Abstract
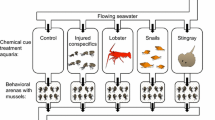
A predator–prey interaction occurs between the asterinid seastars Meridiastra calcar and Parvulastra exigua, which are sympatric on the shores of southeast Australia. These seastars are important as omnivorous predators/scavengers (M. calcar) and surface grazing herbivores (P. exigua). Meridiastra calcar inhabits the lower shore, whilst P. exigua occupies the mid to high shore and they can co-occur in the mid-intertidal zone. Parvulastra exigua exhibits a fleeing response when they encounter M. calcar. We investigated this response to determine the cues that elicit this behaviour. We also determined if M. calcar moves toward P. exigua. Physical contact with M. calcar resulted in an 8.6-fold increase in velocity of P. exigua compared to basal locomotion. Water conditioned by M. calcar (chemosensory cue only) also elicited an increase in velocity, but this was lower, a 3.5-fold increase. The escape trajectory of P. exigua was most linear, 180° away from the point of touch when contacted with M. calcar. In contrast, while touch with a sham stimulus elicited some movement, velocity was low, and the direction of movement was not linear away from the point of stimulation. Meridiastra calcar did not move toward P. exigua in choice experiments, and this was not altered if P. exigua had been damaged. Our results suggest that the escape response of P. exigua is elicited by a physical touch from M. calcar and that they detect chemosensory cues from this predator. Chemosensory cues emanating from M. calcar populations in the lower intertidal zone may influence the behaviour of P. exigua to remain higher on the shore. This first study of a predator–prey interaction in intertidal seastars suggests that this behaviour influences their distribution across the tidal gradient.




Similar content being viewed by others
Availability of data and material
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Code availability
The code is available from the free open access R packages ‘circular’, ‘CircStat’, ‘emmmeans’, ‘ggplot2’.
References
Beer S, Wentzel C, Petie R, Garm A (2016) Active control of the visual field in the starfish Acanthaster planci. Vision Res 127:28–34. https://doi.org/10.1016/j.visres.2016.05.007
Bennett I (1987) Australian Seashores. Angus & Robertson Publishers, Sydney
Brewer R, Konar B (2005) Chemosensory responses and foraging behavior of the seastar Pycnopodia helianthoides. Mar Biol 147:789–795. https://doi.org/10.1007/s00227-005-1608-7
Byrne M (1985) Evisceration behaviour and the seasonal incidence of evisceration in the holothurian Eupentacta quinquesemita (Selenka). Ophelia 24:75–90
Castilla JC, Crisp DJ (1970) Responses of Asterias rubens to olfactory stimuli. J Mar Biol Ass 50:829–847. https://doi.org/10.1017/S0025315400005075
Chivers DP, Smith RJF (1998) Chemical alarm signalling in aquatic predator-prey systems: a review and prospectus. Écoscience 5:338–352. https://doi.org/10.1080/11956860.1998.11682471
Dale J (1997) Chemosensory search behavior in the starfish Asterias forbesi. Biol Bull 193:210–212. https://doi.org/10.1086/BBLv193n2p210
Dartnall AJ (1970) The asterinid sea stars of Tasmania. Pap Proc R Soc Tasmania 104:73–78
Day R, Dowell A, Sant G, Klemke J, Shaw C (1995) Patchy predation: foraging behaviour of Coscinasterias calamaria and escape responses of Haliotis rubra. Mar Freshw Behav and Physiol 26:11–33. https://doi.org/10.1080/10236249509378925
Domenici P, Blagburn JM, Bacon JP (2011a) Animal escapology I: theoretical issues and emerging trends in escape trajectories. J Exp Biol 214:2463–2473. https://doi.org/10.1242/jeb.029652
Domenici P, Blagburn JM, Bacon JP (2011b) Animal escapology II: escape trajectory case studies. J Exp Biol 214:2474–2494. https://doi.org/10.1242/jeb.053801
Escobar J, Navarrete S (2011) Risk recognition and variability in escape responses among intertidal molluskan grazers to the sun star Heliaster helianthus. Mar Ecol Prog Ser 421:151–161. https://doi.org/10.3354/meps08883
Feder HM (1963) Gastropod defensive responses and their effectiveness in reducing predation by starfishes. Ecology 44:505–512. https://doi.org/10.2307/1932529
Francour P (1997) Predation on holothurians: a literature review. Invert Biol 116:52. https://doi.org/10.2307/3226924
Garm A (2017) Sensory biology of starfish—with emphasis on recent discoveries in their visual ecology. Integr Comp Biol 57:1082–1092. https://doi.org/10.1093/icb/icx086
Gaymer C, Himmelman J (2008) A keystone predatory sea star in the intertidal zone is controlled by a higher-order predatory sea star in the subtidal zone. Mar Ecol Prog Ser 370:143–153. https://doi.org/10.3354/meps07663
Guderley HE, Himmelman JH, Nadeau M, Cortes HP, Tremblay I, Janssoone X (2015) Effect of different predators on the escape response of Placopecten magellanicus. Mar Biol 162:1407–1415. https://doi.org/10.1007/s00227-015-2677-x
Güler M, Lök A (2015) Foraging behaviors of sea stars, Marthasterias glacialis and Astropecten aranciacus (Asteroidea) and predator–prey interactions with warty venus clam, Venus verrucosa (Bivalvia). J Exp Mar Biol Ecol 465:99–106. https://doi.org/10.1016/j.jembe.2014.12.018
Hall MR, Kocot KM, Baughman KW, Fernandez-Valverde SL, Gauthier MEA, Hatleberg WL, Krishnan A, McDougall C, Motti CA, Shoguchi E, Wang T, Xiang X, Zhao M, Bose U, Shinzato C, Hisata K, Fujie M, Kanda M, Cummins SF, Satoh N, Degnan SM, Degnan BM (2017) The crown-of-thorns starfish genome as a guide for biocontrol of this coral reef pest. Nature 544:231–234. https://doi.org/10.1038/nature22033
Hart MW, Keever CC, Dartnall AJ, Byrne M (2006) Morphological and genetic variation indicate cryptic species within Lamarck’s little sea star, Parvulastra (=Patiriella) exigua. Biol Bull 210:158–167. https://doi.org/10.2307/4134604
Jellison BM, Gaylord B (2019) Shifts in seawater chemistry disrupt trophic links within a simple shoreline food web. Oecologia. https://doi.org/10.1007/s00442-019-04459-0
Jellison BM, Ninokawa AT, Hill TM, Sanford E, Gaylord B (2016) Ocean acidification alters the response of intertidal snails to a key sea star predator. Proc R Soc B 283:20160890. https://doi.org/10.1098/rspb.2016.0890
Landler L, Ruxton GD, Malkemper EP (2018) Circular data in biology: advice for effectively implementing statistical procedures. Behav Ecol Sociobiol 72:128. https://doi.org/10.1007/s00265-018-2538-y
Lawrence JM (1991) A chemical alarm response in Pycnopodia helianthoides (Echinodermata: Asteroidea). Mar Behav Physiol 19:39–44. https://doi.org/10.1080/10236249109378793
Lawrence JM, Gaymer CF (2012) Autotomy of rays of Heliaster helianthus (Asteroidea: Echinodermata). Zoosymposia 7:173–176. https://doi.org/10.11646/zoosymposia.7.1.16
Legault C, Himmelman JH (1993) Relation between escape behaviour of benthic marine invertebrates and the risk of predation. J Exp Mar Biol Ecol 170:55–74. https://doi.org/10.1016/0022-0981(93)90129-C
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640. https://doi.org/10.1139/z90-092
Mahon AR, Amsler CD, McClintock JB, Baker BJ (2002) Chemo-tactile predator avoidance responses of the common Antarctic limpet Nacella concinna. Polar Biol 25:469–473. https://doi.org/10.1007/s00300-002-0368-9
Manzur T, Gonzalez-Mendez A, Broitman BR (2018) Scales of predator detection behavior and escape in Fissurella limbata: a field and laboratory assessment. Mar Ecol 39:e12492. https://doi.org/10.1111/maec.12492
Margolin AS (1976) Swimming of the sea cucumber Parastichopus californicus (Stimpson) in response to sea stars. Ophelia 15:105–114. https://doi.org/10.1080/00785326.1976.10425452
Markowska M, Kidawa A (2007) Encounters between Antarctic limpets, Nacella concinna, and predatory sea stars, Lysasterias sp., in laboratory and field experiments. Mar Biol 151:1959–1966. https://doi.org/10.1007/s00227-007-0633-0
Martinez AS, Byrne M, Coleman RA (2016) What and when to eat? Investigating the feeding habits of an intertidal herbivorous starfish. Mar Biol 163:166. https://doi.org/10.1007/s00227-016-2942-7
Martinez AS, Byrne M, Coleman RA (2017a). Filling in the grazing puzzle: A synthesis of herbivory in starfish, in: Hawkins SJ, Evans AJ, Dale AC, Firth LB, Hughes DJ, Smith IP (Eds.), Ocean Mar Biol Ann Rev 55: 11-44. https://doi.org/10.1201/b21944-2
Martinez AS, Queiroz EV, Bryson M, Byrne M, Coleman RA (2017b) Incorporating in situ habitat patchiness in site selection models reveals that site fidelity is not always a consequence of animal choice. J Anim Ecol 86:847–856. https://doi.org/10.1111/1365-2656.12668
McClintock JB, Angus RA, Ho C, Amsler CD, Baker BJ (2008) A laboratory study of behavioral interactions of the Antarctic keystone sea star Odontaster validus with three sympatric predatory sea stars. Mar Biol 154:1077–1084. https://doi.org/10.1007/s00227-008-1001-4
Menge BA (2000) Top-down and bottom-up community regulation in marine rocky intertidal habitats. J Exp Mar Biol Ecol 250:257–289. https://doi.org/10.1016/S0022-0981(00)00200-8
Moitoza DJ, Phillips DW (1979) Prey defense, predator preference, and nonrandom diet: the interactions between Pycnopodia helianthoides and two species of sea urchins. Mar Biol 53:299–304. https://doi.org/10.1007/BF00391611
Montgomery EM (2014) Predicting crawling speed relative to mass in sea stars. J Exp Mar Biol Ecol 458:27–33. https://doi.org/10.1016/j.jembe.2014.05.009
Moore PA, Lepper DME (1997) Role of chemical signals in the orientation behavior of the sea star Asterias forbesi. Biol Bull 192:410–417. https://doi.org/10.2307/1542750
Morissette S, Himmelman JH (2000a) Decision of the asteroid Leptasterias polaris to abandon its prey when confronted with its predator, the asteroid Asterias vulgaris. J Exp Mar Biol Ecol 252:151–157. https://doi.org/10.1016/S0022-0981(00)00229-X
Morissette S, Himmelman JH (2000b) Subtidal food thieves: interactions of four invertebrate kleptoparasites with the sea star Leptasterias polaris. Anim Behav 60:531–543. https://doi.org/10.1006/anbe.2000.1500
Motti CA, Bose U, Roberts RE, McDougall C, Smith MK, Hall MR, Cummins SF (2018) Chemical ecology of chemosensation in Asteroidea: insights towards management strategies of pest species. J Chem Ecol 44:147–177. https://doi.org/10.1007/s10886-018-0926-4
Ormond RFG, Campbell AC, Head SH, Moore RJ, Rainbow PR, Saunders AP (1973) Formation and breakdown of aggregations of the crown-of-thorns starfish, Acanthaster planci (L.). Nature 246:167–169. https://doi.org/10.1038/246167a0
Paine RT (1966) Food web complexity and species diversity. American Nat 100:65–75. https://doi.org/10.1086/282400
Paine RT (1969) A note on trophic complexity and community stability. Am Nat 103:91–93. https://doi.org/10.1086/282586
Petie R, Hall MR, Hyldahl M, Garm A (2016) Visual orientation by the crown-of-thorns starfish (Acanthaster planci). Coral Reefs 35:1139–1150. https://doi.org/10.1007/s00338-016-1478-0
Pewsey A, Neuhäuser M, Ruxton GD (2014) Circular statistics in R. Oxford University Press USA-OSO, Oxford, United Kingdom
Phillips DW (1976) The effect of a species-specific avoidance response to predatory starfish on the intertidal distribution of two gastropods. Oecologia 23:83–94. https://doi.org/10.1007/BF00557847
Power ME, Tilman D, Estes JA, Menge BA, Bond WJ, Mills LS, Daily G, Castilla JC, Lubchenco J, Paine RT (1996) Challenges in the quest for keystones. Bioscience 46:609–620. https://doi.org/10.2307/1312990
Pratchett M, Caballes C, Wilmes J, Matthews S, Mellin C, Sweatman H, Nadler L, Brodie J, Thompson C, Hoey J, Bos A, Byrne M, Messmer V, Fortunato S, Chen C, Buck A, Babcock R, Uthicke S (2017) Thirty years of research on crown-of-thorns starfish (1986–2016): scientific advances and emerging opportunities. Diversity 9:41. https://doi.org/10.3390/d9040041
Pruitt JN, Stachowicz JJ, Sih A (2012) Behavioral types of predator and prey jointly determine prey survival: potential implications for the maintenance of within-species behavioral variation. Am Nat 179:217–227. https://doi.org/10.1086/663680
Quinn G, Keough M (2002) Experimental Design and Data Dnalysis for Biologists. Cambridge University Press, Cambridge. https://doi.org/10.1017/CBO9780511806384
R Core Team (2020) R: a language and environment for statistical computing. Austria, Vienna
Schram JB, Amsler CD, McClintock JB (2019) Contrasting chemotactic escape responses of the common Antarctic gastropod Margarella antarctica to four species of sympatric sea stars. Pol Sci 22:100486. https://doi.org/10.1016/j.polar.2019.100486
Schultz JA, Cloutier RN, Côté IM (2016) Evidence for a trophic cascade on rocky reefs following sea star mass mortality in British Columbia. PeerJ 4:e1980. https://doi.org/10.7717/peerj.1980
Shepherd SA, Thomas IM (1982) Marine Invertebrates of Southern Australia. D.J. Woolman, Government Printer, Adelaide
Sih A, Ferrari MCO, Harris DJ (2011) Evolution and behavioural responses to human-induced rapid environmental change. Evol Appl 4:367–387. https://doi.org/10.1111/j.1752-4571.2010.00166.x
Sloan NA (1984) Interference and aggregation: close encounters of the starfish kind. Ophelia 23:23–31. https://doi.org/10.1080/00785236.1984.10426602
Sloan NA, Northway SM (1982) Chemoreception by the asteroid Crossaster papposus (L.). J Exp Mar Biol Ecol 61:85–98. https://doi.org/10.1016/0022-0981(82)90023-5
Stevenson JP (1992) A possible modification of the distribution of the intertidal seastar Patiriella exigua (Lamarck) (Echinodermata: Asteroidea) by Patiriella calcar (Lamarck). J Exp Mar Biol Ecol 155:41–54. https://doi.org/10.1016/0022-0981(92)90026-7
Van Veldhuizen HD, Oakes VJ (1981) Behavioral responses of seven species of asteroids to the asteroid predator, Solaster dawsoni (responses of asteroids to the predator Solaster dawsoni). Oecologia 48:214–220. https://doi.org/10.1007/BF00347967
Vermeij GJ (2020) Getting out of arms’ way: star wars and snails on the seashore. Biol Bull 239:209–217. https://doi.org/10.1086/711487
Walker JA, Ghalambor CK, Griset OL, Mckenney D, Reznick DN (2005) Do faster starts increase the probability of evading predators? Funct Ecology 19:808–815. https://doi.org/10.1111/j.1365-2435.2005.01033.x
Watson SA, Lefevre S, McCormick MI, Domenici P, Nilsson GE, Munday PL (2014) Marine mollusc predator-escape behaviour altered by near-future carbon dioxide levels. Proc R Soc B 281:20132377. https://doi.org/10.1098/rspb.2013.2377
Zimmer RK, Ferrier GA, Kim SJ, Ogorzalek Loo RR, Zimmer CA, Loo JA (2017) Keystone predation and molecules of keystone significance. Ecology 98:1710–1721. https://doi.org/10.1002/ecy
Acknowledgements
We thank Sergio Torres Gabarda of the Sydney Institute of Marine Science (SIMS) for assistance with experiments. We thank Dione Deaker, Regina Balogh, Hamish Campbell, Katie Erickson, Alex McGrath and Sian Liddy for assistance with experiments and collections. Dr Karen Chan (Swarthmore College) is thanked for advice on animal tracking. We thank the reviewers and editor for their insightful comments that improved this manuscript. This work is Sydney Institute of Marine Science contribution number 276.
Funding
This study was not externally funded.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Ethics approval
This study did not require ethics approval. All applicable national and/or institutional guidelines for sampling, care and experimental use of organisms for the study have been followed and all necessary approvals have been obtained.
Additional information
Responsible Editor: M. Thiel.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewers: Undisclosed experts.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
McLaren, E.J., Byrne, M. Predator–prey behavioural interactions between the asterinid seastars Meridiastra calcar and Parvulastra exigua sympatric on the rocky shores of southeast Australia. Mar Biol 168, 124 (2021). https://doi.org/10.1007/s00227-021-03933-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-021-03933-9