA, Cyster JG (2001) Chemokines as regulators of T cell differentiation. Nat Immunol 2(2):102
Butrym A, Kryczek I, Dlubek D, Jaskula E, Lange A, Jurczyszyn A, Mazur G (2018) High expression of CC chemokine receptor 5 (CCR5) promotes disease progression in patients with B-cell non-Hodgkin lymphomas. Curr Probl Cancer 42(2):268–275
Liu Y, Poon RT, Feng X, Yu WC, Luk JM, Fan ST (2004) Reduced expression of chemokine receptors on peripheral blood lymphocytes in patients with hepatocellular carcinoma. Am J Gastroenterol 99(6):1111–1121. https://doi.org/10.1111/j.1572-0241.2004.30265.x
Liu Y, Poon RT, Hughes J, Feng X, Yu WC, Fan ST (2005) Chemokine receptors support infiltration of lymphocyte subpopulations in human hepatocellular carcinoma. Clin Immunol 114(2):174–182
Gonzalez-Martin A, Mira E, Manes S (2012) CCR5 in cancer immunotherapy: More than an “attractive” receptor for T cells. Oncoimmunology 1(1):106–108. https://doi.org/10.4161/onci.1.1.17995
Liu YY, Wang Y, Walsh TR, Yi LX, Zhang R, Spencer J, Doi Y, Tian G, Dong B, Huang X, Yu LF, Gu D, Ren H, Chen X, Lv L, He D, Zhou H, Liang Z, Liu JH, Shen J (2016) Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis 16(2):161–168. https://doi.org/10.1016/S1473-3099(15)00424-7
Ren Z, Peng H, Fu Y-X (2016) PD-1 shapes B cells as evildoers in the tumor microenvironment. Cancer Discov 6(5):477–478
Sakuishi K, Ngiow SF, Sullivan JM, Teng MW, Kuchroo VK, Smyth MJ, Anderson AC (2013) TIM3(+)FOXP3(+) regulatory T cells are tissue-specific promoters of T-cell dysfunction in cancer. Oncoimmunology 2(4):e23849. https://doi.org/10.4161/onci.23849
World Health Organization (2017) Global hepatitis report 2017. World Health Organization, Washington
Mekky MA, Abdel-Malek MO, Osman HA, Abdel-Aziz EM, Hashim A-KA, Hetta HF, Morsy KH (2019) Efficacy of ombitasvir/paritaprevir/ritonavir/ribavirin in management of HCV genotype 4 and end-stage kidney disease. Clin Res Hepatol Gastroenterol 43(1):82–87
Mekky MA, Sayed HI, Abdelmalek MO, Saleh MA, Osman OA, Osman HA, Morsy KH, Hetta HF (2019) Prevalence and predictors of occult hepatitis C virus infection among Egyptian patients who achieved sustained virologic response to sofosbuvir/daclatasvir therapy: a multi-center study. Infect Drug Resist 12:273
Mazouz S, Boisvert M, Shoukry NH, Lamarre D (2018) Reversing immune dysfunction and liver damage after direct-acting antiviral treatment for hepatitis C. Can Liver J 1(2):78–105
Abdel-Hameed EA, Rouster SD, Ji H, Ulm A, Hetta HF, Anwar N, Sherman KE, Shata MTM (2016) Evaluating the role of cellular immune responses in the emergence of HCV NS3 resistance mutations during protease inhibitor therapy. Viral Immunol 29(4):252–258
Zhang Y, Cai P, Liang T, Wang L, Hu L (2017) TIM-3 is a potential prognostic marker for patients with solid tumors: a systematic review and meta-analysis. Oncotarget 8(19):31705
Zahran AM, Saleh M, Mostafa F, Sayed MM, Rayan A, Ali AM, Hetta HF (2018) Up-regulation of regulatory T cells, CD200 and TIM3 expression in cytogenetically normal acute myeloid leukemia. Cancer Biomarkers 22(3):587–595
Ma C, Kesarwala AH, Eggert T, Medina-Echeverz J, Kleiner DE, Jin P, Stroncek DF, Terabe M, Kapoor V, ElGindi M (2016) NAFLD causes selective CD4+ T lymphocyte loss and promotes hepatocarcinogenesis. Nature 531(7593):253
Author information
Authors and Affiliations
Contributions
AMZ, HFH, AR and OE-B conceived and designed the experiments. AR, ASE, EAH, HF and AS recruited patients, carried out the clinical investigations and collected patients’ clinical data. AMZ performed the experiments. AMZ, HFH and OE-B shared in the analysis of the flow cytometry data. OE-B. performed the statistical analysis. OE-B and AMZ accomplished the interpretation of results and wrote the initial draft. All authors participated in critical review and revision of the final manuscript, and HFH managed the submission of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interests.
Ethical approval
The research was done according to the ethical guidelines of the Declaration of Helsinki 1975 and was accepted by the local ethics committee of the Faculty of Medicine, Assiut University, Assiut, Egypt (IRB no: 17300300).
Informed consent
Written informed consent was obtained from all participants before inclusion in the study. Participants consented for the use of their specimens and data for research and publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zahran, A.M., Hetta, H.F., Rayan, A. et al. Differential expression of Tim-3, PD-1, and CCR5 on peripheral T and B lymphocytes in hepatitis C virus-related hepatocellular carcinoma and their impact on treatment outcomes. Cancer Immunol Immunother 69, 1253–1263 (2020). https://doi.org/10.1007/s00262-019-02465-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-019-02465-y