Abstract
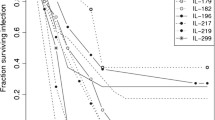
To evaluate the effect of genetic background on antibacterial defense to streptococcal infection, eight genetically diverse strains of mice (A/J, DBA/2J, CAST/Ei, FVB/NJ, BALB/cJ, C57BL/6J, 129/SvImJ, and C3H/HeJ) and tlr2-deficient mice (C57BL/6tlr2−/−) were infected with three doses of Streptococcus zooepidemicus (500, 5,000, or 50,000 colony-forming units) by alveolar challenge. There was a range of susceptibility between the strains at each dose and time point (6, 24, and 96 h). At the lowest dose, the 129/SvImJ and C3H/HeJ strains had significantly higher bacterial counts at all time points after infection, when compared to A/J, DBA/2J, CAST/Ei, FVB/NJ, which were resistant to infection at the low dose of innoculum. At the medium dose, 129/SvImJ and C3H/HeJ had higher bacterial counts, while A/J, DBA/2J, and BALB/cJ showed reduced streptococcal growth. After the highest dose of Streptococcus, there were minimal differences between strains, suggesting the protective impact of modifier genes can be overcome. TLR2-deficient animals contained increased bacterial load with reduced cytokines after 96 h when compared to C57BL/6J controls suggesting a role of innate immunity in late antibacterial defense. Overall, we identify vulnerable (129/SvlmJ and C3H/HeJ) and resistant (A/J, FVB, and DBA) mouse strains to streptococcal lung infection, which demonstrate divergent genetic expression profiles. These results demonstrate that innate differences in pulmonary host defense to S. zooepidemicus are dependent on host genetic factors.







Similar content being viewed by others
References
Branger J, Knapp S, Weijer S, Leemans JC, Pater JM, Speelman P, Florquin S, van der Poll T (2004) Role of Toll-like receptor 4 in gram-positive and gram-negative pneumonia in mice. Infect Immun 72:788–794
Bylund M, Andersson E, Novitch BG, Muhr J (2003) Vertebrate neurogenesis is counteracted by Sox1–3 activity. Nat Neurosci 6:1162–1168
Chanter N, Ward CL, Talbot NC, Flanagan JA, Binns M, Houghton SB, Smith KC, Mumford JA (1999) Recombinant hyaluronate associated protein as a protective immunogen against Streptococcus equi and Streptococcus zooepidemicus challenge in mice. Microb Pathog 27:133–143
Dallaire F, Ouellet N, Bergeron Y, Turmel V, Gauthier MC, Simard M, Bergeron MG (2001) Microbiological and inflammatory factors associated with the development of pneumococcal pneumonia. J Infect Dis 184:292–300
Denny P, Hopes E, Gingles N, Broman KW, McPheat W, Morten J, Alexander J, Andrew PW, Brown SD (2003) A major locus conferring susceptibility to infection by Streptococcus pneumoniae in mice. Mamm Genome 14:448–453
Echchannaoui H, Frei K, Schnell C, Leib SL, Zimmerli W, Landmann R (2002) Toll-like receptor 2-deficient mice are highly susceptible to Streptococcus pneumoniae meningitis because of reduced bacterial clearing and enhanced inflammation. J Infect Dis 186:798–806
Gillespie SH, Balakrishnan I (2000) Pathogenesis of pneumococcal infection. J Med Microbiol 49:1057–1067
Gingles NA, Alexander JE, Kadioglu A, Andrew PW, Kerr A, Mitchell TJ, Hopes E, Denny P, Brown S, Jones HB, Little S, Booth GC, McPheat WL (2001) Role of genetic resistance in invasive pneumococcal infection: identification and study of susceptibility and resistance in inbred mouse strains. Infect Immun 69:426–434
Goldmann O, Chhatwal GS, Medina E (2003) Immune mechanisms underlying host susceptibility to infection with group A streptococci. J Infect Dis 187:854–861
Hegde P, Qi R, Abernathy K, Gay C, Dharap S, Gaspard R, Hughes JE, Snesrud E, Lee N, Quackenbush J (2000) A concise guide to cDNA microarray analysis. Biotechniques 29:548–550 552–4, 556 passim
Hosack DA, Dennis G Jr, Sherman BT, Lane HC, Lempicki RA (2003) Identifying biological themes within lists of genes with EASE. Genome Biol 4:R70
Hoshino K, Takeuchi O, Kawai T, Sanjo H, Ogawa T, Takeda Y, Takeda K, Akira S (1999) Cutting edge: toll-like receptor 4 (TLR4)-deficient mice are hyporesponsive to lipopolysaccharide: evidence for TLR4 as the Lps gene product. J Immunol 162:3749–3752
Kerr AR, Irvine JJ, Search JJ, Gingles NA, Kadioglu A, Andrew PW, McPheat WL, Booth CG, Mitchell TJ (2002) Role of inflammatory mediators in resistance and susceptibility to pneumococcal infection. Infect Immun 70:1547–1557
Khan AQ, Chen Q, Wu ZQ, Paton JC, Snapper CM (2005) Both innate immunity and type 1 humoral immunity to Streptococcus pneumoniae are mediated by MyD88 but differ in their relative levels of dependence on toll-like receptor 2. Infect Immun 73:298–307
Knapp S, Wieland CW, van ’t Veer C, Takeuchi O, Akira S, Florquin S, van der Poll T (2004) Toll-like receptor 2 plays a role in the early inflammatory response to murine pneumococcal pneumonia but does not contribute to antibacterial defense. J Immunol 172:3132–3138
Koedel U, Angele B, Rupprecht T, Wagner H, Roggenkamp A, Pfister HW, Kirschning CJ (2003) Toll-like receptor 2 participates in mediation of immune response in experimental pneumococcal meningitis. J Immunol 170:438–4344
Larkin JE, Frank BC, Gaspard RM, Duka I, Gavras H, Quackenbush J (2004) Cardiac transcriptional response to acute and chronic angiotensin II treatments. Physiol Genomics 18:152–166
Lauw FN, Branger J, Florquin S, Speelman P, van Deventer SJ, Akira S, van der Poll T (2002) IL-18 improves the early antimicrobial host response to pneumococcal pneumonia. J Immunol 168:372–378
Malley R, Henneke P, Morse SC, Cieslewicz MJ, Lipsitch M, Thompson CM, Kurt-Jones E, Paton JC, Wessels MR, Golenbock DT (2003) Recognition of pneumolysin by Toll-like receptor 4 confers resistance to pneumococcal infection. Proc Natl Acad Sci USA 100:1966–1971
Morbidity and Mortality Weekly Report (1983) Group C Streptococcal Infections Associated with Eating Homemade Cheese - New Mexico. October 07, 1983 / 32(39); 510, 515–516. Reported by FH Espinosa, MD, WM Ryan, MD, PL Vigil, MD, DF Gregory, MD, RB Hilley, MD, DA Romig, MD, RB Stamm, MD, ED Suhre, MD, PS Taulbee, MD, LH Zucal, MD, RW Honsinger Jr. MD, PJ Lindberg, MD, M Barcheck, JA Miller, R Mitzelfelt, JM Montes, LJ Nims, OJ Rollag, DVM, N Weber, JM Mann, MD
Morel L (2004) Mouse models of human autoimmune diseases: essential tools that require the proper controls. PLoS Biol 2(8):E241
Poltorak A, He X, Smirnova I, Liu M-Y, Van Huffel C, Du X, Birdwell D, Alejos E, Silva M, Galanos C, Freudenberg M, Ricciardi-Castagnoli P, Layton B, Beutler B (1998) Defective LPS signaling in C3H/Hej and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282:2085–2088
Rijneveld AW, Levi M, Florquin S, Speelman P, Carmeliet P, van Der Poll T (2002) Urokinase receptor is necessary for adequate host defense against pneumococcal pneumonia. J Immunol 168:3507–3511
Rose HD, Allen JR, Witte G (1980) Streptococcus zooepidemicus. (group C) pneumonia in a human. J Clin Microbiol 11:76–78
Saeed AI, Sharov V, White J, Li J, Liang W, Bhagabati N, Braisted J, Klapa M, Currier T, Thiagarajan M, Sturn A, Snuffin M, Rezantsev A, Popov D, Ryltsov A, Kostukovich E, Borisovsky I, Liu Z, Vinsavich A, Trush V, Quackenbush J (2003) TM4: a free, open-source system for microarray data management and analysis. Biotechniques 34:374–378
Sun H, Ringdahl U, Homeister JW, Fay WP, Engleberg NC, Yang AY, Rozek LS, Wang X, Sjobring U, Ginsburg D (2004) Plasminogen is a critical host pathogenicity factor for group A streptococcal infection. Science 305:1283–1286
Takashima K, Tateda K, Matsumoto T, Iizawa Y, Nakao M, Yamaguchi K (1997) Role of tumor necrosis factor alpha in pathogenesis of pneumococcal pneumonia in mice. Infect Immun 65:257–260
Takeuchi O, Hoshino K, Kawai T, Sanjo H, Takada H, Ogawa T, Takeda K, Akira S (1999) Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity 11:443–451
Thoma-Uszynski S, Stenger S, Takeuchi O, Ochoa MT, Engele M, Sieling PA, Barnes PF, Rollinghoff M, Bolcskei PL, Wagner M, Akira S, Norgard MV, Belisle JT, Godowski PJ, Bloom BR, Modlin RL (2001) Induction of direct antimicrobial activity through mammalian toll-like receptors. Science 291:1544–1547
van der Poll T, Marchant A, Keogh CV, Goldman M, Lowry SF (1996) Interleukin-10 impairs host defense in murine pneumococcal pneumonia. J Infect Dis 174:994–1000
van der Poll T, Keogh CV, Guirao X, Buurman WA, Kopf M, Lowry SF (1997) Interleukin-6 gene-deficient mice show impaired defense against pneumococcal pneumonia. J Infect Dis 176:439–444
van der Sluijs KF, van Elden LJ, Nijhuis M, Schuurman R, Pater JM, Florquin S, Goldman M, Jansen HM, Lutter R, van der Poll T (2004) IL-10 is an important mediator of the enhanced susceptibility to pneumococcal pneumonia after influenza infection. J Immunol 172:7603–7609
van Rossum AM, Lysenko ES, Weiser JN (2005) Host and bacterial factors contributing to the clearance of colonization by Streptococcus pneumoniae in a murine model. Infect Immun 73:7718–7726
Wong PM, Chugn SW, Sultzer BM (2000) Genes, receptors, signals and responses to lipopolysaccharide endotoxin. Scand J Immunol 51:123–127
Yoshimura A, Lien E, Ingalls RR, Tuomanen E, Dziarski R, Golenbock D (1999) Cutting edge: recognition of Gram-positive bacterial cell wall components by the innate immune system occurs via Toll-like receptor 2. J Immunol 163:1–5
Acknowledgments
This manuscript was supported by grants from the National Institute of Environmental Health Sciences (ES12717, ES11375, ES12496, and ES11961), the National Institute of Allergy and Infectious Diseases (AI58161), the National Heart, Lung, and Blood Institute (HL67467, HL91335), and the Department of Veterans’ Affairs (Merit Review). The research described in this article has been reviewed by the National Health and Environmental Effects Research Laboratory, US Environmental Protection Agency, and approved for publication. Approval does not signify that the contents necessarily reflect the views and policies of the agency.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hollingsworth, J.W., Whitehead, G., Berman, K.G. et al. Genetic basis of murine antibacterial defense to streptococcal lung infection. Immunogenetics 59, 713–724 (2007). https://doi.org/10.1007/s00251-007-0242-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-007-0242-6