Abstract
Purpose
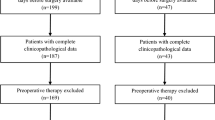
Tumor size is an important prognostic factor without consideration of the necrotic and cystic components within tumor for patients with gastrointestinal stromal tumors (GISTs). We aimed to extract the enhancing viable component from the tumor using computed tomography (CT) post-processing software and evaluate the value of preoperative CT features for predicting the disease-free survival (DFS) after curative resection for patients with primary gastric GISTs.
Methods
132 Patients with primary gastric GISTs who underwent preoperative contrast-enhanced CT and curative resection were retrospectively analyzed. We used a certain CT attenuation of 30 HU to extract the enhancing tissue component from the tumor. Enhancing tissue volume and other CT features were assessed on venous-phase images. We evaluated the value of preoperative CT features for predicting the DFS after surgery. Univariate and multivariate Cox regression analyses were performed to find the independent risk factor for predicting the DFS.
Results
Of the 132 patients, 68 were males and 64 were females, with a mean age of 61 years. The median follow-up duration was 60 months, and 28 patients experienced disease recurrence and distant metastasis during the follow-up period. Serosal invasion (p < 0.001; HR = 5.277) and enhancing tissue volume (p = 0.005; HR = 1.447) were the independent risk factors for predicting the DFS after curative resection for patients with primary gastric GISTs.
Conclusion
Preoperative contrast-enhanced CT could be useful for predicting the DFS after the surgery of gastric GISTs, and serosal invasion and enhancing tissue volume were the independent risk factors.



Similar content being viewed by others
Abbreviations
- GISTs:
-
Gastrointestinal stromal tumors
- CT:
-
Computed tomography
- DFS:
-
Disease-free survival
- 3D:
-
Three-dimensional
- ICC:
-
Interclass and intraclass correlation coefficients
- TNM:
-
Tumor node metastasis
- Mean attenuation_T:
-
Mean attenuation value of total tumor
- Mean attenuation_E:
-
Mean attenuation value of enhancing tissue
- RFS:
-
Recurrence-free survival
References
Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S (1998). Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science 279:577–580. doi: https://doi.org/10.1126/science.279.5350.577.
Joensuu H, Vehtari A, Riihimäki J, Nishida T, Steigen S. E, Brabec P (2012). Risk of recurrence of gastrointestinal stromal tumour after surgery: an analysis of pooled population-based cohorts. Lancet Oncol 13:265–274. doi: https://doi.org/10.1016/s1470-2045(11)70299-6.
DeMatteo RP, Lewis JJ, Leung D, Mudan SS, Woodruff JM, Brennan MF (2000). Two hundred gastrointestinal stromal tumors: recurrence patterns and prognostic factors for survival. Ann Surg 231:51–58. doi: https://doi.org/10.1097/00000658-200001000-00008.
Lin Y, Wang M, Jia J, Wan W, Wang T, Yang W (2020). Development and validation of a prognostic nomogram to predict recurrence in high-risk gastrointestinal stromal tumour: A retrospective analysis of two independent cohorts. EBioMedicine 60:103016. doi: https://doi.org/10.1016/j.ebiom.2020.103016.
Blanke CD, Demetri GD, von Mehren M, Heinrich MC, Eisenberg B, Fletcher JA (2008). Long-term results from a randomized phase II trial of standard- versus higher-dose imatinib mesylate for patients with unresectable or metastatic gastrointestinal stromal tumors expressing KIT. J Clin Oncol 26:620–625. doi: https://doi.org/10.1200/jco.2007.13.4403.
Joensuu H, Hohenberger P, Corless CL (2013). Gastrointestinal stromal tumour. Lancet 382:973–983. doi: https://doi.org/10.1016/s0140-6736(13)60106-3.
Joensuu H (2008). Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol 39:1411–1419. doi: https://doi.org/10.1016/j.humpath.2008.06.025.
Fletcher CDM, Bridge JA, P.C.W. Hogendoorn (2013) WHO Classification of Tumours of Soft Tissue and Bone. 4.
von Mehren M, Randall RL, Benjamin RS, Boles S, Bui MM, Ganjoo KN, et al. Soft Tissue Sarcoma, Version 2.2018 (2018) Journal of the National Comprehensive Cancer Network 16(5): 536–563.
Miettinen M, Lasota J (2006) Gastrointestinal stromal tumors: Pathology and prognosis at different sites. Seminars in Diagnostic Pathology 23(2): 70–83.
Liu X, Qiu H, Zhang P, et al (2017) China Gastrointestinal Stromal Tumor Study, Group. Prognostic role of tumor necrosis in patients undergoing curative resection for gastric gastrointestinal stromal tumor: a multicenter analysis of 740 cases in China. Cancer Med 6: 2796–2803. doi: https://doi.org/10.1002/cam4.1229.
Tyler R, Davies E, Tan D, et al (2021) Tumor necrosis is significantly associated with reduced recurrence-free survival after curative resection of gastrointestinal stromal tumors. J Surg Oncol 123: 432–438. doi: https://doi.org/10.1002/jso.26294.
Xue A, Yuan W, Gao X, et al (2019) Gastrointestinal stromal tumors (GISTs) with remarkable cystic change: a specific subtype of GISTs with relatively indolent behaviors and favorable prognoses. J Cancer Res Clin Oncol 145: 1559–1568. doi: https://doi.org/10.1007/s00432-019-02853-y.
Casali PG, Abecassis N, Aro HT, et al (2018) Gastrointestinal stromal tumours: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 29: iv68–iv78. doi: https://doi.org/10.1093/annonc/mdy095.
Zhou C, Duan X, Zhang X, Hu H, Wang D, Shen J (2016) Predictive features of CT for risk stratifications in patients with primary gastrointestinal stromal tumour. Eur Radiol 26: 3086–3093. doi: https://doi.org/10.1007/s00330-015-4172-7.
Cannella R, Tabone E, Porrello G, et al (2021) Assessment of morphological CT imaging features for the prediction of risk stratification, mutations, and prognosis of gastrointestinal stromal tumors. Eur Radiol 31: 8554–8564. doi: https://doi.org/10.1007/s00330-021-07961-3.
Chen XS, Shan YC, Dong SY, et al (2021) Utility of preoperative computed tomography features in predicting the Ki-67 labeling index of gastric gastrointestinal stromal tumors. Eur J Radiol 142: 109840. doi: https://doi.org/10.1016/j.ejrad.2021.109840.
Camp RL, Dolled-Filhart M, Rimm DL (2004) X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res 10: 7252–7259. doi: https://doi.org/10.1158/1078-0432.Ccr-04-0713.
Sasaki K, Morioka D, Conci S, et al (2018) The Tumor Burden Score: A New “Metro-ticket” Prognostic Tool for Colorectal Liver Metastases Based on Tumor Size and Number of Tumors. Ann Surg 267: 132–141. doi: https://doi.org/10.1097/SLA.0000000000002064.
Chen MK, Chen TH, Liu JP, Chang CC, Chie WC (2004) Better prediction of prognosis for patients with nasopharyngeal carcinoma using primary tumor volume. Cancer 100:2160–2166. doi: https://doi.org/10.1002/cncr.20210.
Ikai I, Takayasu K, Omata M, Okita K, Nakanuma Y, Matsuyama Y (2006) A modified Japan Integrated Stage score for prognostic assessment in patients with hepatocellular carcinoma. J Gastroenterol 41:884–892. doi: https://doi.org/10.1007/s00535-006-1878-y.
Li, C, Fu, W, Huang, L, et al (2021) A CT-based nomogram for predicting the malignant potential of primary gastric gastrointestinal stromal tumors preoperatively. Abdom Radiol 46: 3075–3085. doi: https://doi.org/10.1007/s00261-021-03026-7
D'Ambrosio L, Palesandro E, Boccone P, Tolomeo F, Miano S, Galizia D (2017) Impact of a risk-based follow-up in patients affected by gastrointestinal stromal tumour. Eur J Cancer 78:122–132. doi: https://doi.org/10.1016/j.ejca.2017.03.025.
O'Neill AC, Shinagare AB, Kurra V, Tirumani SH, Jagannathan JP, Baheti AD (2016) Assessment of metastatic risk of gastric GIST based on treatment-naive CT features. Eur J Surg Oncol 42:1222–1228. doi: https://doi.org/10.1016/j.ejso.2016.03.032.
Choi H, Charnsangavej C, Faria SC, Macapinlac HA, Burgess MA, Patel SR (2007) Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with imatinib mesylate: proposal of new computed tomography response criteria. J Clin Oncol 25:1753–1759. doi: https://doi.org/10.1200/jco.2006.07.3049.
Amin MB, Edge SB (2017) AJCC cancer staging manual. Springer.
Zhao WY, Xu J, Wang M, Zhang ZZ, Tu L, Wang CJ (2014) Evaluation of high-risk clinicopathological indicators in gastrointestinal stromal tumors for prognosis and imatinib treatment outcome. BMC Gastroenterol 14:105. doi: https://doi.org/10.1186/1471-230x-14-105.
Chen T, Xu L, Dong X, Li Y, Yu J, Xiong W (2019) The roles of CT and EUS in the preoperative evaluation of gastric gastrointestinal stromal tumors larger than 2 cm. Eur Radiol 29:2481–2489. doi: https://doi.org/10.1007/s00330-018-5945-6.
Iitsuka Y, Kaneshima S, Tanida O, Takeuchi T, Koga S (1979) Intraperitoneal free cancer cells and their viability in gastric cancer. Cancer 44: 1476–1480. doi: https://doi.org/10.1002/1097-0142(197910)44:4<1476:aid-cncr2820440442>3.0.co;2-r.
Mazzei MA, Squitieri NC, Vindigni C, et al (2020) Gastrointestinal stromal tumors (GIST): a proposal of a “CT-based predictive model of Miettinen index” in predicting the risk of malignancy. Abdominal Radiology 45: 2989–2996. doi: https://doi.org/10.1007/s00261-019-02209-7.
Funding
This paper is supported by China International Medical Foundation (Grant Number Z-2014-07-2003-04).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors state that this work has not received any funding and declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, XS., Yuan, W., Xu, ZH. et al. Prognostic value of preoperative CT features for disease-free survival in patients with primary gastric gastrointestinal stromal tumors after resection. Abdom Radiol 48, 494–501 (2023). https://doi.org/10.1007/s00261-022-03725-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-022-03725-9