Abstract
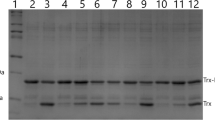
Enterokinase is one of the most frequently used enzymes for the removal of affinity tags from target recombinant proteins. In this study, several fermentation strategies were assayed for the production of human enterokinase in Pichia pastoris under constitutive GAP promoter. Two of them with controlled specific growth rate during whole cultivation showed a very low enterokinase activity, under 1 U/ml, of the fermentation medium. On the contrary, the combined fermentation with a maximum specific growth rate at the initial phase of the fermentation and stationary-like phase during the rest of the fermentation showed a significant accumulation of the enterokinase in the medium, which counted up to 1400 U/ml. Lower cultivation temperature had a negative impact on the enzyme accumulation during this fermentation strategy. Downstream processes were focused on buffer environment optimization directly after cultivation, as at this time, the most amount of the activity is eliminated by endogenous proteases. Slightly positive effect on enzyme activity in the medium had an addition of liquid storage solution of EDTA and KOH to adjust pH to 8 and molarity of the EDTA to 50 mM. During the purification process, a significant amount of the enzyme was detected to be lost, which counted up to 90%. The purified enzyme, enterokinase, kept quality standard of the published enzymes.





Similar content being viewed by others
References
Boer H, Teeri TT, Koivula A (2000) Characterization of Trichoderma reesei cellobiohydrolase Cel7A secreted from Pichia pastoris using two different promoters. Biotechnol Bioeng 69(5):486–494
Bradford MM (1976) Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cereghino JL, Cregg JM (2000) Heterologous protein expression in the methylotrophic yeast Pichia pastoris. FEMS Microbiol Rev 24(1):45–66
Cereghino GP, Cereghino JL, Ilgen C, Cregg JM (2002) Production of recombinant proteins in fermenter cultures of the yeast Pichia pastoris. Curr Opin Biotechnol 13(4):329–332
Chun H, Joo K, Lee J, Shin HC (2011) Design and efficient production of bovine enterokinase light chain with higher specificity in E. coli. Biotechnol Lett 33(6):1227–1232
Cos O, Resina D, Ferrer P, Montesinos JL, Valero F (2005) Heterologous production of Rhizopus oryzae lipase in Pichia pastoris using the alcohol oxidase and formaldehyde dehydrogenase promoters in batch and fed-batch cultures. Biochem Eng J 26(2–3):86–94
Cregg JM, Cereghino JL, Shi J, Higgins DR (2000) Recombinant protein expression in Pichia pastoris. Mol Biotechnol 16(1):23–52
Delroisse J-M, Dannau M, Gilsoul J-J, El Mejdoub T, Destain J, Portetelle D, Thonart P, Haubruge E, Vandenbol M (2005) Expression of a synthetic gene encoding a Tribolium castaneum carboxylesterase in Pichia pastoris. Protein Expr Purif 42(2):286–294
Fang L, Sun QM, Hua ZC (2004) Expression of recombinant Chinese bovine enterokinase catalytic subunit in P. pastoris and its purification and characterization. Acta Biochim Biophys Sin Shanghai 36(7):513–517
Ferrer-Miralles N, Domingo-Espin J, Corchero JL, Vazquez E, Villaverde A (2009) Microbial factories for recombinant pharmaceuticals. Microb Cell Factories 8:17
Garcia-Ortega X, Ferrer P, Montesinos JL, Valero F (2013) Fed-batch operational strategies for recombinant Fab production with Pichia pastoris using the constitutive GAP promoter. Biochem Eng J 79:172–181
Gasparian ME, Ostapchenko VG, Schulga AA, Dolgikh DA, Kirpichnikov MP (2003) Expression, purification, and characterization of human enteropeptidase catalytic subunit in Escherichia coli. Protein Expr Purif 31(1):133–139
Gasparian ME, Ostapchenko VG, Dolgikh DA, Kirpichnikov MP (2006) Biochemical characterization of human enteropeptidase light chain. Biochem Mosc 71(2):113–119
Goodrick JC, Xu M, Finnegan R, Schilling BM, Schiavi S, Hoppe H, Wan NC (2001) High-level expression and stabilization of recombinant human chitinase produced in a continuous constitutive Pichia pastoris expression system. Biotechnol Bioeng 74(6):492–497
Heyland J, Fu J, Blank LM, Schmid A (2010) Quantitative physiology of Pichia pastoris during glucose-limited high-cell density fed-batch cultivation for recombinant protein production. Biotechnol Bioeng 107(2):357–368
Hong F, Meinander NQ, Jonsson LJ (2002) Fermentation strategies for improved heterologous expression of laccase in Pichia pastoris. Biotechnol Bioeng 79(4):438–449
Hosfield T, Lu Q (1999) Influence of the amino acid residue downstream of (Asp)4 Lys on enterokinase cleavage of a fusion protein. Anal Biochem 269(1):10–16
Kim HJ, Kim YH, Roh YH, Seong BL, Shin CS (2005) Optimization of enterokinase fermentation using a recombinant Saccharomyces cerevisiae. Process Biochem 40:717–722
Kobayashi K, Kuwae S, Ohya T, Ohda T, Ohyama M, Ohi H, Tomomitsu K, Ohmura T (2000) High-level expression of recombinant human serum albumin from the methylotrophic yeast Pichia pastoris with minimal protease production and activation. J Biosci Bioeng 89(1):55–61
LaVallie ER, DiBlasio EA, Kovacic S, Grant KL, Schendel PF, McCoy JM (1993) A thioredoxin gene fusion expression system that circumvents inclusion body formation in the E. coli cytoplasm. Biotechnology (N Y) 11(2):187–193
Li Z, Xiong F, Lin Q, d’Anjou M, Daugulis AJ, Yang DS, Hew CL (2001) Low-temperature increases the yield of biologically active herring antifreeze protein in Pichia pastoris. Prot Exp Pur 21(3):438–445
Looser V, Bruhlmann B, Bumbak F, Stenger C, Costa M, Camattari A, Fotiadis D, Kovar K (2015) Cultivation strategies to enhance productivity of Pichia pastoris: a review. Biotechnol Adv 33(6 Pt 2):1177–1193
Lu D (1997) Bovine proenteropeptidase is activated by trypsin, and the specificity of enteropeptidase depends on the heavy chain. J Biol Chem 272(50):31293–31300
Macauley-Patrick S, Fazenda ML, McNeil B, Harvey LM (2005) Heterologou protein production using the Pichia pastoris expression system. Yeast 22(4):249–270
Mattanovich D, Graf A, Stadlmann J, Dragosits M, Redl A, Maurer M, Kleinheinz M, Sauer M, Altmann F, Gasser B (2009) Genome, secretome and glucose transport highlight unique features of the protein production host Pichia pastoris. Microb Cell Factories 8:29
Maurer M, Kuhleitner M, Gasser B, Mattanovich D (2006) Versatile modeling and optimization of fed batch processes for the production of secreted heterologous proteins with Pichia pastoris. Microb Cell Factories 5:37
Mikhailova AG, Likhareva VV, Teich N, Rumsh LD (2007) The ways of realization of high specificity and efficiency of enteropeptidase. Protein Pept Lett 14(3):227–232
Pepeliaev S, Krahulec J, Cerny Z, Jilkova J, Tlusta M, Dostalova J (2011) High level expression of human enteropeptidase light chain in Pichia pastoris. J Biotechnol 156(1):67–75
Potvin G, Ahmad A, Zhang Z (2012) Bioprocess engineering aspects of heterologous protein production in Pichia pastoris: a review. Biochem Eng J 64:91–105
Ragon M, Neugnot-Roux V, Chemardin P, Moulin G, Boze H (2008) Molecular gene cloning and overexpression of the phytase from Debaryomyces castellii CBS 2923. Protein Expr Purif 58(2):275–283
Simeonov P, Berger-Hoffmann R, Hoffmann R, Strater N, Zuchner T (2011) Surface supercharged human enteropeptidase light chain shows improved solubility and refolding yield. Protein Eng Des Sel 24(3):261–268
Sinha J, Plantz BA, Inan MMM (2005) Causes of proteolytic degradation of secreted recombinant proteins produced in methylotrophic yeast Pichia pastoris: case study with recombinant ovine interferon-tau. Biotechnol Bioeng 89(1):102–112
Skala W, Goettig P, Brandstetter H (2013) Do-it-yourself histidine-tagged bovine enterokinase: a handy member of the protein engineer’s toolbox. J Biotechnol 168(4):421–425
Svetina M, Kraševec N, Gaberc-Porekar V, Komel R (2000) Expression of catalytic subunit of bovine enterokinase in the filamentous fungus Aspergilus niger. J Biotechnol 76:245–251
Vogl T, Glieder A (2013) Regulation of Pichia pastoris promoters and its consequences for protein production. New Biotechnol 30(4):385–404
Vozza LA, Wittwer L, Higgins DR, Purcell TJ, Bergseid M, Collins-Racie LA, LaVallie ER, Hoeffler JP (1996) Production of a recombinant bovine enterokinase catalytic subunit in the methylotrophic yeast Pichia pastoris. Bio/Technology 14:77–81
Wu JM, Wang SY, Fu WC (2012) Lower temperature cultures enlarge the effects of Vitreoscilla hemoglobin expression on recombinant Pichia pastoris. Int J Mol Sci 13(10):13212–13226
Young CL, Britton ZT, Robinson AS (2012) Recombinant protein expression and purification: a comprehensive review of affinity tags and microbial applications. Biotechnol J 7(5):620–634
Zhang AL, Zhang TY, Luo JX, Chen SC, Guan WJ, Fu CY, Peng SQ, Li HL (2007) Constitutive expression of human angiostatin in Pichia pastoris by high-density cell culture. J Ind Microbiol Biotechnol 34(2):117–122
Zhang AL, Luo JX, Zhang TY, Pan YW, Tan YH, Fu CY, Tu FZ (2009) Recent advances on the GAP promoter derived expression system of Pichia pastoris. Mol Biol Rep 36(6):1611–1619a
Zhao W, Wang J, Deng R, Wang X (2008) Scale-up fermentation of recombinant Candida rugosa lipase expressed in Pichia pastoris using the GAP promoter. J Ind Microbiol Biotechnol 35(3):189–195
Zheng XL, Kitamoto Y, Sadler JE (2009) Enteropeptidase, a type II transmembrane serine protease. Front Biosci (Elite Ed) 1:242–249
Acknowledgements
This work was supported by the Slovak Research and Development Agency under the contract No. APVV-0119-12.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by the Slovak Research and Development Agency under the contract No. APVV-0119-12.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Melicherová, K., Krahulec, J., Šafránek, M. et al. Optimization of the fermentation and downstream processes for human enterokinase production in Pichia pastoris . Appl Microbiol Biotechnol 101, 1927–1934 (2017). https://doi.org/10.1007/s00253-016-7960-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-7960-3