Abstract
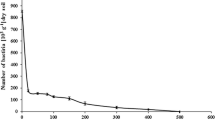
Arthrobacter strain ATCC 33790, a pentachlorophenol (PCP)-metabolizer isolated by the author, has been recovered after 10 years of storage. The freeze-dried preparation grown on half-strength Trypticase Soy Broth adapted to utilize PCP within 1 week. Cultures grown on PCP-nutrient agar were found to utilize PCP in mineral salts medium within 2–3 days. The culture was prepared for continuous growth at pH 6.5 by successive feeding of 100–110 mg solid aliquots of PCP to a 1-l culture initially grown at pH 7.4. Continuous culture growth at pH 6.5 was possible on a mineral salts feed containing 1800 ppm PCP. Continuous cultures grown at pH 6.7 on mineral salts feeds containing 500 and 340 mg PCP/l were especially efficient in removing PCP. Less than 4 mg PCP/l were detected in the effluent at dilution rates near washout. In batch culture studies at pH 6.5 the PCP utilization kinetics were found to be similar at low PCP concentration to those at pH 7.4 for the approximately same inoculum size. Utilization of 35 mg PCP/l was very slow at pH 6.0. Growth rates at pH 6.5 at controlled PCP concentration ranges of 5–35 and 75–115 mg/l were 0.09 h−1 and 0.05 h−1, respectively. The ability of strain ATCC 33790 to utilize PCP in mineral salts media containing naphthalene, methylnaphthalenes, and cresols was examined. Naphthalene, 1-, and 2-methylnaphthalenes at their solubility limit, and o- and m-cresols at 900–1000 mg/l prevented utilization of 80–90 mg PCP/l. PCP was rapidly removed from both commercial sand at 30°C and from clay soil at room temperature. Estimated inoculum sizes of 6.6 × 106, 6.6 × 104, and 656 cells/g were found to be effective in removing approximately half the starting amount of PCP from sand in 3, 19, and 42 h, respectively. Nearly complete disappearance of extractable PCP was observed after 1 day in clay soil inoculated with 6 × 106 cells/g.
Similar content being viewed by others
References
Crawford RL, Mohn WW (1985) Microbiological removal of pentachlorophenol from soil using a Flavobacterium. Enzyme Microb Technol 7:617–620
Edgehill RU (1982) Microbial treatment of water and soil to remove pentachlorophenol. Ph. D. thesis, Cornell University
Edgehill RU (1992) Factors influencing the success of bioremediation. Aust Biotechnol 2:297–301
Edgehill RU, Finn RK (1982) Isolation, characterization, and growth kinetics of bacteria metabolizing pentachlorophenol. Eur J Appl Microbiol Biotechnol 16:179–184
Edgehill RU, Finn RK (1983a) Activated sludge treatment of synthetic wastewater containing pentachlorophenol. Biotechnol Bioeng 25:2165–2176
Edgehill RU, Finn RK (1983b) Microbial treatment of soil to remove pentachlorophenol. Appl Environ Microbiol 45:1122–1125
Lamar RT, Glaser JA, Kirk TK (1990) Fate of pentachlorophenol (PCP) in sterile soils inoculated with the white-rot basidiomycete Phanerochaete chrysosporium: mineralisation, volatilization, and depletion of PCP. Soil Biol Biochem 22:433–440
Middeldorp PJM, Briglia M, Salkinoja-Salonen MS (1990) Biodegradation of pentachlorophenol in natural soil by inoculated Rhodococcus chlorophenolicus. Microb Ecol 23:123–139
Mueller JG, Chapman PJ, Pritchard PH (1989) Creosote-contaminated sites: their potential for bioremediation. Environ Sci Technol 23:1197–1201
Simkins S, Alexander M (1984) Models for mineralization kinetics with the variables of substrate concentration and population density. Appl Environ Microbiol 47:1299–1306
Stanlake GJ, Finn RK (1982) Isolation and characterization of a pentachlorophenol-degrading bacterium. Appl Environ Microbiol 44:1421–1427
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Edgehill, R.U. Pentachlorophenol removal from slightly acidic mineral salts, commercial sand, and clay soil by recovered Arthrobacter strain ATCC 33790. Appl Microbiol Biotechnol 41, 142–148 (1994). https://doi.org/10.1007/BF00166097
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00166097