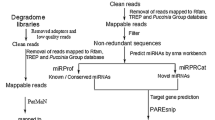
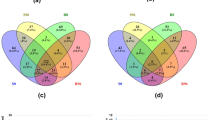
Abstract
Wheat rusts remain a major threat to global wheat production and food security. The R-gene-mediated resistance has been employed as an efficient approach to develop rust-resistant varieties. However, evolution of new fungal races and infection strategies put forward the urgency of unravelling novel molecular players, including non-coding RNAs for plant response. This study identified microRNAs associated with Sr36 and Lr45 disease resistance genes in response to stem and leaf rust, respectively. Here, small RNA sequencing was performed on susceptible and resistant wheat near-isogenic lines inoculated with stem and leaf rust pathotypes. microRNA mining in stem rust–inoculated cultivars revealed a total of distinct 26 known and 7 novel miRNAs, and leaf rust libraries culminated with 22 known and 4 novel miRNAs. The comparative analysis between two disease sets provides a better understanding of altered miRNA profiles associated with respective R-genes and infections. Temporal differential expression pattern of miRNAs pinpoints their role during the progress of infection. Differential expression pattern of miRNAs among various treatments as well as time-course expression of miRNAs revealed stem and leaf rust–responsive miRNAs and their possible role in balancing disease resistance/susceptibility. Disclosure of guide strand, passenger strand and a variant of novel-Tae-miR02 from different subgenome origins might serve as a potential link between stem and leaf rust defence mechanisms downstream to respective R-genes. The outcome from the analysis of microRNA dynamics among two rust diseases and further characterization of identified microRNAs can contribute to significant novel insights on wheat-rust interactions and rust management.
Key points
• Identification and comparative analysis of stem and leaf rust–responsive miRNAs.
• Chromosomal location and functional prediction of miRNAs.
• Time-course expression analysis of pathogen-responsive miRNAs.
Graphical abstract







Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available in the DDBJ repository, under the BioProject Accession number: PRJDB11752 (www.ddbj.nig.ac.jp).
References
Abuqamar S, Ajeb S, Sham A, Enan MR, Iratni R (2013) A mutation in the expansin-like A2 gene enhances resistance to necrotrophic fungi and hypersensitivity to abiotic stress in Arabidopsis thaliana. Mol Plant Pathol 14:813–827. https://doi.org/10.1111/mpp.12049
Ahmed F, Senthil-Kumar M, Lee S, Dai X, Mysore KS, Zhao PX (2014) Comprehensive analysis of small RNA-seq data reveals that combination of miRNA with its isomiRs increase the accuracy of target prediction in Arabidopsis thaliana. RNA Biol 11:1414–1429. https://doi.org/10.1080/15476286.2014.996474
Alaux M, Rogers J, Letellier T, Flores R, Alfama F, Pommier C, Mohellibi N, Durand S, Kimmel E, Michotey C, Guerche C, Loaec M, Lainé M, Steinbach D, Choulet F, Rimbert H, Leroy P, Guilhot N, Salse J, Feuillet C, Paux E, Eversole K, Adam-Blondon A-F, Quesneville H, Consortium IWGS (2018) Linking the international wheat genome sequencing consortium bread wheat reference genome sequence to wheat genetic and phenomic data. Genome Biol 19:111. https://doi.org/10.1186/s13059-018-1491-4
Axtell MJ, Meyers BC (2018) Revisiting criteria for plant microRNA annotation in the era of big data. Plant Cell 30:272–284
Baranova OA, Lapochkina IF, Anisimova AV, Gajnullin NR, Iordanskaya IV, Makarova IY (2016) Identification of Sr genes in new common wheat sources of resistance to stem rust race Ug99 using molecular markers. Russ J Genet Appl Res 6:344–350
Bhardwaj SC, Singh GP, Gangwar OP, Prasad P, Kumar S (2019) Status of wheat rust research and progress in rust management-Indian context. Agronomy 9:892
Budak H, Akpinar BA (2015) Plant miRNAs: biogenesis, organization and origins. Funct Integr Genomics 15:523–531
Burkhardt A, Day B (2016) Transcriptome and small RNAome dynamics during a resistant and susceptible interaction between cucumber and downy mildew. Plant Genome 9:plantgenome2015.08.0069. https://doi.org/10.3835/plantgenome2015.08.0069
Campo S, Peris-Peris C, Siré C, Moreno AB, Donaire L, Zytnicki M, Notredame C, Llave C, San Segundo B (2013) Identification of a novel microRNA (miRNA) from rice that targets an alternatively spliced transcript of the Nramp6 (Natural resistance-associated macrophage protein 6) gene involved in pathogen resistance. New Phytol 199:212–227. https://doi.org/10.1111/nph.12292
Chen Z (2001) A superfamily of proteins with novel cysteine-rich repeats. Plant Physiol 126:473–476. https://doi.org/10.1104/pp.126.2.473
Chen L, Zhang Y-H, Zheng M, Huang T, Cai Y-D (2016) Identification of compound-protein interactions through the analysis of gene ontology, KEGG enrichment for proteins and molecular fragments of compounds. Mol Genet Genomics 291:2065–2079
Chisholm ST, Coaker G, Day B, Staskawicz BJ (2006) Host-microbe interactions: shaping the evolution of the plant immune response. Cell 124:803–814
Conesa A, Götz S (2008) Blast2GO: a comprehensive suite for functional analysis in plant genomics. Int J Plant Genomics 2008:619832
Cosgrove DJ (2015) Plant expansins: diversity and interactions with plant cell walls. Curr Opin Plant Biol 25:162–172. https://doi.org/10.1016/j.pbi.2015.05.014
Dai X, Zhuang Z, Zhao PX (2018) psRNATarget: a plant small RNA target analysis server (2017 release). Nucleic Acids Res 46:W49–W54
Djami-Tchatchou AT, Sanan-Mishra N, Ntushelo K, Dubery IA (2017) Functional roles of microRNAs in agronomically important plants-potential as targets for crop improvement and protection. Front Plant Sci 8:378. https://doi.org/10.3389/fpls.2017.00378
Dutta S, Jha SK, Prabhu KV, Kumar M, Mukhopadhyay K (2019a) Leaf rust (Puccinia triticina) mediated RNAi in wheat (Triticum aestivum L.) prompting host susceptibility. Funct Integr Genomics 19:437–452. https://doi.org/10.1007/s10142-019-00655-6
Dutta S, Kumar M, Mukhopadhyay K (2019b) Identification and validation of leaf rust responsive wheat isomiRs and their target genes in both wheat and Puccinia triticina. Trop Plant Biol 12:318–335. https://doi.org/10.1007/s12042-019-09235-1
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Ellis JG, Lagudah ES, Spielmeyer W, Dodds PN (2014) The past, present and future of breeding rust resistant wheat. Front Plant Sci 5:641
Fang X, Qi Y (2016) RNAi in plants: an argonaute-centered view. Plant Cell 28:272–285. https://doi.org/10.1105/tpc.15.00920
Friedländer MR, Mackowiak SD, Li N, Chen W, Rajewsky N (2012) miRDeep2 accurately identifies known and hundreds of novel microRNA genes in seven animal clades. Nucleic Acids Res 40:37–52
Fujimoto S, Matsunaga S, Yonemura M, Uchiyama S, Azuma T, Fukui K (2004) Identification of a novel plant MAR DNA binding protein localized on chromosomal surfaces. Plant Mol Biol 56:225–239. https://doi.org/10.1007/s11103-004-3249-5
Fujita Y, Fujita M, Shinozaki K, Yamaguchi-Shinozaki K (2011) ABA-mediated transcriptional regulation in response to osmotic stress in plants. J Plant Res 124:509–525. https://doi.org/10.1007/s10265-011-0412-3
Gadjev I, Vanderauwera S, Gechev TS, Laloi C, Minkov IN, Shulaev V, Apel K, Inzé D, Mittler R, Van Breusegem F (2006) Transcriptomic footprints disclose specificity of reactive oxygen species signaling in Arabidopsis. Plant Physiol 141:436–445. https://doi.org/10.1104/pp.106.078717
Gao F, Wang K, Liu Y, Chen Y, Chen P, Shi Z, Luo J, Jiang D, Fan F, Zhu Y, Li S (2015) Blocking miR396 increases rice yield by shaping inflorescence architecture. Nat Plants 2:15196
Garland SH, Lewin L, Abedinia M, Henry R, Blakeney A (1999) The use of microsatellite polymorphisms for the identification of Australian breeding lines of rice (Oryza sativa L.). Euphytica 108:53–63
Garretón V, Carpinelli J, Jordana X, Holuigue L (2002) The as-1 promoter element is an oxidative stress-responsive element and salicylic acid activates it via oxidative species. Plant Physiol 130:1516–1526. https://doi.org/10.1104/pp.009886
Gidhi A, Kumar M, Mukhopadhyay K (2021) The auxin response factor gene family in wheat (Triticum aestivum L.): Genome-wide identification, characterization and expression analyses in response to leaf rust. South African J Bot 140:312–325. https://doi.org/10.1016/j.sajb.2020.06.017
Goedhart J, Luijsterburg MS (2020) VolcaNoseR is a web app for creating, exploring, labeling and sharing volcano plots. Sci Rep 10:20560. https://doi.org/10.1038/s41598-020-76603-3
Griffiths-Jones S, Grocock RJ, van Dongen S, Bateman A, Enright AJ (2006) miRBase: microRNA sequences, targets and gene nomenclature. Nucleic Acids Res 34:D140–D144. https://doi.org/10.1093/nar/gkj112
Gu J, Sun J, Liu N, Sun X, Liu C, Wu L, Liu G, Zeng F, Hou C, Han S, Zhen W, Wang D (2020) A novel cysteine-rich receptor-like kinase gene, TaCRK2, contributes to leaf rust resistance in wheat. Mol Plant Pathol 21:732–746. https://doi.org/10.1111/mpp.12929
Gupta OP, Permar V, Koundal V, Singh UD, Praveen S (2012) MicroRNA regulated defense responses in Triticum aestivum L. during Puccinia graminis f. sp. tritici infection. Mol Biol Rep 39:817–824
Gupta PK (2015) MicroRNAs and target mimics for crop improvement. Curr Sci 1624–1633
Hammond-Kosack KE, Jones JD (1996) Resistance gene-dependent plant defense responses. Plant Cell 8:1773
Hayden MJ, Sharp PJ (2001) Sequence-tagged microsatellite profiling (STMP): a rapid technique for developing SSR markers. Nucleic Acids Res 29:e43–e43. https://doi.org/10.1093/nar/29.8.e43
Hazen SP, Naef F, Quisel T, Gendron JM, Chen H, Ecker JR, Borevitz JO, Kay SA (2009) Exploring the transcriptional landscape of plant circadian rhythms using genome tiling arrays. Genome Biol 10:R17. https://doi.org/10.1186/gb-2009-10-2-r17
Hiebert C, Spielmeyer W, McCartney C, Kassa M, Fetch T, You F, Menzies J, Humphreys G, McCallum B (2015) Stem rust resistance: two approaches. Advances in wheat genetics: from genome to field. Springer, Tokyo, pp 183–191
Hsieh L-C, Lin S-I, Shih AC-C, Chen J-W, Lin W-Y, Tseng C-Y, Li W-H, Chiou T-J (2009) Uncovering small RNA-mediated responses to phosphate deficiency in Arabidopsis by deep sequencing. Plant Physiol 151:2120–2132. https://doi.org/10.1104/pp.109.147280
Hu G, Rijkenberg FHJ (1998) Scanning electron microscopy of early infection structure formation by Puccinia recondita f. sp. tritici on and in susceptible and resistant wheat lines. Mycol Res 102:391–399. https://doi.org/10.1017/S0953756297005054
Jain N, Sinha N, Krishna H, Singh PK, Gautam T, Prasad P, Balyan HS, Gupta PK (2020) A study of miRNAs and lncRNAs during Lr28-mediated resistance against leaf rust in wheat (Triticum aestivum L.). Physiol Mol Plant Pathol 112:101552. https://doi.org/10.1016/j.pmpp.2020.101552
Karlova R, van Haarst JC, Maliepaard C, van de Geest H, Bovy AG, Lammers M, Angenent GC, de Maagd RA (2013) Identification of microRNA targets in tomato fruit development using high-throughput sequencing and degradome analysis. J Exp Bot 64:1863–1878. https://doi.org/10.1093/jxb/ert049
Kaur A, Pati PK, Pati AM, Nagpal AK (2017) In-silico analysis of cis-acting regulatory elements of pathogenesis-related proteins of Arabidopsis thaliana and Oryza sativa. PLoS ONE 12:e0184523
Kolmer JA, Chen X, Jin Y (2009) Diseases which challenge global wheat production-the cereal rusts. Carver BF(ed) Wheat: Science and trade. Wiley Press Hoboken, NJ, USA, pp 89–124
Kumar D, Dutta S, Singh D, Prabhu KV, Kumar M, Mukhopadhyay K (2017a) Uncovering leaf rust responsive miRNAs in wheat (Triticum aestivum L.) using high-throughput sequencing and prediction of their targets through degradome analysis. Planta 245:161–182
Kumar G, Singh A, Vinutha T, Kumar S, Kumar RR, Praveen S (2017b) Possible role of miRNAs and their targets, in modulating leaf morphology and plant growth during leaf curl virus infection in tomato. Indian J Plant Physiol 22:608–615. https://doi.org/10.1007/s40502-017-0345-5
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Langmead B (2010) Aligning short sequencing reads with Bowtie. Curr Protoc Bioinforma 32:11–17
Lescot M, Déhais P, Thijs G, Marchal K, Moreau Y, de Peer Y, Rouzé P, Rombauts S (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res 30:325–327
Li Y, Zhang Q, Zhang J, Wu L, Qi Y, Zhou J-M (2010) Identification of microRNAs involved in pathogen-associated molecular pattern-triggered plant innate immunity. Plant Physiol 152:2222–2231. https://doi.org/10.1104/pp.109.151803
Li W, Wang F, Wang J, Fan F, Zhu J, Yang J, Liu F, Zhong W (2015) Overexpressing CYP71Z2 enhances resistance to bacterial blight by suppressing auxin biosynthesis in rice. PLoS ONE 10:e0119867–e0119867. https://doi.org/10.1371/journal.pone.0119867
Li Y-F, Wei K, Wang M, Wang L, Cui J, Zhang D, Guo J, Zhao M, Zheng Y (2019) Identification and temporal expression analysis of conserved and novel microRNAs in the leaves of winter wheat grown in the field. Front Genet 10:779. https://doi.org/10.3389/fgene.2019.00779
Liu B, Sun G (2017) microRNAs contribute to enhanced salt adaptation of the autopolyploid Hordeum bulbosum compared with its diploid ancestor. Plant J 91:57–69. https://doi.org/10.1111/tpj.13546
Liu B, Li P, Li X, Liu C, Cao S, Chu C, Cao X (2005) Loss of function of OsDCL1 affects microRNA accumulation and causes developmental defects in rice. Plant Physiol 139:296–305. https://doi.org/10.1104/pp.105.063420
Liu P-P, Montgomery TA, Fahlgren N, Kasschau KD, Nonogaki H, Carrington JC (2007) Repression of AUXIN RESPONSE FACTOR10 by microRNA160 is critical for seed germination and post-germination stages. Plant J 52:133–146. https://doi.org/10.1111/j.1365-313X.2007.03218.x
Liu P-P, Bhattacharjee S, Klessig DF, Moffett P (2010) Systemic acquired resistance is induced by R gene-mediated responses independent of cell death. Mol Plant Pathol 11:155–160
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:1–21
Lu H, Zou Y, Feng N (2010) Overexpression of AHL20 negatively regulates defenses in Arabidopsis. J Integr Plant Biol 52:801–808. https://doi.org/10.1111/j.1744-7909.2010.00969.x
Marsalis MA, Goldberg NP (2011) Leaf, stem and stripe rust diseases of wheat. Guide A-415
Martin M (2011) Cutadapt removes adapter sequences from high-throughput sequencing reads. Embnet J 17:10–12
McCouch SR, Chen X, Panaud O, Temnykh S, Xu Y, Cho YG, Huang N, Ishii T, Blair M (1997) Microsatellite marker development, mapping and applications in rice genetics and breeding- Oryza: From Molecule to Plant. In: Sasaki T, Moore G (eds). Springer Netherlands, Dordrecht, pp 89–99
Megraw M, Baev V, Rusinov V, Jensen ST, Kalantidis K, Hatzigeorgiou AG (2006) MicroRNA promoter element discovery in Arabidopsis. RNA 12:1612–1619. https://doi.org/10.1261/rna.130506
Naik BK, Sharma JB, Sivasamy M, Prabhu KV, Tomar RS, Tomar SMS (2015) Molecular mapping and validation of the microsatellite markers linked to the Secale cereale-derived leaf rust resistance gene Lr45 in wheat. Mol Breed 35:1–10
Nair MM, Manickavelu A (2020) MicroRNAs as fine-tuners of gene regulation in plant-microbe interactions. Curr Sci 119:1282–1290
Narang D, Kaur S, Steuernagel B, Ghosh S, Bansal U, Li J, Zhang P, Bhardwaj S, Uauy C, Wulff BBH, Chhuneja P (2020) Discovery and characterisation of a new leaf rust resistance gene introgressed in wheat from wild wheat Aegilops peregrina. Sci Rep 10:1–9
Natarajan B, Kalsi HS, Godbole P, Malankar N, Thiagarayaselvam A, Siddappa S, Thulasiram HV, Chakrabarti SK, Banerjee AK (2018) MiRNA160 is associated with local defense and systemic acquired resistance against Phytophthora infestans infection in potato. J Exp Bot 69:2023–2036. https://doi.org/10.1093/jxb/ery025
Navarro L, Dunoyer P, Jay F, Arnold B, Dharmasiri N, Estelle M, Voinnet O, Jones JDG (2006) A plant miRNA contributes to antibacterial resistance by repressing auxin signaling. Science (80-) 312:436–439. https://doi.org/10.1126/science.1126088
Pérez-Quintero ÁL, Quintero A, Urrego O, Vanegas P, López C (2012) Bioinformatic identification of cassava miRNAs differentially expressed in response to infection by Xanthomonas axonopodis pv. manihotis. BMC Plant Biol 12:1–11
Periyannan S, Milne RJ, Figueroa M, Lagudah ES, Dodds PN (2017) An overview of genetic rust resistance: from broad to specific mechanisms. PLoS Pathog 13:e1006380
Petrov V, Vermeirssen V, De Clercq I, Van Breusegem F, Minkov I, Vandepoele K, Gechev TS (2012) Identification of cis-regulatory elements specific for different types of reactive oxygen species in Arabidopsis thaliana. Gene 499:52–60. https://doi.org/10.1016/j.gene.2012.02.035
Quesada V (2016) The roles of mitochondrial transcription termination factors (MTERFs) in plants. Physiol Plant 157:389–399. https://doi.org/10.1111/ppl.12416
Roberti M, Polosa PL, Bruni F, Manzari C, Deceglie S, Gadaleta MN, Cantatore P (2009) The MTERF family proteins: Mitochondrial transcription regulators and beyond. Biochim Biophys Acta - Bioenerg 1787:303–311. https://doi.org/10.1016/j.bbabio.2009.01.013
Sablok G, Srivastva AK, Suprasanna P, Baev V, Ralph PJ (2015) isomiRs: increasing evidences of isomiRs complexity in plant stress functional biology. Front Plant Sci 6:949. https://doi.org/10.3389/fpls.2015.00949
Sánchez-Retuerta C, Suaréz-López P, Henriques R (2018) Under a new light: regulation of light-dependent pathways by non-coding RNAs. Front Plant Sci 9:962. https://doi.org/10.3389/fpls.2018.00962
Shahmuradov IA, Umarov RK, Solovyev VV (2017) TSSPlant: a new tool for prediction of plant Pol II promoters. Nucleic Acids Res 45:e65–e65
Singh D, Bhaganagare G, Bandopadhyay R, Prabhu KV, Gupta PK, Mukhopadhyay K (2012) Targeted spatio-temporal expression based characterization of state of infection and time-point of maximum defense in wheat NILs during leaf rust infection. Mol Biol Rep 39:9373–9382
Soto-Suárez M, Baldrich P, Weigel D, Rubio-Somoza I, San Segundo B (2017) The Arabidopsis miR396 mediates pathogen-associated molecular pattern-triggered immune responses against fungal pathogens. Sci Rep 7:44898
Szklarczyk D, Franceschini A, Wyder S, Forslund K, Heller D, Huerta-Cepas J, Simonovic M, Roth A, Santos A, Tsafou KP et al (2015) STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res 43:D447–D452
Temnykh S, DeClerck G, Lukashova A, Lipovich L, Cartinhour S, McCouch S (2001) Computational and experimental analysis of microsatellites in rice (Oryza sativa L.): frequency, length variation, transposon associations, and genetic marker potential. Genome Res 11:1441–1452
Tyagi S, Kumar A, Gautam T, Pandey R, Rustgi S, Mir RR (2021) Development and use of miRNA-derived SSR markers for the study of genetic diversity, population structure, and characterization of genotypes for breeding heat tolerant wheat varieties. PLoS ONE 16:1–17. https://doi.org/10.1371/journal.pone.0231063
Untergasser A, Cutcutache I, Koressaar T, Ye J, Faircloth BC, Remm M, Rozen SG (2012) Primer3-new capabilities and interfaces. Nucleic Acids Res 40:e115–e115. https://doi.org/10.1093/nar/gks596
Varkonyi-Gasic E (2017) Stem-Loop qRT-PCR for the Detection of Plant microRNAs. Methods Mol Biol 1456:163–175
Wang J-W, Wang L-J, Mao Y-B, Cai W-J, Xue H-W, Chen X-Y (2005) Control of root cap formation by microRNA-targeted auxin response factors in Arabidopsis. Plant Cell 17:2204–2216. https://doi.org/10.1105/tpc.105.033076
Wang X, McCallum BD, Fetch T, Bakkeren G, Saville BJ (2015) Sr36- and Sr5-mediated resistance response to Puccinia graminis f. sp. tritici is associated with callose deposition in wheat guard cells. Phytopathology® 105:728–737. https://doi.org/10.1094/PHYTO-08-14-0213-R
Wrzaczek M, Brosché M, Salojärvi J, Kangasjärvi S, Idänheimo N, Mersmann S, Robatzek S, Karpiński S, Karpińska B, Kangasjärvi J (2010) Transcriptional regulation of the CRK/DUF26 group of receptor-like protein kinases by ozone and plant hormones in Arabidopsis. BMC Plant Biol 10:95. https://doi.org/10.1186/1471-2229-10-95
Xie Z, Allen E, Fahlgren N, Calamar A, Givan SA, Carrington JC (2005) Expression of Arabidopsis MIRNA Genes. Plant Physiol 138:2145–2154. https://doi.org/10.1104/pp.105.062943
Yang K, Sablok G, Qiao G, Nie Q, Wen X (2017) isomiR2Function: an integrated workflow for identifying microRNA variants in plants. Front Plant Sci 8:322
Yeh Y-H, Chang Y-H, Huang P-Y, Huang J-B, Zimmerli L (2015) Enhanced Arabidopsis pattern-triggered immunity by overexpression of cysteine-rich receptor-like kinases. Front Plant Sci 6:322. https://doi.org/10.3389/fpls.2015.00322
Zhang X, Zhao H, Gao S, Wang W-C, Katiyar-Agarwal S, Huang H-D, Raikhel N, Jin H (2011) Arabidopsis Argonaute 2 regulates innate immunity via miRNA393∗-mediated silencing of a golgi-localized SNARE gene, MEMB12. Mol Cell 42:356–366. https://doi.org/10.1016/j.molcel.2011.04.010
Zhang K, Shi X, Zhao X, Ding D, Tang J, Niu J (2015) Investigation of miR396 and growth-regulating factor regulatory network in maize grain filling. Acta Physiol Plant 37:28. https://doi.org/10.1007/s11738-014-1767-6
Zhang Y, Xia R, Kuang H, Meyers BC (2016) The diversification of plant NBS-LRR defense genes directs the evolution of microRNAs that target them. Mol Biol Evol 33:2692–2705. https://doi.org/10.1093/molbev/msw154
Acknowledgements
The authors would like to acknowledge Dr. Rebekah Nisha, Dr. Shajitha P and other staffs of the ICAR-Indian Agricultural Research Institute, Wellington, for their help in pathogen inoculation and plant material collection. The authors would like to thank the Central University of Kerala for research facilities. The authors also thank the Department of Science and Technology-Innovation in Science Pursuit for Inspired Research (DST-INSPIRE) and Council of Scientific and Industrial Research-University Grants Commission (CSIR-UGC) for providing financial support.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. MMN and SJ are supported by DST-INSPIRE fellowship and CSIR-UGC fellowship respectively.
Author information
Authors and Affiliations
Contributions
MMN: conceptualization, methodology, investigation, software, validation, data curation, formal analysis, visualization, writing—original draft preparation. HKS: software, data curation, formal analysis, visualization. SJ: data curation, validation, visualization, writing—reviewing and editing. KTS: software. CM: resources, methodology, writing—reviewing and editing. MS: resources, writing—reviewing and editing. MA: conceptualization, funding, supervision, writing—reviewing and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nair, M.M., Kumar, S.H.K., Jyothsna, S. et al. Stem and leaf rust–induced miRNAome in bread wheat near-isogenic lines and their comparative analysis. Appl Microbiol Biotechnol 106, 8211–8232 (2022). https://doi.org/10.1007/s00253-022-12268-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-022-12268-4