Abstract
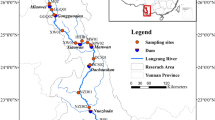
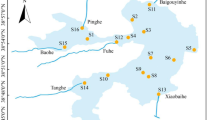
Over the past few decades, anaerobic ammonium oxidation (anammox) has been extensively documented at different scales in natural ecosystems. Previous studies have stated that the community composition of anammox bacteria is shaped mainly by environmental factors, whereas spatial factors have been largely overlooked. This study investigated biogeographical patterns of anammox bacterial communities using 42 sediment samples along a 4300-km stretch of the Yangtze River, the longest river in Asia. A significant distance-decay relationship was observed for anammox bacterial community similarity, which was significantly influenced by mean dendritic distance rather than environmental factors. This implied that dispersal limitation plays an important role in shaping biogeographical pattern of anammox community. Furthermore, our results revealed that neutral processes played vital role in shaping community assembly of anammox bacteria, and their communities were seriously dispersal limited. These findings contrast with previous studies on community similarities between broad taxonomic groups, which are mainly determined by niche-based selection owing to greater niche distances within broad taxonomic groups than in anammox bacteria. Importantly, the slope of the distance-decay curve was much steeper than previously reported for whole bacteria, which indicating the species turnover rate of anammox bacterial community was significantly higher than that of the whole bacterial community. Anammox bacteria harbor strong adhesion ability and low dispersal potential, and ultimately exhibited a high species turnover rate. Together, in the context of biogeography, our results highlight the importance of dispersal limitation in shaping the biogeographical pattern of anammox bacterial community.




Similar content being viewed by others
References
Amato KR, Yeoman CJ, Kent A, Righini N, Carbonero F, Estrada A, Gaskins HR, Stumpf RM, Yildirim S, Torralba M, Gillis M, Wilson BA, Nelson KE, White BA, Leigh SR (2013) Habitat degradation impacts black howler monkey (Alouatta pigra) gastrointestinal microbiomes. ISME J 7(7):1344–1353
Anderson MJ, Crist TO, Chase JM, Vellend M, Inouye BD, Freestone AL, Sanders NJ, Cornell HV, Comita LS, Davies KF (2011) Navigating the multiple meanings of β diversity: a roadmap for the practicing ecologist. Ecol Lett 14(1):19–28
Angermeyer A, Crosby SC, Huber JA (2016) Decoupled distance-decay patterns between dsrA and 16S rRNA genes among salt marsh sulfate-reducing bacteria. Environ Microbiol 18(1):75–86
Astorga A, Oksanen J, Luoto M, Soininen J, Virtanen R, Muotka T (2012) Distance decay of similarity in freshwater communities: do macro- and microorganisms follow the same rules? Glob Ecol Biogeogr 21(3):365–375
Babbin AR, Ward BB (2013) Controls on nitrogen loss processes in Chesapeake Bay sediments. Environ Sci Technol 47(9):4189–4196
Bao S (2000) Chemical analysis for agricultural soil. China Agriculture press, Beijing
Bastian M, Heymann S, Jacomy M (2009) Gephi: an open source software for exploring and manipulating networks. Proc Int AAAI Conf Weblogs Soc Media 8:361–362
Bell T (2010) Experimental tests of the bacterial distance-decay relationship. ISME J 4(11):1357–1365
Borcard D, Legendre P (2002) All-scale spatial analysis of ecological data by means of principal coordinates of neighbour matrices. Ecol Model 153(1–2):51–68
Burns JH, Strauss SY (2011) More closely related species are more ecologically similar in an experimental test. Proc Natl Acad Sci U S A 108(13):5302–5307
Burns AR, Stephens WZ, Stagaman K, Wong S, Rawls JF, Guillemin K, Bohannan BJ (2016) Contribution of neutral processes to the assembly of gut microbial communities in the zebrafish over host development. ISME J 10(3):655–664
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7(5):335–336
Caporaso JG, Lauber CL, Walters WA, Berglyons D, Huntley J, Fierer N, Owens SM, Betley J, Fraser L, Bauer M (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J 6(8):1621–1624
Chase JM, Kraft NJ, Smith KG, Vellend M, Inouye BD (2011) Using null models to disentangle variation in community dissimilarity from variation in α-diversity. Ecosphere 2(2):1–11
Chen L, Liu S, Chen Q, Zhu G, Wu X, Wang J, Li X, Hou L, Ni J (2019) Anammox response to natural and anthropogenic impacts over the Yangtze River. Sci Total Environ 665:171–180
Cirpus IE, Geerts W, Hermans JH, Op den Camp HJ, Strous M, Kuenen JG, Jetten MS (2006) Challenging protein purification from anammox bacteria. Int J Biol Macromol 39(1–3):88–94
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Austral Ecol 18(1):117–143
Dray S, Legendre P, Blanchet F (2007) Packfor: forward selection with permutation, R package version 0.0–7
Flemming HC, Wingender J (2010) The biofilm matrix. Nat Rev Microbiol 8(9):623–633
Gaidos E, Rusch A, Ilardo M (2011) Ribosomal tag pyrosequencing of DNA and RNA from benthic coral reef microbiota: community spatial structure, rare members and nitrogen-cycling guilds. Environ Microbiol 13(5):1138–1152
Gobet A, Böer SI, Huse SM, Van Beusekom JE, Quince C, Sogin ML, Boetius A, Ramette A (2012) Diversity and dynamics of rare and of resident bacterial populations in coastal sands. ISME J 6(3):542–553
Godoy O, Kraft NJ, Levine JM (2014) Phylogenetic relatedness and the determinants of competitive outcomes. Ecol Lett 17(7):836–844
Goslee SC, Urban DL (2007) The ecodist package for dissimilarity-based analysis of ecological data. J Stat Softw 22(7):1–19
Guo B, Zhou Y, Zhu J, Liu W, Wang F, Wang L, Yan F, Wang F, Yang G, Luo W (2016) Spatial patterns of ecosystem vulnerability changes during 2001–2011 in the three-river source region of the Qinghai-Tibetan Plateau, China. J Arid Land 8(1):23–35
Hall BG (2013) Building phylogenetic trees from molecular data with MEGA. Mol Biol Evol 30(5):1229–1235
Hanson CA, Fuhrman JA, Hornerdevine MC, Martiny JBH (2012) Beyond biogeographic patterns: processes shaping the microbial landscape. Nat Rev Microbiol 10(7):497–506
Harhangi HR, Le Roy M, van Alen T, Hu BL, Groen J, Kartal B, Tringe SG, Quan ZX, Jetten MS, Op den Camp HJ (2012) Hydrazine synthase, a unique phylomarker with which to study the presence and biodiversity of anammox bacteria. Appl Environ Microbiol 78(3):752–758
Horner-Devine MC, Lage M, Hughes JB, Bohannan BJ (2004) A taxa-area relationship for bacteria. Nature 432(7018):750–753
Hou X, Liu S, Zhang Z (2015) Role of extracellular polymeric substance in determining the high aggregation ability of anammox sludge. Water Res 75:51–62
Hubbell S (2001) The unified neutral theory of biogeography and biodiversity. Princeton University. Press
Humbert S, Tarnawski S, Fromin N, Mallet M-P, Aragno M, Zopfi J (2010) Molecular detection of anammox bacteria in terrestrial ecosystems: distribution and diversity. ISME J 4(3):450–454
Jain A, Nandakumar K, Ross A (2005) Score normalization in multimodal biometric systems. Pattern Recogn 38(12):2270–2285
Lansdown K, McKew BA, Whitby C, Heppell C, Dumbrell AJ, Binley A, Olde L, Trimmer M (2016) Importance and controls of anaerobic ammonium oxidation influenced by riverbed geology. Nat Geosci 9(5):357–360
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129(2):271–280
Legendre P, Mi X, Ren H, Ma K, Yu M, Sun I-F, He F (2009) Partitioning beta diversity in a subtropical broad-leaved forest of China. Ecology 90(3):663–674
Li H, Yang X, Weng B, Su J, Nie S, Gilbert JA, Zhu Y-G (2016) The phenological stage of rice growth determines anaerobic ammonium oxidation activity in rhizosphere soil. Soil Biol Biochem 100:59–65
Liu T, Zhang AN, Wang J, Liu S, Jiang X, Dang C, Ma T, Liu S, Chen Q, Xie S (2018) Integrated biogeography of planktonic and sedimentary bacterial communities in the Yangtze River. Microbiome 6(1):16
Lotti T, Kleerebezem R, Hu Z, Kartal B, Jetten MS, van Loosdrecht MC (2014) Simultaneous partial nitritation and anammox at low temperature with granular sludge. Water Res 66:111–121
Madsen EL (2011) Microorganisms and their roles in fundamental biogeochemical cycles. Curr Opin Biotechnol 22(3):456–464
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27(2 Part 1):209–220
Mari L, Casagrandi R, Bertuzzo E, Rinaldo A, Gatto M (2014) Metapopulation persistence and species spread in river networks. Ecol Lett 17(4):426–434
Martiny JB, Eisen JA, Penn K, Allison SD, Hornerdevine MC (2011) Drivers of bacterial beta-diversity depend on spatial scale. Proc Natl Acad Sci U S A 108(19):7850–7854
Meyer KM, Memiaghe H, Korte L, Kenfack D, Alonso A, Bohannan BJM (2018) Why do microbes exhibit weak biogeographic patterns? ISME J 12(6):1404–1413
Milliman JD, Meade RH (1983) World-wide delivery of river sediment to the oceans. J Geol 91(1):1–21
Müller AL, De Rezende JR, Hubert CR, Kjeldsen KU, Lagkouvardos I, Berry D, Jørgensen BB, Loy A (2014) Endospores of thermophilic bacteria as tracers of microbial dispersal by ocean currents. ISME J 8(6):1153–1165
Nie S, Li H, Yang X, Zhang Z, Weng B, Huang F, Zhu G, Zhu Y (2015) Nitrogen loss by anaerobic oxidation of ammonium in rice rhizosphere. ISME J 9(9):2059–2067
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’hara R, Simpson GL, Solymos P, Stevens MHH, Wagner H (2013) Package ‘vegan’. Community ecology package, version 2(9)
Oshiki M, Satoh H, Okabe S (2016) Ecology and physiology of anaerobic ammonium oxidizing bacteria. Environ Microbiol 18(9):2784–2796
Pinheiro J, Bates D, DebRoy S, Sarkar D (2014) Linear and nonlinear mixed effects models. R package version 3
Ranjard L, Dequiedt S, Prévost-Bouré NC, Thioulouse J, Saby N, Lelievre M, Maron P, Morin F, Bispo A, Jolivet C (2013) Turnover of soil bacterial diversity driven by wide-scale environmental heterogeneity. Nat Commun 4:1434
Read DS, Gweon HS, Bowes MJ, Newbold LK, Field D, Bailey MJ, Griffiths RI (2015) Catchment-scale biogeography of riverine bacterioplankton. ISME J 9(2):516–526
Robeson MS, King AJ, Freeman KR, Birky CW, Martin AP, Schmidt SK (2011) Soil rotifer communities are extremely diverse globally but spatially autocorrelated locally. Proc Natl Acad Sci U S A 108(11):4406–4410
Roguet A, Laigle GS, Therial C, Bressy A, Soulignac F, Catherine A, Lacroix G, Jardillier L, Bonhomme C, Lerch TZ (2015) Neutral community model explains the bacterial community assembly in freshwater lakes. FEMS Microbiol Ecol 91(11):fiv125
Rossetti C, Pomati F, Calamari D (2001) Microorganisms’ activity and energy fluxes in lake Varese (Italy): a field method. Water Res 35(5):1318–1324
Russel J, Røder HL, Madsen JS, Burmølle M, Sørensen SJ (2017) Antagonism correlates with metabolic similarity in diverse bacteria. Proc Natl Acad Sci U S A 114(40):10684–10688
Savio D, Sinclair L, Ijaz UZ, Parajka J, Reischer GH, Stadler P, Blaschke AP, Bloschl G, Mach RL, Kirschner AK, Farnleitner AH, Eiler A (2015) Bacterial diversity along a 2600 km river continuum. Environ Microbiol 17(12):4994–5007
Shen L, Liu S, Lou L, Liu W, Xu X, Zheng P, Hu B (2013) Broad distribution of diverse anaerobic ammonium-oxidizing bacteria in Chinese agricultural soils. Appl Environ Microbial 79(19):6167–6172
Sloan WT, Lunn M, Woodcock S, Head IM, Nee S, Curtis TP (2006) Quantifying the roles of immigration and chance in shaping prokaryote community structure. Environ Microbiol 8(4):732–740
Sloan WT, Woodcock S, Lunn M, Head IM, Curtis TP (2007) Modeling taxa-abundance distributions in microbial communities using environmental sequence data. Microb Ecol 53(3):443–455
Soininen J, Mcdonald R, Hillebrand H (2007) The distance decay of similarity in ecological communities. Ecography 30(1):3–12
Sonthiphand P, Hall MW, Neufeld JD (2014) Biogeography of anaerobic ammonia-oxidizing (anammox) bacteria. Front Microbiol 5:399
Staley C, Gould TJ, Wang P, Phillips J, Cotner JB, Sadowsky MJ (2015) Species sorting and seasonal dynamics primarily shape bacterial communities in the Upper Mississippi River. Sci Total Environ 505:435–445
Strous M, Van Gerven E, Zheng P, Kuenen JG, Jetten MS (1997) Ammonium removal from concentrated waste streams with the anaerobic ammonium oxidation (anammox) process in different reactor configurations. Water Res 31(8):1955–1962
Strous M, Heijnen J, Kuenen JG, Jetten M (1998) The sequencing batch reactor as a powerful tool for the study of slowly growing anaerobic ammonium-oxidizing microorganisms. Appl Microbiol Biotechnol 50(5):589–596
Strous M, Fuerst JA, Kramer EH, Logemann S, Muyzer G, van de Pas-Schoonen KT, Webb R, Kuenen JG, Jetten MS (1999) Missing lithotroph identified as new planctomycete. Nature 400(6743):446–449
van de Graaf AA, Mulder A, Robertson LA, Kuenen JG (1995) Anaerobic ammonium oxidation discovered in a denitrifying fluidized bed reactor. FEMS Microbiol Ecol 16(3):177–184
Van de Graaf AA, de Bruijn P, Robertson LA, Jetten MS, Kuenen JG (1996) Autotrophic growth of anaerobic ammonium-oxidizing micro-organisms in a fluidized bed reactor. Microbiology 142(8):2187–2196
Wang S, Zhu G, Peng Y, Jetten MS, Yin C (2012) Anammox bacterial abundance, activity, and contribution in riparian sediments of the Pearl River estuary. Environ Sci Technol 46(16):8834–8842
Wang X, Lü X, Yao J, Wang Z, Deng Y, Cheng W, Zhou J, Han X (2017) Habitat-specific patterns and drivers of bacterial β-diversity in China’s drylands. ISME J 11(6):1345–1358
Xue Y, Yu Z, Chen H, Yang JR, Liu M, Liu L, Huang B, Yang J (2017) Cyanobacterial bloom significantly boosts hypolimnelic anammox bacterial abundance in a subtropical stratified reservoir. FEMS Microbiol Ecol 93(10):fix118
Xue Y, Chen H, Yang JR, Liu M, Huang B, Yang J (2018) Distinct patterns and processes of abundant and rare eukaryotic plankton communities following a reservoir cyanobacterial bloom. ISME J 12(9):2263–2277
Yang X, Li H, Nie S, Su J, Weng B, Zhu G, Yao H, Gilbert JA, Zhu Y (2015) Potential contribution of anammox to nitrogen loss from paddy soils in Southern China. Appl Environ Microbiol 81(3):938–947
Zhao S, Zhuang L, Wang C, Li Y, Wang S, Zhu G (2017) High-throughput analysis of anammox bacteria in wetland and dryland soils along the altitudinal gradient in Qinghai-Tibet Plateau. Microbiologyopen 7(2):e00556
Zhou J, Deng Y, Luo F, He Z, Yang Y (2011) Phylogenetic molecular ecological network of soil microbial communities in response to elevated CO2. MBio 2(4):e00122–e00111
Zhu G, Wang S, Wang Y, Wang C, Risgaardpetersen N, Jetten MS, Yin C (2011) Anaerobic ammonia oxidation in a fertilized paddy soil. ISME J 5(12):1905–1912
Zhu G, Wang S, Wang W, Wang Y, Zhou L, Jiang B, Camp HJMOD, RisgaardPetersen N, Schwark L, Peng Y (2013) Hotspots of anaerobic ammonium oxidation at land-freshwater interfaces. Nat Geosci 6(2):103–107
Funding
This study received financial support from the Natural Science Foundations of China (Grant No. 51721006) for financial support. This study also received financial support from the Natural Science Foundation of Qinghai Province (No. 2018-ZJ-909).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics statement
This article does not contain any studies involving human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 760 kb)
Rights and permissions
About this article
Cite this article
Chen, L., Liu, S., Chen, Q. et al. Dispersal limitation drives biogeographical patterns of anammox bacterial communities across the Yangtze River. Appl Microbiol Biotechnol 104, 5535–5546 (2020). https://doi.org/10.1007/s00253-020-10511-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10511-4