Abstract
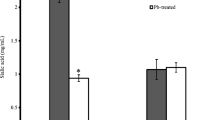
Lead (Pb2+) is a heavy metal that has long been used by humans for a wide range of technological purposes, which is the main reason for its current widespread distribution. Pb2+ is thought to enter erythrocytes through anion exchange and to remain in the cell by binding to thiol groups. Pyruvate kinase (PK) is a thiol-containing enzyme that plays a key role in erythrocyte cellular energy homeostasis. δ-aminolevulinic acid dehydratase (δ-ALAD) is the second enzyme in the heme biosynthetic pathway and plays a role in the pathogenesis of Pb poisoning. Our primary objective was to investigate the effect of Pb2+ on the activity of the thiolenzymes δ-ALAD and PK and on the concentration of glutathione (GSH), a nonenzymatic antioxidant defense, in erythrocytes from Pb-exposed workers. The study sample comprised 22 male Pb workers and 21 normal volunteers (15 men and 6 women). The Pb-exposed workers were employed in manufacturing and recycling of automotive batteries. Basic red-cell parameters were assayed and total white blood cell counts performed. PK and δ-ALAD activity and blood Pb (BPb) concentrations were determined in all subjects. Pb-exposed individuals had significantly greater BPb levels than controls. Both PK and δ-ALAD activity levels were significantly lower in Pb-exposed individuals than in controls. Pb significantly inhibited PK and δ-ALAD activity in a dose-dependent manner. We found that erythrocyte GSH levels were lower in Pb-exposed individuals than normal volunteers. Pb-exposed individuals had lower values than controls for several red cell parameters (hemoglobin, hematocrit, red blood cell count, mean corpuscular volume). These results suggest that Pb inhibits δ-ALAD and PK activity by interacting with their thiol groups. It is therefore possible that Pb disrupts energy homeostasis and may be linked with decreased glucose metabolism because it affects the heme synthesis pathway in erythrocytes, contributing to the cell dysfunction observed in these in Pb-exposed individuals. These results indicate an apparent dose-effect relationship between PK activity and BPb. PK activity in human erythrocytes can be used for biological monitoring of Pb exposure. Study of the mechanisms by which Pb acts may contribute to greater understanding of the symptoms caused by Pb.






Similar content being viewed by others
References
Agency for Toxic Substances and Disease Registry: Toxicological Profile for Lead (2007) United States Department of Heath and Human Services, Public Health Service, ATSDR
Armstrong JS, Jones DP (2002) Glutathione depletion enforces the mitochondrial permeability transition and causes cell death in Bcl-2 overexpressing HL60 cells. FASEB J 16(10):1263–1265
Berlin A, Schaller KH (1974) European standardized method for the determination of delta-aminolevulinic acid dehydratase activity in blood. Z Klin Chem Klin Biochem 12:389–390
Bernard A, Lauwerys R (1987) Metal-induced alterations of delta-aminolevulinic acid dehydratase. Ann N Y Acad Sci 514:41–47
Berry E, Toms NJ (2006) Pyruvate and oxaloacetate limit zinc-induced oxidative HT-22 neuronal cell injury. Neurotoxicology 27(6):1043–1051
Bianchi P, Zanella A (2000) Hematologically important mutations: red cell pyruvate kinase (third update). Blood Cells Mol Dis 26:47–53
Brito JA, McNeill FE, Webber CE, Wells S, Richard N, Carvalho ML et al (2002) Evaluation of a novel structural model to describe the endogenous release of lead from bone. J Environ Monit 4:194–201
Browne RW, Armstrong D (1998) Reduced glutathione and glutathione disulfide. Methods Mol Biol 108:347–352
Calderón-Salinas JV, Hernández-Luna C, Valdez-Anaya B, Maldonado-Vega M, López-Miranda A (1996a) Evolution of lead toxicity in a population of children. Hum Exp Toxicol 15:376–382
Calderón-Salinas JV, Valdez-Anaya B, Mazúñiga-Charles Albores-Medina A (1996b) Lead exposure in a population of Mexican children. Hum Exp Toxicol 15(4):305–311
Calderón-Salinas JV, Quintanar-Escorza MA, Hernández-Luna CE, González- Martínez MT (1999a) Effect of lead on the calcium transport in human erythrocyte. Hum Exp Toxicol 18(3):146–153
Calderón-Salinas JV, Quintanar-Escorza MA, González-Martínez MT, Hernández-Luna CE (1999b) Lead and calcium transport in human erythrocyte. Hum Exp Toxicol 18(5):327–332
Courtois E, Marques M, Barrientos A (2003) Lead-induced down-regulation of soluble guanylate cyclase in isolated rat aortic segments mediated by reactive oxygen species and cyclo-oxygenase-2. J Am Soc Nephrol 14:1464–1470
Das UN (2006) Pyruvate is an endogenous anti-inflammatory and anti-oxidant molecule. Int Med J Exp Clin Res 12:79–84
Dusinska M, Collins AR (2008) The comet assay in human biomonitoring: gene-environment interactions. Mutagenesis 23:191–205
Ekong EB, Jaar BG, Weaver VM (2006) Lead-related nephrotoxicity: a review of epidemiologic evidence. Kidney Int 70:2074–2084
Ercal N, Gurer-Orhan H, Aykin-Burns N (2011) Toxic metals and oxidative stress. Part 1. Mechanisms involved in metal-induced oxidative damage. Curr Top Med Chem 1:529–539
Farant JP, Wigfield DC (1982) Biomonitoring lead exposure with delta-aminolevulinate dehydratase (ALA-D) activity ratios. Int Arch Occup Environ Health 51:15–24
Garza A, Vega R, Soto E (2006) Cellular mechanisms of lead neurotoxicity. Med Sci Monit 12(3):57–65
Gilbert HF (1984) Redox control of enzyme activities by thiol/disulfide exchange. Methods Enzymol 107:330–351
Goering PL (1993) Lead–protein interactions as a basis for lead toxicity. Neurotoxicology 14(2–3):45–60
Greenwood NN, Earnshaw A (1997) Chemistry of the elements, 2nd edn. Butterworth-Heinemann, London
Gurer H, Ercal N (2000) Can antioxidants be beneficial in the treatment of lead poisoning? Free Radic Biol Med 29:927–945
Gurer H, Neal R, Yang P, Oztezcan S, Ercal N (1999) Captropril as an antioxidant in lead-exposed Fischer 344 rats. Hum Exp Toxicol 18:27–32
Gurer-Ohrnan H, Sabir HU, Ozgünes H (2004) Correlation between clinical indicators of lead poisoning and oxidative stress parameters in controls and lead-exposed workers. Toxicology 195(2–3):147–154
Hall ER, Cottam GL (1978) Isoenzymes of pyruvate kinase in vertebrates: their physical, chemical, kinetic and immunological properties. Int J Biochem 9:785–793
Huang M, Krepkiy D, Hu W, Petering DH (2004) Zn-, Cd-, and Pb-transcription factor IIIA: properties, DNA binding, and comparison with TFIIIA finger 3 metal complexes. J Inorg Biochem 98:775–785
Korsrud GO, Meldrum JB (1988) Effect of diet on response in rats to lead acetate given orally or in the drinking water. Biol Trace Elem Res 17:167–173
Lauwerys R, Buchet JP, Roels HA, Materne DO (1974) Relationship between urinary delta-aminolevulinic acid excretion and the inhibition or read cell delta-aminolevulinate dehydratase by lead. Clin Toxicol 7:383–388
Lawton LJ, Donaldson WE (1991) Lead-induced tissue fatty acid alterations and lipid peroxidation. Biol Trace Elem Res 28:83–97
Leech NL, Barrett KC, Morgan GA (2005) SPSS for intermediate statistics. Use and interpretation, 2nd edn. Lawrence Erlbaum Associates, London
Leong SF, Lai JCK, Lim L, Clark JB (1981) Energy-metabolising enzymes in brain regions of adult and aging rats. J Neurochem 37:1548–1556
Lepper TW, Evandro Oliveira E, Koch GDW, Berlese DB, Feksa LR (2010) Lead inhibits in vitro creatine kinase and pyruvate kinase activity in brain cortex of rats. Toxicol in Vitro 24:1045–1051
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Meister A (1994) Glutathione, ascorbate, and cellular protection. Cancer Res 54:1969–1975
Menke A, Muntner P, Batuman P, Silbergeld EK, Guallar E (2006) Blood lead below 0.48 μmol/L (100 μg L−1) and mortality among US adults. Circulation 114:1388–1394
Meredith PA, Moore MR, Campbell BC, Thompson GG, Goldberg A (1978) Delta-aminolevulinic acid metabolism in normal and lead-exposed humans. Toxicology 9:1–9
Muntener P, He J, Vupputuri S, Coresh J, Batuman V (2003) Blood lead and chronic kidney disease in the general United States population: results from NHANES III. Kidney Int 63:1044–1050
Nash D, Magder L, Lustberg M, Sherwin RW, Rubin RJ, Kaufmann RB, Silbergeld EK (2003) Blood lead, blood pressure, and hypertension in perimenopausal and postmenopausal women. JAMA 289(12):1523–1532
Navarro-Moreno LG, Quintanar-Escorza MA, González S, Mondragón R, Cerbón-Solorzáno J, Valdés J et al (2009) Effects of lead intoxication on intercellular junctions and biochemical alterations of the renal proximal tubule cells. Toxicol In Vitro 23:1298–1304
Noguchi T, Yamada K, Inoue H, Matsuda T, Tanaka T (1987) The L- and R-type isozymes of rat pyruvate kinase are produced from a single gene by use of different promoters. J Biol Chem 262:14366–14371
Nunes-Tavares N, Valverde RHF, Araújo GMN, Hassón-Voloch A (2005) Toxicity induced by Hg2+ on choline acetyltransferase activity from E. electricus (L.) electrocytes: the protective effect of 2,3 dimercapto-propanol (BAL). Med Sci Monit 11(4):100–105
Occupational Safety and Health Administration (2008) Employee standard summary—1910.1025 App B [displayed 6 January 2008]. Available at: http://www.osha.gov/Publications/OSHA3348-metal-scrap-recycling.pdf
Olewińska E, Kasperczyk A, Kapka L, Kozłowska A, Pawlas N, Dobrakowski M et al (2010) Level of DNA damage in lead-exposed workers. Ann Agric Environ Med 17:231–236
Onalaja AO, Claudio L (2000) Genetic susceptibility to lead poisoning. Environ Health Perspect 108:23–28
Quintanar-Escorza MA, González-Martínez MT, Navarro L, Maldonado M, Arévalo B, Calderón-Salinas JV (2007) Intracellular free calcium concentration and calcium transport in human erythrocyte of lead-exposed workers. Toxicol Appl Pharmacol 220:1–8
Reed DJ (1990) Glutathione: toxicological implications. Annu Rev Pharmacol Toxicol 30:603–631
Rendón-Ramírez A, Cerbón-Solórzano J, Maldonado-Vega M, Quintanar-Escorza MA, Calderón-Salinas JV (2007) Vitamin-E reduces the oxidative damage on delta-aminolevulinic dehydratase induced by lead intoxication in rat erythrocytes. Toxicol in Vitro 21(6):1121–1126
Ribarov SR, Benov LC (1981) Relationship between the hemolytic action of heavy metals and lipid peroxidation. Biochim Biophys Acta 640:721–726
Sakai T (2000) Biomarkers of lead exposure. Ind Health 38:127–142
Selander S, Cramer K (1970) Interrelationship between lead in blood, lead in urine and ALA in urine during lead worse. Br J Ind Med 27:28–39
Sithisarankul P, Schwartz BS, Lee BK, Strickland PT (1998) Urinary 5-aminolevuinic acid (ALA) adjusted by creatinine a surrogate for plasma ALA. J Occup Environ Med 40:901–908
Todd AC, Wetmur JG, Moline JM, Godbold JH, Levin SM, Landrigan PJ (1996) Unraveling the chronic toxicity of lead: an essential priority for environmental health. Environ Health Perspect 104:141–146
Valle BL, Ulmer DD (1972) Biochemical effects of mercury, cadmium and lead. Annu Rev Biochem 41:91–128
van Bemmel DM, Boffetta P, Liao LM, Berndt SI, Menashe I, Yeager M et al (2011) Comprehensive analysis of 5-aminolevulinic acid dehydrogenase (ALAD) variants and renal cell carcinoma risk among individuals exposed to lead. PLoS One 6(7):1–7
Warren MJ, Cooper JB, Wood SP, Shoolingin JPM (1998) Lead poisoning, haem synthesis and 5-aminolaevulinic acid dehydratase. Trends Biochem Sci 23:217–221
World Health Organization (1995) International Programme on Chemical Safety Environmental Health criteria 165: Inorganic lead. WHO, Geneva
Wu G, Fang YZ, Yang S, Lupton JR, Turner ND (2004) Glutathione metabolism and its implications for health. J Nutr 134:489–492
Acknowledgments
This research was partly supported by grants from Feevale University, CNPq, FAPERGS, and PROPESQ/UFRGS, Brazil.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Feksa, L.R., Oliveira, E., Trombini, T. et al. Pyruvate Kinase Activity and δ-Aminolevulinic Acid Dehydratase Activity as Biomarkers of Toxicity in Workers Exposed to Lead. Arch Environ Contam Toxicol 63, 453–460 (2012). https://doi.org/10.1007/s00244-012-9786-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-012-9786-z