Abstract
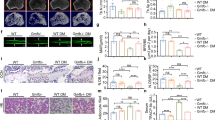
Hyperglycemia induces osteoclastogenesis and bone resorption through complicated, undefined mechanisms. Ca2+/calmodulin-dependent protein kinase II (CaMKII) promotes osteoclastogenesis, and could be activated by hyperglycemia. Here, we investigated whether CaMKII is involved in hyperglycemia-induced osteoclastogenesis and subsequent bone resorption. Osteoclast formation, bone resorption, CaMKII expression and phosphorylation were measured under high glucose in vitro and in streptozotocin-induced hyperglycemia rats with or without CaMKII inhibitor KN93. The results showed that 25 mmol/L high glucose in vitro promoted cathepsin K and tartrate-resistant acid phosphatase expression (p < 0.05) and osteoclast formation (p < 0.01) associated with enhancing β isoform expression (p < 0.05) and CaMKII phosphorylation (p < 0.001). Hyperglycemia promoted the formation of osteoclasts and resorption of trabecular and alveolar bone, and inhibited sizes of femur and mandible associated with enhanced CaMKII phosphorylation (p < 0.001) in rats. All these changes could be alleviated by KN93. These findings imply that CaMKII participates not only in hyperglycemia-induced osteoclastogenesis and subsequent bone resorption, but also in the hyperglycemia-induced developmental inhibition of bone.







Similar content being viewed by others
References
Seppala B, Ainamo J (1994) A site-by-site follow-up study on the effect of controlled versus poorly controlled insulin-dependent diabetes mellitus. J Clin Periodontol 21(3):161–165
Loureiro MB, Ururahy MA, Freire-Neto FP, Oliveira GH, Duarte VM, Luchessi AD, Brandao-Neto J, Hirata RD, Hirata MH, Maciel-Neto JJ, Arrais RF, Almeida MG, Rezende AA (2014) Low bone mineral density is associated to poor glycemic control and increased OPG expression in children and adolescents with type 1 diabetes. Diab Res Clin Pract 103(3):452–457. https://doi.org/10.1016/j.diabres.2013.12.018
Novotna M, Podzimek S, Broukal Z, Lencova E, Duskova J (2015) Periodontal diseases and dental caries in children with type 1 diabetes mellitus. Mediat Inflamm 2015:379626. https://doi.org/10.1155/2015/379626
Iitsuka N, Hie M, Tsukamoto I (2013) Zinc supplementation inhibits the increase in osteoclastogenesis and decrease in osteoblastogenesis in streptozotocin-induced diabetic rats. Eur J Pharmacol 714(1–3):41–47. https://doi.org/10.1016/j.ejphar.2013.05.020
Ma R, Wang L, Zhao B, Liu C, Liu H, Zhu R, Chen B, Li L, Zhao D, Mo F, Li Y, Niu J, Jiang G, Fu M, Bromme D, Gao S, Zhang D (2017) Diabetes perturbs bone microarchitecture and bone strength through regulation of sema3A/IGF-1/beta-catenin in rats. Cell Physiol Biochem 41 (1):55–66. https://doi.org/10.1159/000455936
Li H, Yang H, Ding Y, Aprecio R, Zhang W, Wang Q, Li Y (2013) Experimental periodontitis induced by Porphyromonas gingivalis does not alter the onset or severity of diabetes in mice. J Periodontal Res 48(5):582–590. https://doi.org/10.1111/jre.12041
Erdal N, Gurgul S, Demirel C, Yildiz A (2012) The effect of insulin therapy on biomechanical deterioration of bone in streptozotocin (STZ)-induced type 1 diabetes mellitus in rats. Diab Res Clin Pract 97(3):461–467. https://doi.org/10.1016/j.diabres.2012.03.005
de Oliveira G, Basso TLD, Fontanari LA, Faloni APS, Marcantonio EJ, Orrico SRP (2017) Glycemic control protects against trabecular bone microarchitectural damage in a juvenile male rat model of streptozotocin-induced diabetes. Endocr Res 42(3):171–179. https://doi.org/10.1080/07435800.2017.1292521
Reni C, Mangialardi G, Meloni M, Madeddu P (2016) Diabetes stimulates osteoclastogenesis by acidosis-induced activation of transient receptor potential cation channels. Sci Rep 6:30639. https://doi.org/10.1038/srep30639
Balci Yuce H, Karatas O, Aydemir Turkal H, Pirim Gorgun E, Ocakli S, Benli I, Cayli S (2016) The effect of melatonin on bone loss, diabetic control, and apoptosis in rats with diabetes with ligature-induced periodontitis. J Periodontol 87(4):e35–e43. https://doi.org/10.1902/jop.2015.150315
Seales EC, Micoli KJ, McDonald JM (2006) Calmodulin is a critical regulator of osteoclastic differentiation, function, and survival. J Cell Biochem 97(1):45–55. https://doi.org/10.1002/jcb.20659
Chang EJ, Ha J, Huang H, Kim HJ, Woo JH, Lee Y, Lee ZH, Kim JH, Kim HH (2008) The JNK-dependent CaMK pathway restrains the reversion of committed cells during osteoclast differentiation. J Cell Sci 121(Pt 15):2555–2564. https://doi.org/10.1242/jcs.028217
Yoon SH, Ryu J, Lee Y, Lee ZH, Kim HH (2011) Adenylate cyclase and calmodulin-dependent kinase have opposite effects on osteoclastogenesis by regulating the PKA-NFATc1 pathway. J Bone Miner Res 26(6):1217–1229. https://doi.org/10.1002/jbmr.310
Nishio S, Teshima Y, Takahashi N, Thuc LC, Saito S, Fukui A, Kume O, Fukunaga N, Hara M, Nakagawa M, Saikawa T (2012) Activation of CaMKII as a key regulator of reactive oxygen species production in diabetic rat heart. J Mol Cell Cardiol 52(5):1103–1111. https://doi.org/10.1016/j.yjmcc.2012.02.006
Li J, Wang P, Yu S, Zheng Z, Xu X (2012) Calcium entry mediates hyperglycemia-induced apoptosis through Ca(2+)/calmodulin-dependent kinase II in retinal capillary endothelial cells. Mol Vis 18:2371–2379
Li J, Zhao SZ, Wang PP, Yu SP, Zheng Z, Xu X (2012) Calcium mediates high glucose-induced HIF-1alpha and VEGF expression in cultured rat retinal Muller cells through CaMKII-CREB pathway. Acta Pharmacol Sinica 33(8):1030–1036. https://doi.org/10.1038/aps.2012.61
Yousif MH, Akhtar S, Walther T, Benter IF (2008) Role of Ca2+/calmodulin-dependent protein kinase II in development of vascular dysfunction in diabetic rats with hypertension. Cell Biochem Funct 26(2):256–263. https://doi.org/10.1002/cbf.1446
Li J, Wang P, Ying J, Chen Z, Yu S (2016) Curcumin attenuates retinal vascular leakage by inhibiting calcium/calmodulin-dependent protein kinase ii activity in streptozotocin-induced diabetes. Cell Physiol Biochem 39 (3):1196–1208. https://doi.org/10.1159/000447826
Jelicic Kadic A, Boric M, Kostic S, Sapunar D, Puljak L (2014) The effects of intraganglionic injection of calcium/calmodulin-dependent protein kinase II inhibitors on pain-related behavior in diabetic neuropathy. Neuroscience 256:302–308. https://doi.org/10.1016/j.neuroscience.2013.10.032
Bouxsein ML, Boyd SK, Christiansen BA, Guldberg RE, Jepsen KJ, Muller R (2010) Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J Bone Miner Res 25(7):1468–1486. https://doi.org/10.1002/jbmr.141
Kim JM, Jeong D, Kang HK, Jung SY, Kang SS, Min BM (2007) Osteoclast precursors display dynamic metabolic shifts toward accelerated glucose metabolism at an early stage of RANKL-stimulated osteoclast differentiation. Cell Physiol Biochem 20 (6):935–946. https://doi.org/10.1159/000110454
Wittrant Y, Gorin Y, Woodruff K, Horn D, Abboud HE, Mohan S, Abboud-Werner SL (2008) High d(+)glucose concentration inhibits RANKL-induced osteoclastogenesis. Bone 42(6):1122–1130. https://doi.org/10.1016/j.bone.2008.02.006
Park JH, Lee NK, Lee SY (2017) Current understanding of RANK signaling in osteoclast differentiation and maturation. Mol Cells 40(10):706–713. https://doi.org/10.14348/molcells.2017.0225
Larsen KI, Falany ML, Ponomareva LV, Wang W, Williams JP (2002) Glucose-dependent regulation of osteoclast H(+)-ATPase expression: potential role of p38 MAP-kinase. J Cell Biochem 87(1):75–84. https://doi.org/10.1002/jcb.10252
Yao CH, Zhang P, Zhang L (2012) Differential protein and mRNA expression of CaMKs during osteoclastogenesis and its functional implications. Biochem Cell Biol 90 (4):532–539. https://doi.org/10.1139/o2012-002
Nagasaki N, Hirano T, Kawaguchi SY (2014) Opposite regulation of inhibitory synaptic plasticity by alpha and beta subunits of Ca(2+)/calmodulin-dependent protein kinase II. J Physiol 592(22):4891–4909. https://doi.org/10.1113/jphysiol.2014.280230
Lin YC, Redmond L (2008) CaMKIIbeta binding to stable F-actin in vivo regulates F-actin filament stability. Proc Natl Acad Sci USA 105(41):15791–15796. https://doi.org/10.1073/pnas.0804399105
Waggener CT, Dupree JL, Elgersma Y, Fuss B (2013) CaMKIIbeta regulates oligodendrocyte maturation and CNS myelination. J Neurosci 33(25):10453–10458. https://doi.org/10.1523/jneurosci.5875-12.2013
Sugawara T, Hisatsune C, Miyamoto H, Ogawa N, Mikoshiba K (2017) Regulation of spinogenesis in mature Purkinje cells via mGluR/PKC-mediated phosphorylation of CaMKIIbeta. Proc Natl Acad Sci USA 114(26):E5256–E5265. https://doi.org/10.1073/pnas.1617270114
Zhou M, Liu Z, Yu J, Li S, Tang M, Zeng L, Wang H, Xie H, Peng L, Huang H, Zhou C, Xie P, Zhou J (2018) Quantitative proteomic analysis reveals synaptic dysfunction in the amygdala of rats susceptible to chronic mild stress. Neuroscience 376:24–39. https://doi.org/10.1016/j.neuroscience.2018.02.010
Kramerova I, Torres JA, Eskin A, Nelson SF, Spencer MJ (2018) Calpain 3 and CaMKIIbeta signaling are required to induce HSP70 necessary for adaptive muscle growth after atrophy. Hum Mol Genet 27(9):1642–1653. https://doi.org/10.1093/hmg/ddy071
Liu Q, Yang HX, Wan XH, Zhang M, Zhang J, Lu L, Xie M, Ren HT, Yu SB, Liu XD, Wang M (2018) Calcium-/calmodulin-dependent protein kinase II in occlusion-induced degenerative cartilage of rat mandibular condyle. J Oral Rehabil. https://doi.org/10.1111/joor.12629
Smogorzewski M, Galfayan V, Massry SG (1998) High glucose concentration causes a rise in [Ca2+]i of cardiac myocytes. Kidney Int 53(5):1237–1243. https://doi.org/10.1046/j.1523-1755.1998.00868.x
Symonian M, Smogorzewski M, Marcinkowski W, Krol E, Massry SG (1998) Mechanisms through which high glucose concentration raises [Ca2+]i in renal proximal tubular cells. Kidney Int 54(4):1206–1213. https://doi.org/10.1046/j.1523-1755.1998.00109.x
Boldizsar F, Berki T, Miseta A, Nemeth P (2002) Effect of hyperglycemia on the basal cytosolic free calcium level, calcium signal and tyrosine-phosphorylation in human T-cells. Immunology letters 82(1–2):159–164
Feng N, Anderson ME (2017) CaMKII is a nodal signal for multiple programmed cell death pathways in heart. J Mol Cell Cardiol 103:102–109. https://doi.org/10.1016/j.yjmcc.2016.12.007
Gaertner TR, Kolodziej SJ, Wang D, Kobayashi R, Koomen JM, Stoops JK, Waxham MN (2004) Comparative analyses of the three-dimensional structures and enzymatic properties of alpha, beta, gamma and delta isoforms of Ca2+-calmodulin-dependent protein kinase II. J Biol Chem 279(13):12484–12494. https://doi.org/10.1074/jbc.M313597200
Erickson JR, Pereira L, Wang L, Han G, Ferguson A, Dao K, Copeland RJ, Despa F, Hart GW, Ripplinger CM, Bers DM (2013) Diabetic hyperglycaemia activates CaMKII and arrhythmias by O-linked glycosylation. Nature 502(7471):372–376. https://doi.org/10.1038/nature12537
Nakashima T, Takayanagi H (2011) New regulation mechanisms of osteoclast differentiation. Ann N Y Acad Sci 1240:E13–E18. https://doi.org/10.1111/j.1749-6632.2011.06373.x
Ang ES, Zhang P, Steer JH, Tan JW, Yip K, Zheng MH, Joyce DA, Xu J (2007) Calcium/calmodulin-dependent kinase activity is required for efficient induction of osteoclast differentiation and bone resorption by receptor activator of nuclear factor kappa B ligand (RANKL). J Cell Physiol 212(3):787–795. https://doi.org/10.1002/jcp.21076
Shanbhogue VV, Hansen S, Frost M, Jorgensen NR, Hermann AP, Henriksen JE, Brixen K (2015) Bone geometry, volumetric density, microarchitecture, and estimated bone strength assessed by HR-pQCT in adult patients with type 1 diabetes mellitus. J bone Miner Res 30(12):2188–2199. https://doi.org/10.1002/jbmr.2573
Abdalrahaman N, McComb C, Foster JE, McLean J, Lindsay RS, McClure J, McMillan M, Drummond R, Gordon D, McKay GA, Shaikh MG, Perry CG, Ahmed SF (2015) Deficits in trabecular bone microarchitecture in young women with type 1 diabetes mellitus. J Bone Miner Res 30(8):1386–1393. https://doi.org/10.1002/jbmr.2465
Oikawa A, Siragusa M, Quaini F, Mangialardi G, Katare RG, Caporali A, van Buul JD, van Alphen FP, Graiani G, Spinetti G, Kraenkel N, Prezioso L, Emanueli C, Madeddu P (2010) Diabetes mellitus induces bone marrow microangiopathy. Arterioscler Thromb Vasc Biol 30(3):498–508. https://doi.org/10.1161/atvbaha.109.200154
Kim HJ, Park KM, Tak HJ, Choi JW, Kang SH, Park W, Lee SH (2018) Three-dimensional growth pattern of the rat mandible revealed by periodic live micro-computed tomography. Arch Oral Biol 87:94–101. https://doi.org/10.1016/j.archoralbio.2017.12.012
Bonfig W, Kapellen T, Dost A, Fritsch M, Rohrer T, Wolf J, Holl RW (2012) Growth in children and adolescents with type 1 diabetes. J Pediatr 160(6):900–903 e902. https://doi.org/10.1016/j.jpeds.2011.12.007
Acknowledgements
The work was supported by the National Key Research and Development Program of China (2017YFA0104800) and National Natural Science Foundation of China (Grant No. 81200792).
Author information
Authors and Affiliations
Contributions
YS, SG, YW and WT designed the study; YS and SG performed the experiments; YS and GC analyzed the data; YS prepared the first draft of the paper; YS, SG, YD, YW and WT revised the paper. All authors reviewed and approved the final version. WT is guarantor. All authors agree to be accountable for the work and to ensure that any questions relating to the accuracy and integrity of the paper are investigated and properly resolved.
Corresponding authors
Ethics declarations
Conflict of interest
Yanxin Shen, Shujuan Guo, Guoqing Chen, Yi Ding, Yafei Wu and Weidong Tian declare no conflict of interest.
Human and Animal Rights and Informed Consent
All procedures performed in studies involving animals were in accordance with the ethical standards of the ethics committee of Sichuan University.
Rights and permissions
About this article
Cite this article
Shen, Y., Guo, S., Chen, G. et al. Hyperglycemia Induces Osteoclastogenesis and Bone Destruction Through the Activation of Ca2+/Calmodulin-Dependent Protein Kinase II. Calcif Tissue Int 104, 390–401 (2019). https://doi.org/10.1007/s00223-018-0499-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-018-0499-9