Abstract
Immunological and molecular advances have modernized diagnostic testing for many diseases. Although interferon gamma-release and polymerase chain reaction assays have been developed to detect Mycobacterium tuberculosis (Mtb) infection, purified protein derivative (PPD)–based tuberculin skin testing (TST) remains the most widely used method. Indeed, the TST is a simple and cost-effective tool that can be easily applied for widespread screening for Mtb infection. However, the lack of specificity has been a limitation of these tests, and, more recently, supply issues have arisen. Building upon the skin tests that historically have been used within TB and leprosy control programs, we discuss recent developments using modern technologies for improving mycobacterial skin testing as well as practical advantages inherent to the technique. Furthermore, we outline how this knowledge could be applied to develop similar tests that could benefit diagnostic strategies for other infections.
Key points
• Skin testing provides a significantly cheaper alternative to most modern technologies.
• Skin tests provide a lab-independent diagnostic strategy that can be widely administered.
• Diseases for which T cell responses are more robust or durable than antibody responses are accessible for skin testing.
Similar content being viewed by others
Introduction
Skin testing is a widely known diagnostic method used in allergy and infectious diseases involving intradermal injection of a small amount of antigen to assess either immediate IgE-mediated allergic or antigen-specific T cell–mediated delayed-type hypersensitivity (DTH), responses. Exemplified by the tuberculin skin test (TST) that was originally created by Robert Koch and developed into the intradermal technique in 1912 by Charles Mantoux, multiple iterations have been used to detect mycobacterial infections and guide disease control efforts.
Tuberculosis
Estimates are that nearly 2 billion people latently infected with Mycobacterium tuberculosis, and 5–10% will progress to TB disease during their lifetime (Getahun et al. 2015; Houben and Dodd 2016; WHO 2018a). Indeed, a now relatively stable number of around 10 million people are sickened with tuberculosis (TB) each year. Among these cases, in 2018, there were an estimated 1.2 million TB-associated deaths among HIV-individuals and a further 0.25 million among HIV-infected people (WHO 2019). This makes TB the leading cause of death from a single infectious agent and propels it into the top 10 causes of death overall. The World Health Organization (WHO) End TB Strategy recognizes that unless M. tuberculosis (Mtb) transmission is disrupted, global TB elimination targets are unlikely to be attained (Lonnroth and Raviglione 2016; Uplekar et al. 2015; WHO 2018b). Accordingly, the US Centers for Disease Control and Prevention (CDC) Division of Tuberculosis Elimination’s strategic plan includes accelerating the decline in TB through targeted testing and treatment of latent TB infection (LTBI) (CDC 2020).
At present, TB diagnosis uses indirect methods such as chest X-rays a well as direct detection methods that include 4-week culture of Mtb from sputum samples. Nucleic acid amplification tests (NAAT; e.g., GeneXpert) have sped up the provision of more sensitive and specific results (Boehme et al. 2011; Nicol et al. 2011), and IFN-γ release assays (IGRA) that assess cytokine production following incubation of unfractionated blood with Mtb antigens have also been developed (Arenas Miras Mdel et al. 2013; Arias Guillen 2011; Rangaka et al. 2012) (Fig. 1). Clinicians have great familiarity with skin testing, which requires no laboratory-based technology, and TST has been the standard TB screening/surveillance tool for decades (Bass Jr 2003; Snider Jr 1982).
TST uses purified protein derivative (PPD; tuberculin) to detect T cell responses against Mtb. PPD was produced by first steaming cultures of Mtb then repeatedly precipitating with ammonium sulfate to purify the proteins while reducing polysaccharide, nucleic acid, and lipid content. In 1944, a large lot of PPD-Standard (PPD-S) was made that comprised approximately 92.9% protein, 5.9% polysaccharide, and 1.2% nucleic acid. Eight years later, the World Health Organization (WHO) adopted the PPD-S as the international standard (Guld et al. 1958; Seibert and Glen 1941). The Food and Drug Administration (FDA) required that all lots of PPD be qualified and show a potency equivalent to PPD-S. A standard preparation derived from one solitary batch, Master Batch Connaught Tuberculin (CT68), was then adopted to eliminate batch-to-batch variation (Landi and Held 1980; Sbarbaro 1978). There are now, however, many varieties of PPD in use: PPD-S in the USA; PPD RT23 is the most widely used PPD product outside of North America; PPD RT23 Mexico is used in Latin America; PPD-s is used in Japan; and PPD-L is used in Russia (Comstock et al. 1964; Kimura et al. 2005; Rangel-Frausto et al. 2001; Starshinova et al. 2018; Yang et al. 2012). PPD-S2, the current US standard that was developed to address the eventual depletion of PPD-S, is used in the commercially available Aplisol® and Tubersol® (Jensen et al. 2005; Villarino et al. 2000). Although results with Aplisol® and Tubersol® were comparable to those of the original PPD-S, shifting the use between Tubersol® and Aplisol® has resulted in aberrations that are unexplained (Gillenwater et al. 2006; Mehta et al. 2009; Villarino et al. 1999).
Both Aplisol and Tubersol have experienced regular shortages (CDC 2013b) and routine health department activities had been threatened or already curtailed in 56% US jurisdictions due to reported shortages of PPD TST antigen solutions (10 Tubersol only, four Aplisol only, and 15 both) (CDC 2013a). In June 2019, the US Centers for Disease Control (CDC) announced an anticipated 3–10-month shortage of Aplisol (2019). This limitation could potentially be overcome through the use of defined, species-specific proteins. Mass spectrometry and molecular analyses identified over one hundred proteins from four different PPD, and revealed that roughly 60% of the total protein content is contributed by heat shock proteins GroEl, GroEs, DnaK, and HspX (Borsuk et al. 2009; Cho et al. 2012). These heat shock proteins are conserved among most mycobacterial species, and it is believed that this broad mixture limits the ability of PPD-based TST to distinguish Mtb infection from either exposure to non-tuberculous mycobacteria or vaccination with BCG (Farhat et al. 2006; Huebner et al. 1993).
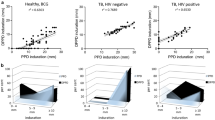
Many TB experts have favored the development of a skin test that could be more accurate for the differential diagnosis of active TB and LTBI (Pai et al. 2016; Rangaka et al. 2012; Rose et al. 1995). We previously demonstrated in preclinical assessments that a novel recombinant protein encoded by the Rv0061 gene that is unique to Mtb had potential as a skin test for Mtb infection. Named DPPD after the first 4 amino acids in the N-terminus sequence, this protein elicited DTH in 100% of M. tuberculosis–infected guinea pigs but not in animals sensitized with other representative Mycobacteria species (M. bovis–BCG, M. avium, M. kansasii, M. fortuitum, M. gordonae, M. chelonae, M. scrofulaceum, M. smegmatis, M. terrae, and M. vaccae) (Coler et al. 2000). Recombinant DPPD produced either in nonpathogenic Mycobacterium smegmatis or in Escherichia coli as a fusion driven from three tandem DPPD gene copies elicited DTH responses in M. tuberculosis–infected guinea pigs that were indistinguishable from those elicited by PPD. (Liu et al. 2004). A proof-of-concept study indicated that intradermal DPPD skin testing detected individuals exposed to Mtb with higher sensitivity and specificity than had previously been reported with PPD (Campos-Neto et al. 2001). In further evaluation of DPPD, irrespective of the TB presentation as cavitary, disseminated, or lymphadenopathic presentation, all HIV-negative active TB patients assessed had indurations > 10 mm following DPPD inoculation. Although there was a highly significant correlation between PPD and DPPD results among TB patients, assessment among participants without active TB but with previous BCG vaccination revealed a specificity of 86.4% DPPD that was much improved over the 42.1% observed for PPD (Badaro et al. 2020).
Leprosy (Hansen’s disease)
Manifesting from Mycobacterium leprae infection, leprosy (Hansen’s disease) is a leading cause of non-traumatic peripheral neuropathy. The extensive provision of curative multiple drug therapy (MDT) over the last 30 years has reduced the prevalence of this complex disease, but a steady detection rate is now being maintained (WHO 2010). Ongoing M. leprae transmission is indicated by case detection rates among children below 15 years of age (Sachdeva et al. 2011a; Singal et al. 2011), and the impact of continued transmission on the true incidence of leprosy is continually identified as a priority by WHO (Sachdeva et al. 2011b). As with TB, epidemiological screening tools that enable early detection of M. leprae–infected individuals are deemed critical for identifying those individuals most in need of prophylactic intervention.
Leprosy presents across a wide array of symptoms that can complicate diagnosis even by experts (Ridley and Jopling 1966; Scollard 2004). Patients are often in clinics with limited facilities and WHO diagnostic criteria has been simplified to allow widespread adoption of either one of two recommended treatment regimen. Leprosy patients are defined into the operational categories, multibacillary (MB) or paucibacillary (PB). At the extreme MB pole, LL patients demonstrate high titers of anti-M. leprae antibodies but an absence of an inflammatory T cell response (Ridley and Jopling 1966). PB patients, encompassing TT and a number of BT forms, are currently characterized as having one or few skin lesions and granulomatous dermatopathology. PB patients have low infectious burdens and low or absent antigen-specific antibody responses, but they demonstrate a specific cell-mediated immunity against M. leprae that provides hope for alternate T cell–based diagnostic strategies, including skin tests.
Skin testing for leprosy has occurred with various antigen preparations generated by crude fractionation of M. leprae (Lepromin A, Rees Antigen, Dharmendra, and Convit’s antigen) over the years. These have proven safe, and lepromin A has been used for nearly half a century (Convit et al. 1992; Dharmendra 2012; Meyers et al. 1975; Millar et al. 1975). Given the inability to culture M. leprae in vitro, however, each of these complex mixtures was derived from M. leprae grown in animals (or extracted from human lesions). These processes neither allow for consistent mass production nor do they meet modern safety standards (Meyers et al. 1975). “Semi-refined” antigen preparations incorporating over 100 components reflective of those contained within Convit and Rees were generated more recently under good manufacturing practices (GMP). Upon clinical evaluation, although Mycobacterium leprae soluble antigens devoid of glycolipids particularly lipoarabinomannan (MLSA-LAM)) and MLCwA (M. leprae cell wall associated antigens) were well-tolerated, the advancement of GMP MLSA-LAM and MLCwA could not be justified as sensitivity was only 20–25% (Marques et al. 2008; Rivoire et al. 2014a, b).
Analogous to the situation with TB, we used in vitro antigen recall data to prioritize the recombinant chimeric LID-1 fusion protein as a skin test candidate antigen for leprosy/M. leprae infection. LID-1 was formulated to achieve maximum performance at a minimal dose in preclinical evaluation of DTH in M. leprae–immune guinea pigs. Select formulations and doses were then evaluated in armadillos, the only M. leprae infection model that recapitulates human disease (Truman et al. 2014). Data indicated that intradermal inoculation of formulated LID-1 could satisfactorily distinguish uninfected from M. leprae–infected animals manifesting with symptoms distinctly similar to the PB presentation of patients, suggesting that evaluation among various groups in leprosy-endemic regions is merited.
Skin tests in immune-compromised subjects
Among groups for whom immunological-based TB testing results are traditionally poor, Mtb infection is especially problematic in immune-compromised individuals that have an increased likelihood of progression to disease such that TB is a leading cause of death in HIV/AIDS patients. The use of TST for the diagnosis of active disease in HIV-infected individuals has been controversial with previous studies reporting reduced sensitivity in this population (Goldstein et al. 1994; Graham et al. 1992; Johnson et al. 1992; Lifson et al. 1993; Mamani et al. 2013; Whittle et al. 1993).
CD4 T cell counts are likely a major determinant of the induration size following skin testing in HIV-positive TB patients. Accordingly, although expanded testing among individuals with defined CD4 T cell counts is required to validate it, we observed that the magnitude of response to DPPD correlated strongly with CD4 T cell counts of HIV-infected patients. Whereas sensitivity of the standard TST test in this population is less than 50%, the DPPD-based test gave 90% sensitivity including even in individuals with very low CD4 T cell counts. These data indicate a potentially significant benefit of skin testing over ex vivo assessment of small blood volumes, likely because antigen-specific T cells that may be limited within the small blood volumes used for IGRA are afforded the time to relocate to, and accumulate at, the antigen inoculation site in a skin test. Thus, defined skin tests such as DPPD have both specificity (i.e., non-reactive to BCG or mycobacteria other than Mtb) and sensitivity (i.e., able to detect Mtb in immune-compromised individuals) advantages over traditional TST.
Logistics and economics of skin testing
The use of lab-based IGRA and NAAT in resource-limited settings has been restricted by their cost and modest availability (Morrison et al. 2008; Pai et al. 2014; Rangaka et al. 2012). Even in the USA, 2013 private insurance claims data relating to TB testing indicated that IGRA were used far less often than TST (13.7% versus 86.3%, respectively) (Owusu-Edusei Jr. et al. 2017). This was despite medical expenditures for TB-specific tests among the employer-based privately insured population costing an estimated $53.0 million. TST was not only the most commonly used test but also provides a clear economic advantage as it was, by far, the least expensive ($9) (Owusu-Edusei Jr. et al. 2017). In addition, skin testing provides a practical advantage as it is well-suited for use in decentralized field studies or surveillance programs where even rudimentary laboratory support is lacking (Fig. 1).
Beyond mycobacteria...
As exemplified by PB leprosy, skin tests lend themselves to detection of pathogens that cause low-level infections that do not elicit strong antibody responses but do elicit inflammatory CD4 T cell responses. Accordingly, the majority of people with asymptomatic, self-resolving Leishmania donovani and L. infantum infections, and cutaneous or mucosal leishmaniasis patients, have positive responses in the leishmanin (Montenegro) skin test commonly produced from cultured promastigotes that are washed and inactivated in 0.5% phenol saline (Magill 2013) or, more recently, with parasite extract (Reed et al. 1986).
Of particular current interest, it is noteworthy that early diagnostic efforts in response to the COVID-19/SARS-CoV-2 pandemic focused on nucleic acid– and antibody-detection tests, but both these platforms have left testing gaps. Antibody-based tests typically fill the void that nucleic acid detection leave (i.e., fleeting positivity and a lack of information relating to history of infection or immune status), but the rapid waning of antibody titers is a natural phenomenon common to coronavirus immunity also been reported after SARS-CoV-2 infection (Alshukairi et al. 2016; Callow et al. 1990; Ibarrondo et al. 2020; Long et al. 2020; Shin et al. 2019). Detailed evaluations are now indicating that antigen-specific T cell responses generated by both symptomatic and asymptomatic SARS-CoV-2 infection are more stable and longer-lasting than humoral responses. Thus, given the inherent benefits documented above, there appears to be significant merits in developing SARS-CoV-2-specific skin tests for integration into COVID-19 epidemiology and containment strategies.
References
Alshukairi AN, Khalid I, Ahmed WA, Dada AM, Bayumi DT, Malic LS, Althawadi S, Ignacio K, Alsalmi HS, Al-Abdely HM, Wali GY, Qushmaq IA, Alraddadi BM, Perlman S (2016) Antibody response and disease severity in healthcare worker MERS survivors. Emerg Infect Dis 22(6). https://doi.org/10.3201/eid2206.160010
Arenas Miras Mdel M, Hidalgo Tenorio C, Jimenez Alonso J (2013) Tuberculosis in patients with systemic lupus erythematosus: Spain’s situation. Reumatol Clin 9(6):369–372. https://doi.org/10.1016/j.reuma.2012.06.011
Arias Guillen M (2011) Advances in the diagnosis of tuberculosis infection. Arch Bronconeumol 47(10):521–530. https://doi.org/10.1016/j.arbres.2011.06.018
Badaro R, Machado BAS, Duthie MS, Araujo-Neto CA, Pedral-Sampaio D, Nakatani M, Reed SG (2020) The single recombinant M. tuberculosis protein DPPD provides enhanced performance of skin testing among HIV-infected tuberculosis patients. AMB Express 10(1):133. https://doi.org/10.1186/s13568-020-01068-6
Bass JB Jr (2003) How good is the tuberculin skin test? Infect Control Hosp Epidemiol 24(11):797–798. https://doi.org/10.1086/502138
Boehme CC, Nicol MP, Nabeta P, Michael JS, Gotuzzo E, Tahirli R, Gler MT, Blakemore R, Worodria W, Gray C, Huang L, Caceres T, Mehdiyev R, Raymond L, Whitelaw A, Sagadevan K, Alexander H, Albert H, Cobelens F, Cox H, Alland D, Perkins MD (2011) Feasibility, diagnostic accuracy, and effectiveness of decentralised use of the Xpert MTB/RIF test for diagnosis of tuberculosis and multidrug resistance: a multicentre implementation study. Lancet 377(9776):1495–1505. https://doi.org/10.1016/S0140-6736(11)60438-8
Borsuk S, Newcombe J, Mendum TA, Dellagostin OA, McFadden J (2009) Identification of proteins from tuberculin purified protein derivative (PPD) by LC-MS/MS. Tuberculosis (Edinb) 89(6):423–430. https://doi.org/10.1016/j.tube.2009.07.003
Callow KA, Parry HF, Sergeant M, Tyrrell DA (1990) The time course of the immune response to experimental coronavirus infection of man. Epidemiol Infect 105(2):435–446. https://doi.org/10.1017/s0950268800048019
Campos-Neto A, Rodrigues-Junior V, Pedral-Sampaio DB, Netto EM, Ovendale PJ, Coler RN, Skeiky YA, Badaro R, Reed SG (2001) Evaluation of DPPD, a single recombinant Mycobacterium tuberculosis protein as an alternative antigen for the Mantoux test. Tuberculosis (Edinb) 81(5-6):353–358
CDC (2013a) Extent and effects of recurrent shortages of purified-protein derivative tuberculin skin test antigen solutions - United States, 2013. MMWR Morb Mortal Wkly Rep 62(49):1014–1015
CDC (2013b) National shortage of purified-protein derivative tuberculin products. MMWR Morb Mortal Wkly Rep 62(16):312
CDC (2020) Division of tuberculosis elimination strategic plan 2016-2020. Accessed 23 Aug 2020
Cho YS, Dobos KM, Prenni J, Yang H, Hess A, Rosenkrands I, Andersen P, Ryoo SW, Bai GH, Brennan MJ, Izzo A, Bielefeldt-Ohmann H, Belisle JT (2012) Deciphering the proteome of the in vivo diagnostic reagent “purified protein derivative” from Mycobacterium tuberculosis. Proteomics 12(7):979–991. https://doi.org/10.1002/pmic.201100544
Coler RN, Skeiky YA, Ovendale PJ, Vedvick TS, Gervassi L, Guderian J, Jen S, Reed SG, Campos-Neto A (2000) Cloning of a Mycobacterium tuberculosis gene encoding a purifed protein derivative protein that elicits strong tuberculosis-specific delayed-type hypersensitivity. J Infect Dis 182(1):224–233. https://doi.org/10.1086/315677
Comstock GW, Edwards LB, Philip RN, Winn WA (1964) A comparison in the United States of America of two tuberculins, Ppd-S and Rt 23. Bull World Health Organ 31:161–170
Convit J, Sampson C, Zuniga M, Smith PG, Plata J, Silva J, Molina J, Pinardi ME, Bloom BR, Salgado A (1992) Immunoprophylactic trial with combined Mycobacterium leprae/BCG vaccine against leprosy: preliminary results. Lancet 339(8791):446–450
Dharmendra (2012) The immunological skin tests in leprosy. Part I. The isolation of a protein antigen of Mycobacterium leprae. 1942. Indian J Med Res 136(3):7p following 502
Farhat M, Greenaway C, Pai M, Menzies D (2006) False-positive tuberculin skin tests: what is the absolute effect of BCG and non-tuberculous mycobacteria? Int J Tuberc Lung Dis 10(11):1192–1204
Getahun H, Matteelli A, Chaisson RE, Raviglione M (2015) Latent Mycobacterium tuberculosis infection. N Engl J Med 372(22):2127–2135. https://doi.org/10.1056/NEJMra1405427
Gillenwater KA, Sapp SC, Pearce K, Siberry GK (2006) Increase in tuberculin skin test converters among health care workers after a change from Tubersol to Aplisol. Am J Infect Control 34(10):651–654. https://doi.org/10.1016/j.ajic.2006.05.288
Goldstein S, Perlman DC, Salomon N (1994) Two-stage tuberculin skin testing in an HIV-infected population: a preliminary report. Mt Sinai J Med 61(4):337–340
Graham NM, Nelson KE, Solomon L, Bonds M, Rizzo RT, Scavotto J, Astemborski J, Vlahov D (1992) Prevalence of tuberculin positivity and skin test anergy in HIV-1-seropositive and -seronegative intravenous drug users. JAMA 267(3):369–373
Guld J, Bentzon MW, Bleiker MA, Griep WA, Magnusson M, Waaler H (1958) Standardization of a new batch of purified tuberculin (PPD) intended for international use. Bull World Health Organ 19(5):845–951
Houben RM, Dodd PJ (2016) The global burden of latent tuberculosis infection: a re-estimation using mathematical modelling. PLoS Med 13(10):e1002152. https://doi.org/10.1371/journal.pmed.1002152
Huebner RE, Schein MF, Bass JB Jr (1993) The tuberculin skin test. Clin Infect Dis 17(6):968–975
Ibarrondo FJ, Fulcher JA, Goodman-Meza D, Elliott J, Hofmann C, Hausner MA, Ferbas KG, Tobin NH, Aldrovandi GM, Yang OO (2020) Rapid decay of anti-SARS-CoV-2 antibodies in persons with mild Covid-19. N Engl J Med 383:1085–1087. https://doi.org/10.1056/NEJMc2025179
Jensen PA, Lambert LA, Iademarco MF, Ridzon R, Cdc (2005) Guidelines for preventing the transmission of Mycobacterium tuberculosis in health-care settings, 2005. MMWR Recomm Rep 54(RR-17):1–141
Johnson MP, Coberly JS, Clermont HC, Chaisson RE, Davis HL, Losikoff P, Ruff AJ, Boulos R, Halsey NA (1992) Tuberculin skin test reactivity among adults infected with human immunodeficiency virus. J Infect Dis 166(1):194–198
Kimura M, Comstock GW, Mori T (2005) Comparison of erythema and induration as results of tuberculin tests. Int J Tuberc Lung Dis 9(8):853–857
Landi S, Held HR (1980) Preparation and characterization of a large batch of tuberculin purified protein derivate (PPD-CT68). Ann Sclavo 22(6):899–907
Lifson AR, Watters JK, Thompson S, Crane CM, Wise F (1993) Discrepancies in tuberculin skin test results with two commercial products in a population of intravenous drug users. J Infect Dis 168(4):1048–1051
Liu C, Flamoe E, Chen HJ, Carter D, Reed SG, Campos-Neto A (2004) Expression and purification of immunologically reactive DPPD, a recombinant Mycobacterium tuberculosis skin test antigen, using Mycobacterium smegmatis and Escherichia coli host cells. Can J Microbiol 50(2):97–105
Long QX, Liu BZ, Deng HJ, Wu GC, Deng K, Chen YK, Liao P, Qiu JF, Lin Y, Cai XF, Wang DQ, Hu Y, Ren JH, Tang N, Xu YY, Yu LH, Mo Z, Gong F, Zhang XL, Tian WG, Hu L, Zhang XX, Xiang JL, Du HX, Liu HW, Lang CH, Luo XH, Wu SB, Cui XP, Zhou Z, Zhu MM, Wang J, Xue CJ, Li XF, Wang L, Li ZJ, Wang K, Niu CC, Yang QJ, Tang XJ, Zhang Y, Liu XM, Li JJ, Zhang DC, Zhang F, Liu P, Yuan J, Li Q, Hu JL, Chen J, Huang AL (2020) Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat Med 26(6):845–848. https://doi.org/10.1038/s41591-020-0897-1
Lonnroth K, Raviglione M (2016) The WHO’s new end TB strategy in the post-2015 era of the sustainable development goals. Trans R Soc Trop Med Hyg 110(3):148–150. https://doi.org/10.1093/trstmh/trv108
Magill A (2013) Hunter’s tropical medicine and emerging infectious disease: 99 - Leishmaniasis, 9th edn. Elsevier
Mamani M, Majzoobi MM, Torabian S, Mihan R, Alizadeh K (2013) Latent and active tuberculosis: evaluation of injecting drug users. Iran Red Crescent Med J 15(9):775–779. https://doi.org/10.5812/ircmj.6283
Marques MA, Neves-Ferreira AG, da Silveira EK, Valente RH, Chapeaurouge A, Perales J, da Silva Bernardes R, Dobos KM, Spencer JS, Brennan PJ, Pessolani MC (2008) Deciphering the proteomic profile of Mycobacterium leprae cell envelope. Proteomics 8(12):2477–2491. https://doi.org/10.1002/pmic.200700971
Mehta SR, MacGruder C, Looney D, Johns S, Smith DM (2009) Differences in tuberculin reactivity as determined in a veterans administration employee health screening program. Clin Vaccine Immunol 16(4):541–543. https://doi.org/10.1128/CVI.00375-08
Meyers WM, Kvernes S, Binford CH (1975) Comparison of reactions to human and armadillo lepromins in leprosy. Int J Lepr Other Mycobact Dis 43(3):218–225
Millar JW, Gannon C, Chan CS (1975) Comparison in leprosy patients of Fernandex and Mitsuda reactions using human and armadillo antigens. A double-blind study. Int J Lepr Other Mycobact Dis 43(3):226–233
Morrison J, Pai M, Hopewell PC (2008) Tuberculosis and latent tuberculosis infection in close contacts of people with pulmonary tuberculosis in low-income and middle-income countries: a systematic review and meta-analysis. Lancet Infect Dis 8(6):359–368. https://doi.org/10.1016/S1473-3099(08)70071-9
Nicol MP, Workman L, Isaacs W, Munro J, Black F, Eley B, Boehme CC, Zemanay W, Zar HJ (2011) Accuracy of the Xpert MTB/RIF test for the diagnosis of pulmonary tuberculosis in children admitted to hospital in Cape Town, South Africa: a descriptive study. Lancet Infect Dis 11(11):819–824. https://doi.org/10.1016/S1473-3099(11)70167-0
Owusu-Edusei K Jr, Winston CA, Marks SM, Langer AJ, Miramontes R (2017) Tuberculosis test usage and medical expenditures from outpatient insurance claims data, 2013. Tuberc Res Treat 2017:3816432–3816439. https://doi.org/10.1155/2017/3816432
Pai M, Denkinger CM, Kik SV, Rangaka MX, Zwerling A, Oxlade O, Metcalfe JZ, Cattamanchi A, Dowdy DW, Dheda K, Banaei N (2014) Gamma interferon release assays for detection of Mycobacterium tuberculosis infection. Clin Microbiol Rev 27(1):3–20. https://doi.org/10.1128/CMR.00034-13
Pai M, Behr MA, Dowdy D, Dheda K, Divangahi M, Boehme CC, Ginsberg A, Swaminathan S, Spigelman M, Getahun H, Menzies D, Raviglione M (2016) Tuberculosis. Nat Rev Dis Primers 2:16076. https://doi.org/10.1038/nrdp.2016.76
Rangaka MX, Wilkinson KA, Glynn JR, Ling D, Menzies D, Mwansa-Kambafwile J, Fielding K, Wilkinson RJ, Pai M (2012) Predictive value of interferon-gamma release assays for incident active tuberculosis: a systematic review and meta-analysis. Lancet Infect Dis 12(1):45–55. https://doi.org/10.1016/S1473-3099(11)70210-9
Rangel-Frausto MS, Ponce-De-Leon-Rosales S, Martinez-Abaroa C, Haslov K (2001) Tuberculosis and tuberculin quality: best intentions, misleading results. Infect Control Hosp Epidemiol 22(8):481–484. https://doi.org/10.1086/501937
Reed SG, Badaro R, Masur H, Carvalho EM, Lorenco R, Lisboa A, Teixeira R, Johnson WD Jr, Jones TC (1986) Selection of a skin test antigen for American visceral leishmaniasis. Am J Trop Med Hyg 35(1):79–85. https://doi.org/10.4269/ajtmh.1986.35.79
Ridley DS, Jopling WH (1966) Classification of leprosy according to immunity. A five-group system. Int J Lepr Other Mycobact Dis 34(3):255–273
Rivoire BL, Groathouse NA, TerLouw S, Neupane KD, Ranjit C, Sapkota BR, Khadge S, Kunwar CB, Macdonald M, Hawksworth R, Thapa MB, Hagge DA, Tibbals M, Smith C, Dube T, She D, Wolff M, Zhou E, Makhene M, Mason R, Sizemore C, Brennan PJ (2014a) Safety and efficacy assessment of two new leprosy skin test antigens: randomized double blind clinical study. PLoS Negl Trop Dis 8(5):e2811. https://doi.org/10.1371/journal.pntd.0002811
Rivoire BL, TerLouw S, Groathouse NA, Brennan PJ (2014b) The challenge of producing skin test antigens with minimal resources suitable for human application against a neglected tropical disease; leprosy. PLoS Negl Trop Dis 8(5):e2791. https://doi.org/10.1371/journal.pntd.0002791
Rose DN, Schechter CB, Adler JJ (1995) Interpretation of the tuberculin skin test. J Gen Intern Med 10(11):635–642
Sachdeva S, Amin SS, Khan Z, Sharma PK, Bansal S (2011a) Childhood leprosy: lest we forget. Trop Dr 41(3):163–165. https://doi.org/10.1258/td.2011.100477
Sachdeva S, Khan Z, Ansari MA, Amin SS (2011b) Leprosy: down but not out. Trop Dr 41(1):28–30. https://doi.org/10.1258/td.2010.100107
Sbarbaro J (1978) Skin test antigens: an evaluation whose time has come. Am Rev Respir Dis 118(1-5)
Scollard DM (2004) Classification of leprosy: a full color spectrum, or black and white? Int J Lepr Other Mycobact Dis 72(2):166–168
Seibert F, Glen J (1941) PPD-S was comprised of approximately 92.1% protein, 5.9% polysaccharides and 1.2% nucleic acid. Am Rev Tuberc 44:9–24
Shin HS, Kim Y, Kim G, Lee JY, Jeong I, Joh JS, Kim H, Chang E, Sim SY, Park JS, Lim DG (2019) Immune responses to Middle East Respiratory Syndrome Coronavirus during the acute and convalescent phases of human infection. Clin Infect Dis 68(6):984–992. https://doi.org/10.1093/cid/ciy595
Singal A, Sonthalia S, Pandhi D (2011) Childhood leprosy in a tertiary-care hospital in Delhi, India: a reappraisal in the post-elimination era. Lepr Rev 82(3):259–269
Snider DE Jr (1982) The tuberculin skin test. Am Rev Respir Dis 125(3 Pt 2):108–118. https://doi.org/10.1164/arrd.1982.125.3P2.108
Starshinova A, Zhuravlev V, Dovgaluk I, Panteleev A, Manina V, Zinchenko U, Istomina E, Pavlova M, Yablonskiy P (2018) A comparison of intradermal test with recombinant tuberculosis allergen (diaskintest) with other immunologic tests in the diagnosis of tuberculosis infection. Int J Mycobacteriol 7:32–39
Truman RW, Ebenezer GJ, Pena MT, Sharma R, Balamayooran G, Gillingwater TH, Scollard DM, McArthur JC, Rambukkana A (2014) The armadillo as a model for peripheral neuropathy in leprosy. ILAR J 54(3):304–314. https://doi.org/10.1093/ilar/ilt050
Uplekar M, Weil D, Lonnroth K, Jaramillo E, Lienhardt C, Dias HM, Falzon D, Floyd K, Gargioni G, Getahun H, Gilpin C, Glaziou P, Grzemska M, Mirzayev F, Nakatani H, Raviglione M, for WsGTBP (2015) WHO’s new end TB strategy. Lancet 385(9979):1799–1801. https://doi.org/10.1016/S0140-6736(15)60570-0
Villarino ME, Burman W, Wang YC, Lundergan L, Catanzaro A, Bock N, Jones C, Nolan C (1999) Comparable specificity of 2 commercial tuberculin reagents in persons at low risk for tuberculous infection. JAMA 281(2):169–171
Villarino ME, Brennan MJ, Nolan CM, Catanzaro A, Lundergan LL, Bock NN, Jones CL, Wang YC, Burman WJ (2000) Comparison testing of current (PPD-S1) and proposed (PPD-S2) reference tuberculin standards. Am J Respir Crit Care Med 161(4 Pt 1):1167–1171. https://doi.org/10.1164/ajrccm.161.4.9906050
Whittle H, Egboga A, Todd J, Morgan G, Rolfe M, Sabally S, Wilkins A, Corrah T (1993) Immunological responses of Gambians in relation to clinical stage of HIV-2 disease. Clin Exp Immunol 93(1):45–50
WHO (2010) Enhanced global strategy for further reducing the disease burden due to leprosy: 2011–2015. World Health Organization Geneva
WHO (2018a) Global Tuberculosis Report 2018. World Health Organization, Geneva
WHO (2018b) Latent tuberculosis infection: updated and consolidated guidelines for programmatic management
WHO (2019) Global tuberculosis report 2019, Geneva
Yang H, Kruh-Garcia NA, Dobos KM (2012) Purified protein derivatives of tuberculin--past, present, and future. FEMS Immunol Med Microbiol 66(3):273–280. https://doi.org/10.1111/j.1574-695X.2012.01002.x
Funding
The authors’ skin test research has been conducted under NIH SBIR contract HHSN272201800019C and an interagency agreement AAI15006 between the US Department of Health and Human Services Health Resources and Services Administration (HRSA) and NIAID, with additional support from the American Leprosy Missions. The funders had no input to the production of this manuscript and the opinions provided are the authors’ own.
Author information
Authors and Affiliations
Contributions
MSD and SGR conceived, wrote, and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any experiments with animals or human participants performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Duthie, M.S., Reed, S.G. Skin tests for the detection of Mycobacterial infections: achievements, current perspectives, and implications for other diseases. Appl Microbiol Biotechnol 105, 503–508 (2021). https://doi.org/10.1007/s00253-020-11062-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-11062-4