Abstract
Introduction
Diffusion-weighted imaging (DWI) studies focusing on apparent diffusion coefficient (ADC) abnormalities have provided conflicting results about the nature and fate of perihematomal edema.
Methods
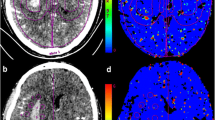
We investigated 35 patients with supratentorial spontaneous intracerebral hemorrhage (SICH) by using DWI scanning obtained at 48 h and 7 days after symptom onset. Regional ADC (rADC) values were measured in three manually outlined regions of interest: (1) the perihematomal hyperintense area, (2) 1 cm of normal appearing brain tissue surrounding the perilesional hyperintense rim, and (3) a mirror area, including the clot and the perihematomal region, located in the contralateral hemisphere.
Results
rADC mean levels were lower at 7 days than at 48 h in each ROI (p < 0.00001), showing a progressive normalization of initial vasogenic values. Perihematomal vasogenic rADC values were more frequent (p < 0.00001) at 48 h than at 7 days, whereas perihematomal cytotoxic and normal rADC levels were more represented (p < 0.02 and p < 0.001, respectively) at 7 days than at 48 h. A neurological worsening was more frequent (p < 0.02) in patients with than in those without perihematomal cytotoxic rADC values at 7 days.
Conclusion
Our findings suggest that the transition from acute to subacute phases after SICH is characterized by a progressive resolution of perihematomal vasogenic edema associated with an increase in cytotoxic ADC values. In the subset of patients with perihematomal cytotoxic rADC levels in subacute stage after bleeding, irreversible damage development seems to be related to poor clinical outcome.




Similar content being viewed by others
References
Qureshi AI, Mendelow AD, Hanley DF (2009) Intracerebral haemorrhage. Lancet 373:1643–1644
Xi G, Keep RF, Hoff JT (2006) Mechanisms of brain injury after intracerebral haemorrhage. Lancet Neurol 5:53–63
Leira R, Dávalos A, Silva Y, Gil-Peralta A, Tejada J, Garcia M, Castillo J, for the Stroke Project, Cerebrovascular Diseases Group of the Spanish Neurological Society (2004) Early neurologic deterioration in intracerebral hemorrhage. Predictors and associated factors. Neurology 63:461–467
Davis SM, Broderick J, Hennerici M, Brun NC, Diringer MN, Mayer SA, Begtrup K, Steiner T, for the Recombinant Activated Factor VII Intracerebral Hemorrhage Trial Investigation (2006) Hematoma growth is a determinant of mortality and poor outcome after intracerebral hemorrhage. Neurology 66:1175–1181
Dowlatshahi D, Demchuck AM, Flaherty ML, Ali M, Lyden PL, Smith EE, on behalf of the VISTA Collaboration (2011) Defining hematoma expansion in intracerebral hemorrhage. Relationship with patient outcomes. Neurology 76:1238–1244
Gebel JM, Jauch EC, Brott TG, Khoury J, Sauerbeck L, Salisbury S, Spilker J, Tomsick TA, Duldner J, Broderick JP (2002) Relative edema volume is a predictor of outcome in patients with hyperacute spontaneous intracerebral hemorrhage. Stroke 33:2636–2641
Venkatasubramanian C, Mlynash M, Finley-Caulfield A, Eyngorn I, Kalimuthu R, Snider RW, Wijman CA (2011) Natural history of perihematomal edema after intracerebral haemorrhage measured by serial magnetic resonance imaging. Stroke 42:73–80
Staykov D, Wagner I, Volbers B, Hauer E-V, Doerfler A, Schwab S, Bardutzky J (2011) Natural course of perihemorrhagic edema after intracerebral haemorrhage. Stroke 42:2625–2629
Arima H, Wang JG, Huang Y, Heeley E, Skulina C, Parsons MW, Peng B, Li Q, Su S, Tao QL, Li YC, Jiang JD, Tai LW, Zhang JL, Xu E, Cheng Y, Morgenstern LB (2009) Anderson CS for the INTERACT Investigators. Neurology 73:1963–1968
Huisman TAG (2003) Diffusion-weighted imaging: basic concepts and application in cerebral stroke and head trauma. Eur Radiol 13:2283–2297
Orakcioglu B, Fierbach JB, Steiner T, Kollmar R, Jüttler E, Becker K, Schwab S, Heiland S, Meyding-Lamadé UK, Schellinger PD (2005) Evolution of early perihemorrhagic changes—ischemia vs. edema: an MRI study in rats. Exp Neurol 193:369–376
Orakcioglu B, Becker K, Sakowitz O, Herweh C, Köhrmann M, Huttner HB, Steiner T, Unterberg A, Schellinger PD (2008) MRI of the perihemorrhagic zone in a rat ICH model: effect of hematoma evacuation. Neurocrit Care 8:448–455
Kidwell CS, Saver JL, Mattiello J, Warach S, Liebeskind DS, Starkman S, Vespa PM, Villablanca JP, Martin NA, Frazee J, Alger JR (2001) Diffusion–perfusion MR evaluation of perihematomal injury in hyperacute intracerebral hemorrhage. Neurology 57:1611–1617
Schellinger PD, Fiebach JB, Hoffmann K, Becker K, Orakcioglu B, Kollmar R, Jüttler E, Schramm P, Schwab S, Sartor K, Hacke W (2003) Stroke MRI in intracerebral hemorrhage. Is there a perihemorrhagic penumbra? Stroke 34:1674–1680
Morita N, Harada M, Yoneda K, Nishitani H, Uno M (2002) A characteristic feature of acute haematomas in the brain on echo-planar diffusion-weighted imaging. Neuroradiology 44:907–911
Carhuapoma JR, Wang P, Beauchamp NJ, Keyl PM, Hanley DF, Barker PB (2000) Diffusion-weighted MRI and proton MR spectroscopy imaging in the study of secondary neuronal injury after intracerebral hemorrhage. Stroke 31:726–732
Carhuapoma JR, Barker PB, Hanley DF, Wang P, Beauchamp NJ (2002) Human brain hemorrhage: quantification of perihematoma edema by use of diffusion-weighted MR imaging. AJNR Am J Neuroradiol 23:1322–1326
Butcher KS, Baird T, MacGregor L, Desmond P, Tress B, Davis S (2004) Perihematomal edema in primary intracerebral hemorrhage is plasma derived. Stroke 35:1879–1885
Forbes KP, Pipe JG, Heiserman JE (2003) Diffusion-weighted imaging provides support for secondary neuronal damage from intraparenchymal hematoma. Neuroradiology 45:363–367
Kamal AK, Dyke JP, Katz JM, Liberato B, Filippi CG, Zimmerman RD, Uluğ AM (2003) Temporal evolution of diffusion after spontaneous supratentorial intracranial hemorrhage. AJNR Am J Neuroradiol 24:895–901
Fainardi E, Borrelli M, Saletti A, Schivalocchi R, Russo M, Azzini C, Cavallo M, Ceruti S, Tamarozzi R, Chieregato A (2006) Evaluation of perihematomal regional apparent diffusion coefficient abnormalities by diffusion-weighted imaging. Acta Neurochir 96(Suppl):81–84
Olivot J-M, Mlynash M, Klienman JT, Straka M, Venkatasubramanian C, Bammer R, Moseley ME, Albers GW, Wijman AC, for the DASH Investigators (2010) MRI profile of the perihematomal region in acute intracerebral hemorrhage. Stroke 41:2681–2683
Chieregato A, Fainardi E (2008) Potential role of surgery in traumatic focal brain lesions as revealed by functional imaging. Intensive Care Med 34:2125–2126
Morgenstern JB, Hemphill JC III, Anderson C, Becker K, Broderick JP, Connolly ES Jr, Greenberg SM, Huang JN, Macdonald RL, Messé SR, Mitchell PH, Selim M, Tamargo RJ, American Heart Association Stroke Council and Council on Cardiovascular Nursing (2010) Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American heart Association/American Stroke Association. Stroke 41:2108–2129
Brott T, Adams HP, Olinger CP, Marler JR, Barsan WG, Biller J, Spilker J, Holleran R, Eberle R, Hertzberg V, Rorick M, Moomaw CJ, Walker M (1989) Measurements of acute cerebral infarction: a clinical examination scale. Stroke 20:864–870
The National Institute of Neurological Disorders and Stroke rt-PA Study Group (1995) Tissue plasminogen activator for acute ischemic stroke. N Engl J Med 333:1581–1587
Ulug AZ, Beauchamp N, Bryan RN, van Zijl PCM (1997) Absolute quantitation of diffusion constants in human stroke. Stroke 28:483–490
Alemany Ripoll M, Stenborg A, Sonninen P, Terent A, Raininko R (2004) Detection and appearance of intraparenchymal haematomas of the brain at 1.5T with spin-echo, FLAIR and GE sequences: poor relationship to the age of the haematoma. Neuroradiology 46:435–443
Volbers B, Staykov D, Wagner I, Dörfler A, Saake M, Schwab S, Bardutzky J (2011) Semi-automatic volumetric assessment of perihemorrhagic edema with computed tomography. Eur J Neurol 18:1323–1328
Burgess RE, Warach S, Schaewe TJ, Copenhaver BR, Alger JR, Vespa P, Martin N, Saver JL, Chelsea S, Kidwell CS (2008) Development and validation of a simple conversion model for comparison of intracerebral hemorrhage volumes measured on CT and gradient recalled echo MRI. Stroke 39:2017–2020
Brott T, Broderick J, Kothari R, Barsan W, Tomsick T, Sauerbeck L, Spilker J, Duldner J, Khoury J (1997) Early hemorrhage growth in patients with intracerebral hemorrhage. Stroke 28:1–5
Maldjian JA, Listerud J, Moonis G, Siddiqi F (2001) Computing diffusion rates in T2-dark hematomas and areas of low T2 signal. AJNR Am J Neuroradiol 22:112–118
Lou M, Lieb K, Selim M (2009) The relationship between hematoma iron content and perihematomal edema: an MRI study. Cerebrovasc Dis 27:266–271
Khatri IA, Verma N, Alkawi A, Janjua N, Kirmani JF (2009) Cerebral vasospasm in intracerebral hemorrhage—case report. J Vasc Interv Neurol 2:139–141
Wardlaw JM (2010) Blood–brain barrier and cerebral small vessel disease. J Neurol Sci 299:66–71
Kidwell CS, Burgess R, Menon R, Warach S, Latour LL (2011) Hyperacute injury marker (HARM) in primary hemorrhage. A distinct form of CNS barrier disruption. Neurology 77:1725–1728
Latour LL, Kang D-W, Ezzedine MA, Chalela JA, Warach S (2004) Early blood–brain barrier disruption in human focal brain ischemia. Ann Neurol 56:468–477
Lou M, Al-Hazzani A, Goddeau RP, Novak V, Selim M (2010) Relationship between white-matter hyperintensities and hematoma volume and growth in patients with intracerebral hemorrhage. Stroke 41:34–40
Lee S-H, Kim BJ, Ryu W-S, Kim CK, Kim N, Park B-J, Yoon B-W (2010) White matter lesions and poor outcome after intracerebral hemorrhage. A nationwide cohort study. Neurology 74:1502–1510
Prabhakaran S, Gupta R, Ouyang B, John S, Temes RE, Mohammad Y, Lee VH, Bleck TP (2010) Acute brain infarcts after spontaneous intracerebral hemorrhage. A diffusion-weighted imaging study. Stroke 41:89–94
Gregorie SM, Charidimou A, Gadapa N, Dolan E, Antoun N, Peeters A, Vandermeeren Y, Laloux P, Baron J-C, Jäger HR, Werring DJ (2011) Acute ischaemic brain lesions in intracerebral haemorrhage: multicentre cross-sectional magnetic resonance imaging study. Brain 134:2376–2386
Steiner T, Bösel J (2010) Options to restrict hematoma expansion after spontaneous intracerebral hemorrhage. Stroke 41:402–409
Sasaki M, Yamada K, Watanabe Y, Matsui M, Ida M, Fujiwara S, Shibata E, for the Acute Stroke IMAGING Standardization Group-Japan (ASIST-Japan) Investigators (2008) Variability in absolute apparent diffusion coefficient values across different platforms may be substantial: a multivendor multi-institutional comparison study. Radiology 249:624–630
Helenius J, Soinne L, Salonen O, Kaste M, Tatlisumak T (2002) Leukoaraiosis, ischemic stroke, and normal white matter on diffusion-weighted MRI. Stroke 33:45–50
Acknowledgments
The authors thank Dr. Elizabeth Jenkins for helpful corrections to the manuscript.
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fainardi, E., Borrelli, M., Saletti, A. et al. Temporal changes in perihematomal apparent diffusion coefficient values during the transition from acute to subacute phases in patients with spontaneous intracerebral hemorrhage. Neuroradiology 55, 145–156 (2013). https://doi.org/10.1007/s00234-012-1093-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-012-1093-x