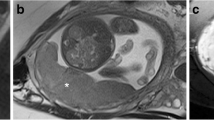
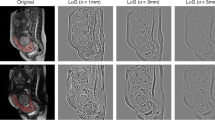
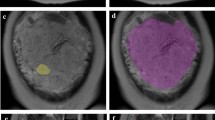
Abstract
Purpose
To retrospectively evaluate the performance of different manual segmentation methods of placenta MR images for predicting Placenta Accreta Spectrum (PAS) disorders in patients with placenta previa (PP) using a Machine Learning (ML) Radiomics analysis.
Methods
64 patients (n=41 with PAS and n= 23 without PAS) with PP who underwent MRI examination for suspicion of PAS were retrospectively selected. All MRI examinations were acquired on a 1.5 T using T2-weighted (T2w) sequences on axial, sagittal and coronal planes. Ten different manual segmentation methods were performed on sagittal placental T2-weighted images obtaining five sets of 2D regions of interest (ROIs) and five sets of 3D volumes of interest (VOIs) from each patient. In detail, ROIs and VOIs were positioned on the following areas: placental tissue, retroplacental myometrium, cervix, placenta with underneath myometrium, placenta with underneath myometrium and cervix. For feature stability testing, the same process was repeated on 30 randomly selected placental MRI examinations by two additional radiologists, working independently and blinded to the original segmentation. Radiomic features were extracted from all available ROIs and VOIs. 100 iterations of 5-fold cross-validation with nested feature selection, based on recursive feature elimination, were subsequently run on each ROI/VOI to identify the best-performing method to classify instances correctly.
Results
Among the segmentation methods, the best performance in predicting PAS was obtained by the VOIs covering the retroplacental myometrium (Mean validation score: 0.761, standard deviation: 0.116).
Conclusion
Our preliminary results show that the VOI including the retroplacental myometrium using T2w images seems to be the best method when segmenting images for the development of ML radiomics predictive models to identify PAS in patients with PP.
Graphical abstract



Similar content being viewed by others
References
R.M. Silver, K.D. Barbour, Placenta Accreta Spectrum, Obstet. Gynecol. Clin. North Am. 42 (2015) 381–402. https://doi.org/https://doi.org/10.1016/j.ogc.2015.01.014.
E. Jauniaux, F. Chantraine, R.M. Silver, J. Langhoff-Roos, FIGO consensus guidelines on placenta accreta spectrum disorders: Epidemiology, Int. J. Gynecol. Obstet. 140 (2018) 265–273. https://doi.org/https://doi.org/10.1002/ijgo.12407.
E. Jauniaux, D. Ayres‐de‐Campos, J. Langhoff‐Roos, K.A. Fox, S. Collins, G. Duncombe, P. Klaritsch, F. Chantraine, J. Kingdom, L. Grønbeck, K. Rull, M. Tikkanen, L. Sentilhes, T. Asatiani, W. Leung, T. AIhaidari, D. Brennan, M. Seoud, A.M. Hussein, R. Jegasothy, K.N. Shah, D. Bomba‐Opon, C. Hubinont, P. Soma‐Pillay, N.T. Mandić, P. Lindqvist, B. Arnadottir, I. Hoesli, R. Cortez, FIGO classification for the clinical diagnosis of placenta accreta spectrum disorders, Int. J. Gynecol. Obstet. 146 (2019) 20–24, doi: https://doi.org/10.1002/ijgo.12761.
J.L. Hecht, R. Baergen, L.M. Ernst, P.J. Katzman, S.M. Jacques, E. Jauniaux, T.Y. Khong, L.A. Metlay, L. Poder, F. Qureshi, J.T. Rabban, D.J. Roberts, S. Shainker, D.S. Heller, Classification and reporting guidelines for the pathology diagnosis of placenta accreta spectrum (PAS) disorders: recommendations from an expert panel, Mod. Pathol. (2020). https://doi.org/https://doi.org/10.1038/s41379-020-0569-1.
H. Imafuku, K. Tanimura, Y. Shi, A. Uchida, M. Deguchi, Y. Terai, Clinical factors associated with a placenta accreta spectrum, Placenta. (2021). https://doi.org/https://doi.org/10.1016/j.placenta.2021.08.001.
R.M. Silver, M.B. Landon, D.J. Rouse, K.J. Leveno, C.Y. Spong, E.A. Thom, A.H. Moawad, S.N. Caritis, M. Harper, R.J. Wapner, Y. Sorokin, M. Miodovnik, M. Carpenter, A.M. Peaceman, M.J. O’Sullivan, B. Sibai, O. Langer, J.M. Thorp, S.M. Ramin, B.M. Mercer, Maternal Morbidity Associated With Multiple Repeat Cesarean Deliveries, Obstet. Gynecol. 107 (2006) 1226–1232. https://doi.org/https://doi.org/10.1097/01.AOG.0000219750.79480.84.
A. Mehrabadi, J.A. Hutcheon, S. Liu, S. Bartholomew, M.S. Kramer, R.M. Liston, K.S. Joseph, Contribution of Placenta Accreta to the Incidence of Postpartum Hemorrhage and Severe Postpartum Hemorrhage, Obstet. Gynecol. 125 (2015) 814–821. https://doi.org/https://doi.org/10.1097/AOG.0000000000000722.
L. Allen, E. Jauniaux, S. Hobson, J. Papillon-Smith, M.A. Belfort, FIGO consensus guidelines on placenta accreta spectrum disorders: Nonconservative surgical management, Int. J. Gynecol. Obstet. 140 (2018) 281–290. https://doi.org/https://doi.org/10.1002/ijgo.12409.
V. Romeo, L. Sarno, A. Volpe, M.I. Ginocchio, R. Esposito, P.P. Mainenti, M. Petretta, R. Liuzzi, M. D’Armiento, P. Martinelli, A. Brunetti, S. Maurea, US and MR imaging findings to detect placental adhesion spectrum (PAS) in patients with placenta previa: a comparative systematic study, Abdom. Radiol. 44 (2019) 3398–3407. https://doi.org/https://doi.org/10.1007/s00261-019-02185-y.
M. De Oliveira Carniello, L.G. Oliveira Brito, L.O. Sarian, J.R. Bennini, Diagnosis of placenta accreta spectrum in high‐risk women using ultrasonography or magnetic resonance imaging: systematic review and meta‐analysis, Ultrasound Obstet. Gynecol. 59 (2022) 428–436, doi: https://doi.org/10.1002/uog.24861.
S. Hong, Y. Le, K.U. Lio, T. Zhang, Y. Zhang, N. Zhang, Performance comparison of ultrasonography and magnetic resonance imaging in their diagnostic accuracy of placenta accreta spectrum disorders: a systematic review and meta-analysis, Insights Imaging. 13 (2022) 1–13. https://doi.org/https://doi.org/10.1186/S13244-022-01192-W/TABLES/2.
S. Maurea, V. Romeo, P.P. Mainenti, M.I. Ginocchio, G. Frauenfelder, F. Verde, R. Liuzzi, M. D’Armiento, L. Sarno, M. Morlando, M. Petretta, P. Martinelli, A. Brunetti, Diagnostic accuracy of magnetic resonance imaging in assessing placental adhesion disorder in patients with placenta previa: Correlation with histological findings, Eur. J. Radiol. 106 (2018) 77–84. https://doi.org/https://doi.org/10.1016/j.ejrad.2018.07.014.
H. Kapoor, M. Hanaoka, A. Dawkins, A. Khurana, Review of MRI imaging for placenta accreta spectrum: Pathophysiologic insights, imaging signs, and recent developments, Placenta. 104 (2021) 31–39. https://doi.org/https://doi.org/10.1016/j.placenta.2020.11.004.
L. Alamo, A. Anaye, J. Rey, A. Denys, G. Bongartz, S. Terraz, S. Artemisia, R. Meuli, S. Schmidt, Detection of suspected placental invasion by MRI: Do the results depend on observer’ experience?, Eur. J. Radiol. 82 (2013) e51–e57. https://doi.org/https://doi.org/10.1016/j.ejrad.2012.08.022.
C.L.A. Ghezzi, C.K. Silva, A.S. Casagrande, S.S. Westphalen, C.C. Salazar, J. Vettorazzi, Diagnostic performance of radiologists with different levels of experience in the interpretation of MRI of the placenta accreta spectrum disorder, Br. J. Radiol. (2021). https://doi.org/https://doi.org/10.1259/bjr.20210827.
S. Maurea, F. Verde, P.P. Mainenti, L. Barbuto, F. Iacobellis, V. Romeo, R. Liuzzi, G. Raia, G. De Dominicis, C. Santangelo, L. Romano, A. Brunetti, Qualitative evaluation of MR images for assessing placenta accreta spectrum disorders in patients with placenta previa: A pilot validation study, Eur. J. Radiol. (2022). https://doi.org/https://doi.org/10.1016/j.ejrad.2021.110078.
P. Jha, L. Pōder, C. Bourgioti, N. Bharwani, S. Lewis, A. Kamath, S. Nougaret, P. Soyer, M. Weston, R.P. Castillo, A. Kido, R. Forstner, G. Masselli, Society of Abdominal Radiology (SAR) and European Society of Urogenital Radiology (ESUR) joint consensus statement for MR imaging of placenta accreta spectrum disorders., Eur. Radiol. 30 (2020) 2604–2615. https://doi.org/https://doi.org/10.1007/s00330-019-06617-7.
P. Lambin, R.T.H. Leijenaar, T.M. Deist, J. Peerlings, E.E.C. de Jong, J. van Timmeren, S. Sanduleanu, R.T.H.M. Larue, A.J.G. Even, A. Jochems, Y. van Wijk, H. Woodruff, J. van Soest, T. Lustberg, E. Roelofs, W. van Elmpt, A. Dekker, F.M. Mottaghy, J.E. Wildberger, S. Walsh, Radiomics: the bridge between medical imaging and personalized medicine, Nat. Rev. Clin. Oncol. 14 (2017) 749–762. https://doi.org/https://doi.org/10.1038/nrclinonc.2017.141.
J. Guiot, A. Vaidyanathan, L. Deprez, F. Zerka, D. Danthine, A. Frix, P. Lambin, F. Bottari, N. Tsoutzidis, B. Miraglio, S. Walsh, W. Vos, R. Hustinx, M. Ferreira, P. Lovinfosse, R.T.H. Leijenaar, A review in radiomics: Making personalized medicine a reality via routine imaging, Med. Res. Rev. 42 (2022) 426–440. https://doi.org/https://doi.org/10.1002/med.21846.
V. Romeo, S. Maurea, The new era of advanced placental tissue characterization using MRI texture analysis: Clinical implications, EBioMedicine. 51 (2020) 102588. https://doi.org/10.1016/j.ebiom.2019.11.049.
V. Romeo, C. Ricciardi, R. Cuocolo, A. Stanzione, F. Verde, L. Sarno, G. Improta, P.P. Mainenti, M. D’Armiento, A. Brunetti, S. Maurea, Machine learning analysis of MRI-derived texture features to predict placenta accreta spectrum in patients with placenta previa, Magn. Reson. Imaging. 64 (2019). https://doi.org/10.1016/j.mri.2019.05.017.
H. Sun, H. Qu, L. Chen, W. Wang, Y. Liao, L. Zou, Z. Zhou, X. Wang, S. Zhou, Identification of suspicious invasive placentation based on clinical MRI data using textural features and automated machine learning., Eur. Radiol. 29 (2019) 6152–6162. https://doi.org/https://doi.org/10.1007/s00330-019-06372-9.
Q. Wu, K. Yao, Z. Liu, L. Li, X. Zhao, S. Wang, H. Shang, Y. Lin, Z. Wen, X. Zhang, J. Tian, M. Wang, Radiomics analysis of placenta on T2WI facilitates prediction of postpartum haemorrhage: A multicentre study, EBioMedicine. 50 (2019) 355–365. https://doi.org/https://doi.org/10.1016/j.ebiom.2019.11.010.
A. Stanzione, R. Cuocolo, L. Ugga, F. Verde, V. Romeo, A. Brunetti, S. Maurea, Oncologic Imaging and Radiomics: A Walkthrough Review of Methodological Challenges, Cancers (Basel). 14 (2022) 4871. https://doi.org/https://doi.org/10.3390/cancers14194871.
C. Ricciardi, R. Cuocolo, F. Verde, G. Improta, A. Stanzione, V. Romeo, S. Maurea, M. D’Armiento, L. Sarno, M. Guida, M. Cesarelli, Resolution Resampling of Ultrasound Images in Placenta Previa Patients: Influence on Radiomics Data Reliability and Usefulness for Machine Learning, in: 2021: pp. 1011–1018. https://doi.org/10.1007/978-3-030-64610-3_113.
A. Zwanenburg, M. Vallières, M.A. Abdalah, H.J.W.L. Aerts, V. Andrearczyk, A. Apte, S. Ashrafinia, S. Bakas, R.J. Beukinga, R. Boellaard, M. Bogowicz, L. Boldrini, I. Buvat, G.J.R. Cook, C. Davatzikos, A. Depeursinge, M.-C. Desseroit, N. Dinapoli, C.V. Dinh, S. Echegaray, I. El Naqa, A.Y. Fedorov, R. Gatta, R.J. Gillies, V. Goh, M. Götz, M. Guckenberger, S.M. Ha, M. Hatt, F. Isensee, P. Lambin, S. Leger, R.T.H. Leijenaar, J. Lenkowicz, F. Lippert, A. Losnegård, K.H. Maier-Hein, O. Morin, H. Müller, S. Napel, C. Nioche, F. Orlhac, S. Pati, E.A.G. Pfaehler, A. Rahmim, A.U.K. Rao, J. Scherer, M.M. Siddique, N.M. Sijtsema, J. Socarras Fernandez, E. Spezi, R.J.H.M. Steenbakkers, S. Tanadini-Lang, D. Thorwarth, E.G.C. Troost, T. Upadhaya, V. Valentini, L. V. van Dijk, J. van Griethuysen, F.H.P. van Velden, P. Whybra, C. Richter, S. Löck, The Image Biomarker Standardization Initiative: Standardized Quantitative Radiomics for High-Throughput Image-based Phenotyping, Radiology. 295 (2020) 328–338. https://doi.org/10.1148/radiol.2020191145.
F. Pedregosa, G. Varoquaux, A. Gramfort, V. Michel, B. Thirion, O. Grisel, M. Blondel, A. Müller, J. Nothman, G. Louppe, P. Prettenhofer, R. Weiss, V. Dubourg, J. Vanderplas, A. Passos, D. Cournapeau, M. Brucher, M. Perrot, É. Duchesnay, Scikit-learn: Machine Learning in Python, (2012).
B. Baeßler, K. Weiss, D. Pinto dos Santos, Robustness and Reproducibility of Radiomics in Magnetic Resonance Imaging, Invest. Radiol. 54 (2019) 221–228. https://doi.org/https://doi.org/10.1097/RLI.0000000000000530.
A. Stanzione, R. Galatola, R. Cuocolo, V. Romeo, F. Verde, P.P. Mainenti, A. Brunetti, S. Maurea, Radiomics in Cross-Sectional Adrenal Imaging: A Systematic Review and Quality Assessment Study, Diagnostics. 12 (2022) 578. https://doi.org/https://doi.org/10.3390/diagnostics12030578.
B. Kocak, E. Ates, E.S. Durmaz, M.B. Ulusan, O. Kilickesmez, Influence of segmentation margin on machine learning–based high-dimensional quantitative CT texture analysis: a reproducibility study on renal clear cell carcinomas, Eur. Radiol. (2019). https://doi.org/https://doi.org/10.1007/s00330-019-6003-8.
X. Zhang, L. Zhong, B. Zhang, L. Zhang, H. Du, L. Lu, S. Zhang, W. Yang, Q. Feng, The effects of volume of interest delineation on MRI-based radiomics analysis: evaluation with two disease groups, Cancer Imaging. 19 (2019) 89. https://doi.org/https://doi.org/10.1186/s40644-019-0276-7.
M. Pavic, M. Bogowicz, X. Würms, S. Glatz, T. Finazzi, O. Riesterer, J. Roesch, L. Rudofsky, M. Friess, P. Veit-Haibach, M. Huellner, I. Opitz, W. Weder, T. Frauenfelder, M. Guckenberger, S. Tanadini-Lang, Influence of inter-observer delineation variability on radiomics stability in different tumor sites, Acta Oncol. (Madr). 57 (2018) 1070–1074. https://doi.org/https://doi.org/10.1080/0284186X.2018.1445283.
S. Gitto, R. Cuocolo, I. Emili, L. Tofanelli, V. Chianca, D. Albano, C. Messina, M. Imbriaco, L.M. Sconfienza, Effects of Interobserver Variability on 2D and 3D CT- and MRI-Based Texture Feature Reproducibility of Cartilaginous Bone Tumors, J. Digit. Imaging. 34 (2021) 820–832. https://doi.org/https://doi.org/10.1007/s10278-021-00498-3.
Q. Qiu, J. Duan, Z. Duan, X. Meng, C. Ma, J. Zhu, J. Lu, T. Liu, Y. Yin, Reproducibility and non-redundancy of radiomic features extracted from arterial phase CT scans in hepatocellular carcinoma patients: Impact of tumor segmentation variability, Quant. Imaging Med. Surg. (2019). https://doi.org/10.21037/qims.2019.03.02.
B. Kocak, E.S. Durmaz, C. Erdim, E. Ates, O.K. Kaya, O. Kilickesmez, Radiomics of Renal Masses: Systematic Review of Reproducibility and Validation Strategies, Am. J. Roentgenol. 214 (2020) 129–136. https://doi.org/https://doi.org/10.2214/AJR.19.21709.
A. Stanzione, F. Verde, R. Cuocolo, V. Romeo, P. Paolo Mainenti, A. Brunetti, S. Maurea, Placenta Accreta Spectrum Disorders and Radiomics: Systematic review and quality appraisal, Eur. J. Radiol. 155 (2022) 110497. https://doi.org/10.1016/j.ejrad.2022.110497.
B.D. Einerson, J. Comstock, R.M. Silver, D.W. Branch, P.J. Woodward, A. Kennedy, Placenta Accreta Spectrum Disorder, Obstet. Gynecol. 135 (2020) 1104–1111. https://doi.org/https://doi.org/10.1097/AOG.0000000000003793.
E. Jauniaux, D. Jurkovic, A.M. Hussein, G.J. Burton, New insights into the etiopathology of placenta accreta spectrum, Am. J. Obstet. Gynecol. 227 (2022) 384–391. https://doi.org/https://doi.org/10.1016/j.ajog.2022.02.038.
A.M. Hussein, R.A. Elbarmelgy, R.M. Elbarmelgy, M.M. Thabet, E. Jauniaux, Prospective evaluation of impact of <scp>post‐Cesarean</scp> section uterine scarring in perinatal diagnosis of placenta accreta spectrum disorder, Ultrasound Obstet. Gynecol. 59 (2022) 474–482. https://doi.org/https://doi.org/10.1002/uog.23732.
E. Jauniaux, A.M. Hussein, N. Zosmer, R.M. Elbarmelgy, R.A. Elbarmelgy, H. Shaikh, G.J. Burton, A new methodologic approach for clinico-pathologic correlations in invasive placenta previa accreta, Am. J. Obstet. Gynecol. 222 (2020) 379.e1-379.e11. https://doi.org/https://doi.org/10.1016/j.ajog.2019.11.1246.
E. Jauniaux, A.M. Hussein, R.M. Elbarmelgy, R.A. Elbarmelgy, G.J. Burton, Failure of placental detachment in accreta placentation is associated with excessive fibrinoid deposition at the utero-placental interface, Am. J. Obstet. Gynecol. (2022). https://doi.org/https://doi.org/10.1016/j.ajog.2021.08.026.
Author information
Authors and Affiliations
Contributions
I confirm that all the authors have contributed significantly to this manuscript, have seen and approved the final manuscript, and have agreed to its submission to the Abdominal Radiology.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Ethical approval
IRB approved.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Verde, F., Stanzione, A., Cuocolo, R. et al. Segmentation methods applied to MRI-derived radiomic analysis for the prediction of placenta accreta spectrum in patients with placenta previa. Abdom Radiol 48, 3207–3215 (2023). https://doi.org/10.1007/s00261-023-03963-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-023-03963-5