Abstract
Purpose
To evaluate the validity of PI-RADS categories 1 and 2 version 2.1 (V2.1) as predictors of the absence of carcinoma and to reevaluate lesions that were analysed as suspicious prior to PI-RADS or according to PI-RADS versions 1 and 2 and classified as PI-RADS 1 or 2 in V2.1.
Methods
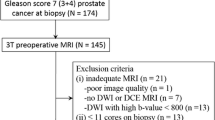
Retrospective evaluation of 1170 multiparametric MRIs performed at one academic teaching hospital (2012–2019). Study cohort comprised 188 men that achieved PI-RADS scores 1 or 2 (V2.1) and underwent systematic and targeted biopsy, split into one group with suspect findings in the original reports that were created prior to PI-RADS or with version 1 and 2, and another group with unremarkable reports. Differences in presence of prostate cancer and PSA density were assessed by Chi-square and Fisher’s exact test, and the negative predictive value (NPV) for both groups was conducted.
Results
The NPV for clinically significant carcinoma (csCa) was 89.1% for 55 men with suspect findings in the original report and 93.2% for 133 men with negative MRI. There was no difference between the groups regarding the detection of csCa (p = 0.103). PSA density was significantly higher in the group with suspect original reports (p = 0.015).
Conclusion
A PI-RADS score 1 or 2 appears less likely to miss existing prostate cancer, although a small amount of csCa can be overlooked. In case of clinical suspicion or elevated PSA density and PI-RADS score 1 or 2, an individual decision has to be taken if biopsy is necessary or if monitoring is sufficient.
Graphical abstract




Similar content being viewed by others
References
German Cancer Society, German Cancer Aid, AWMF (2021) S3-Leitlinie Prostatakarzinom, Langversion 6.0 [S3-guideline Prostate Carcinoma, long version 6.0]. http://www.leitlinienprogramm-onkologie.de/leitlinien/prostatakarzinom/. Accessed 26 Jun 2021
Mottet N, Bellmunt J, Bolla M et al. (2017) EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol 71:618–629. https://doi.org/10.1016/j.eururo.2016.08.003
Barentsz JO, Richenberg J, Clements R et al. (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22:746–757. https://doi.org/10.1007/s00330-011-2377-y
Weinreb JC, Barentsz JO, Choyke PL et al. (2016) PI-RADS Prostate Imaging - Reporting and Data System: 2015, Version 2. Eur Urol 69:16–40. https://doi.org/10.1016/j.eururo.2015.08.052
Turkbey B, Rosenkrantz AB, Haider MA et al. (2019) Prostate Imaging Reporting and Data System Version 2.1: 2019 Update of Prostate Imaging Reporting and Data System Version 2. Eur Urol 76:340–351. https://doi.org/10.1016/j.eururo.2019.02.033
Chen M, Dang H-D, Wang J-Y et al. (2008) Prostate cancer detection: comparison of T2-weighted imaging, diffusion-weighted imaging, proton magnetic resonance spectroscopic imaging, and the three techniques combined. Acta Radiol 49:602–610. https://doi.org/10.1080/02841850802004983
Visschere P de, Oosterlinck W, Meerleer G de et al. (2010) Clinical and imaging tools in the early diagnosis of prostate cancer, a review. JBR-BTR 93:62–70. https://doi.org/10.5334/jbr-btr.121
Tanimoto A, Nakashima J, Kohno H et al. (2007) Prostate cancer screening: the clinical value of diffusion-weighted imaging and dynamic MR imaging in combination with T2-weighted imaging. J Magn Reson Imaging 25:146–152. https://doi.org/10.1002/jmri.20793
D'Amico AV, Whittington R, Malkowicz SB et al. (1998) Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 280:969–974. https://doi.org/10.1001/jama.280.11.969
Westphalen AC, Fazel F, Nguyen H et al. (2019) Detection of clinically significant prostate cancer with PIRADS v2 scores, PSA density, and ADC values in regions with and without mpMRI visible lesions. Int Braz J Urol 45:713–723. https://doi.org/10.1590/S1677-5538.IBJU.2018.0768
Liu C, Liu S-L, Wang Z-X et al. (2018) Using the prostate imaging reporting and data system version 2 (PI-RIDS v2) to detect prostate cancer can prevent unnecessary biopsies and invasive treatment. Asian J Androl 20:459–464. https://doi.org/10.4103/aja.aja_19_18
Anastay V, Gondran-Tellier B, McManus R et al. (2020) Nonsuspicious prebiopsy multiparametric MRI: is prostate biopsy still necessary? Abdom Radiol (NY) 45:4160–4165. https://doi.org/10.1007/s00261-020-02728-8
Buisset J, Norris JM, Puech P et al. (2021) Negative Prebiopsy Magnetic Resonance Imaging and Risk of Significant Prostate Cancer: Baseline and Long-Term Followup Results. J Urol 205:725–731. https://doi.org/10.1097/JU.0000000000001414
Moldovan PC, van den Broeck T, Sylvester R et al. (2017) What Is the Negative Predictive Value of Multiparametric Magnetic Resonance Imaging in Excluding Prostate Cancer at Biopsy? A Systematic Review and Meta-analysis from the European Association of Urology Prostate Cancer Guidelines Panel. Eur Urol 72:250–266. https://doi.org/10.1016/j.eururo.2017.02.026
Santok GDR, Abdel Raheem A, Kim LH et al. (2017) Prostate-specific antigen 10-20 ng/mL: A predictor of degree of upgrading to ≥8 among patients with biopsy Gleason score 6. Investig Clin Urol 58:90–97. https://doi.org/10.4111/icu.2017.58.2.90
van der Kwast, Theo H, Amin MB, Billis A et al. (2011) International Society of Urological Pathology (ISUP) Consensus Conference on Handling and Staging of Radical Prostatectomy Specimens. Working group 2: T2 substaging and prostate cancer volume. Mod Pathol 24:16–25. https://doi.org/10.1038/modpathol.2010.156
Nilsson P, Ströberg P (2019) Are TRUS-guided prostate biopsies in clinical practice robust enough to make a correct assessment of the surgical strategy in prostatectomies? Poor correlation between preoperative prostate biopsies and postoperative specimens. Scand J Urol 53:282–286. https://doi.org/10.1080/21681805.2019.1653362
Baur ADJ, Maxeiner A, Franiel T et al. (2014) Evaluation of the prostate imaging reporting and data system for the detection of prostate cancer by the results of targeted biopsy of the prostate. Invest Radiol 49:411–420. https://doi.org/10.1097/RLI.0000000000000030
Ryznarová Z, Keller J, Záleský M et al. (2019) Comparison of Prostate Imaging Reporting and Data System (PI-RADS) version 1 and version 2 and combination with apparent diffusion coefficient as a predictor of biopsy outcome. Neuro Endocrinol Lett 40:41–50
Kam J, Yuminaga Y, Krelle M et al. (2019) Evaluation of the accuracy of multiparametric MRI for predicting prostate cancer pathology and tumour staging in the real world: an multicentre study. BJU Int 124:297–301. https://doi.org/10.1111/bju.14696
American College of Radiology (2019) Revisions in PI-RADS v2.1. https://www.acr.org/-/media/ACR/Files/RADS/Pi-RADS/ACR-PIRADS-V21Final-Slides.pdf?la=en. Accessed 26 Jun 2021
Oto A, Kayhan A, Jiang Y et al. (2010) Prostate cancer: differentiation of central gland cancer from benign prostatic hyperplasia by using diffusion-weighted and dynamic contrast-enhanced MR imaging. Radiology 257:715–723. https://doi.org/10.1148/radiol.10100021
Girouin N, Mège-Lechevallier F, Tonina Senes A et al. (2007) Prostate dynamic contrast-enhanced MRI with simple visual diagnostic criteria: is it reasonable? Eur Radiol 17:1498–1509. https://doi.org/10.1007/s00330-006-0478-9
Sureka B, Elhence P, Khera PS et al. (2019) Quantitative contrast-enhanced perfusion kinetics in multiparametric MRI in differentiating prostate cancer from chronic prostatitis: results from a pilot study. Br J Radiol 92:20190181. https://doi.org/10.1259/bjr.20190181
Uysal A, Karaosmanoğlu AD, Karcaaltıncaba M et al. (2020) Prostatitis, the Great Mimicker of Prostate Cancer: Can We Differentiate Them Quantitatively With Multiparametric MRI? AJR Am J Roentgenol 215:1104–1112. https://doi.org/10.2214/AJR.20.22843
Kitzing YX, Prando A, Varol C et al. (2016) Benign Conditions That Mimic Prostate Carcinoma: MR Imaging Features with Histopathologic Correlation. Radiographics 36:162–175. https://doi.org/10.1148/rg.2016150030
Gaur S, Harmon S, Gupta RT et al. (2019) A Multireader Exploratory Evaluation of Individual Pulse Sequence Cancer Detection on Prostate Multiparametric Magnetic Resonance Imaging (MRI). Acad Radiol 26:5–14. https://doi.org/10.1016/j.acra.2018.03.024
Turnbull LW, Buckley DL, Turnbull LS et al. (1999) Differentiation of prostatic carcinoma and benign prostatic hyperplasia: correlation between dynamic Gd-DTPA-enhanced MR imaging and histopathology. J Magn Reson Imaging 9:311–316. https://doi.org/10.1002/(sici)1522-2586(199902)9:2<311:aid-jmri24>3.0.co;2-w
Distler FA, Radtke JP, Bonekamp D et al. (2017) The Value of PSA Density in Combination with PI-RADS™ for the Accuracy of Prostate Cancer Prediction. J Urol 198:575–582. https://doi.org/10.1016/j.juro.2017.03.130
Nordström T, Akre O, Aly M et al. (2018) Prostate-specific antigen (PSA) density in the diagnostic algorithm of prostate cancer. Prostate Cancer Prostatic Dis 21:57–63. https://doi.org/10.1038/s41391-017-0024-7
Woo S, Suh CH, Kim SY et al. (2018) Head-To-Head Comparison Between High- and Standard-b-Value DWI for Detecting Prostate Cancer: A Systematic Review and Meta-Analysis. AJR Am J Roentgenol 210:91–100. https://doi.org/10.2214/AJR.17.18480
Funding
No funds, grants or other support were received.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Katja Bogner, Karl Engelhard and Sajad Hamel. The first draft of the manuscript was written by Katja Bogner and all authors commented on previous versions of the manuscript. Wolfgang Wuest and Katja Bogner revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Disclosures
Wolfgang Wuest: Siemens speaker bureau and consultancy. The other authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Friedrich-Alexander-University Erlangen-Nuremberg (Approval Number 384_20 Bc) and informed research consent was waived.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bogner, K., Engelhard, K., Wuest, W. et al. Prostate cancer in PI-RADS scores 1 and 2 version 2.1: a comparison to previous PI-RADS versions. Abdom Radiol 47, 2187–2196 (2022). https://doi.org/10.1007/s00261-022-03444-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-022-03444-1