Abstract
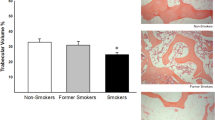
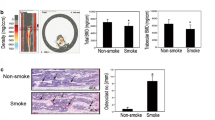
There is no clear evidence to show the direct causal relationship between passive cigarette smoking and osteoporosis. Furthermore, the underlying mechanism is unknown. The objective of this study is to demonstrate the effects of long-term passive cigarette smoking on bone metabolism and microstructure by a mouse model and cell culture systems. BALB/c mice were exposed to 2 or 4 % cigarette smoke for 14 weeks. The bone turnover biochemical markers in urine and serum and also the bone micro-architecture by micro-CT were compared with the control group exposed to normal ambient air. In the cell culture experiments, mouse MC3T3-E1 and RAW264.7 cell lines to be employed as osteoblast and osteoclast, respectively, were treated with the sera obtained from 4 % smoking or control mice. Their actions on cell viability, differentiation, and function on these bone cells were assessed. The urinary mineral and deoxypyridinoline (DPD) levels, and also the serum alkaline phosphatase activity, were significantly higher in the 4 % smoking group when compared with the control group, indicating an elevated bone metabolism after cigarette smoking. In addition, femoral osteopenic condition was observed in the 4 % smoking group, as shown by the decrease of relative bone volume and trabecular thickness. In isolated cell studies, osteoblast differentiation and bone formation were inhibited while osteoclast differentiation was increased. The current mouse smoking model and the isolated cell studies demonstrate that passive cigarette smoke could induce osteopenia by exerting a direct detrimental effect on bone cells differentiation and further on bone remodeling process.






Similar content being viewed by others
Abbreviations
- ALP:
-
Alkaline phosphatase
- ALT:
-
Alanine transaminase
- AST:
-
Aspartate aminotransferase
- BMD:
-
Bone mineral density
- BV/TV:
-
Bone volume per tissue volume
- Ca:
-
Calcium
- Cr:
-
Creatinine
- DPD:
-
Deoxypyridinoline
- μCT:
-
Micro-computed tomography
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide
- P:
-
Phosphorus
- SMI:
-
Structural model index
- Tb. N.:
-
Trabecular number
- Tb. Sp.:
-
Trabecular spacing
- Tb. Th.:
-
Trabecular thickness
- TRAP:
-
Tartrate-resistant acid phosphatase
References
Breitling LP (2013) Current genetics and epigenetics of smoking/tobacco-related cardiovascular disease. Arterioscler Thromb Vasc Biol 33(7):1468–1472
Hecht SS (2006) Cigarette smoking: cancer risks, carcinogens, and mechanisms. Langenbecks Arch Surg 391(6):603–613
Forey BA, Thornton AJ, Lee PN (2011) Systematic review with meta-analysis of the epidemiological evidence relating smoking to COPD, chronic bronchitis and emphysema. BMC Pulm Med 11:36
Kanis JA, Johnell O, Oden A, Johansson H, McCloskey E (2008) FRAX and the assessment of fracture probability in men and women from the UK. Osteoporos Int 19(4):385–397
Hollenbach KA, Barrett-Connor E, Edelstein SL, Holbrook T (1993) Cigarette smoking and bone mineral density in older men and women. Am J Public Health 83(9):1265–1270
Hermann AP, Brot C, Gram J, Kolthoff N, Mosekilde L (2000) Premenopausal smoking and bone density in 2015 perimenopausal women. J Bone Miner Res 15(4):780–787
Taes Y, Lapauw B, Vanbillemont G, Bogaert V, De Bacquer D, Goemaere S, Zmierczak H, Kaufman JM (2010) Early smoking is associated with peak bone mass and prevalent fractures in young, healthy men. J Bone Miner Res 25(2):379–387
Kim KH, Lee CM, Park SM, Cho B, Chang Y, Park SG, Lee K (2013) Secondhand smoke exposure and osteoporosis in never-smoking postmenopausal women: the Fourth Korea National Health and Nutrition Examination Survey. Osteoporos Int 24(2):523–532
Oberg M, Jaakkola MS, Woodward A, Peruga A, Pruss-Ustun A (2011) Worldwide burden of disease from exposure to second-hand smoke: a retrospective analysis of data from 192 countries. Lancet 377(9760):139–146
Hopper JL, Seeman E (1994) The bone density of female twins discordant for tobacco use. N Engl J Med 330(6):387–392
Balansky RM, D’Agostini F, Flora S (1999) Induction, persistence and modulation of cytogenetic alterations in cells of smoke-exposed mice. Carcinogenesis 20(8):1491–1497
Botelho FM, Nikota JK, Bauer CM, Morissette MC, Iwakura Y, Kolbeck R, Finch D, Humbles AA, Stampfli MR (2012) Cigarette smoke-induced accumulation of lung dendritic cells is interleukin-1alpha-dependent in mice. Respir Res 13:81
Marchetti F, Rowan-Carroll A, Williams A, Polyzos A, Berndt-Weis ML, Yauk CL (2011) Sidestream tobacco smoke is a male germ cell mutagen. Proc Natl Acad Sci USA 108(31):12811–12814
Kim DY, Kwon EY, Hong GU, Lee YS, Lee SH, Ro JY (2011) Cigarette smoke exacerbates mouse allergic asthma through Smad proteins expressed in mast cells. Respir Res 12:49
Ma D, Li Y, Hackfort B, Zhao Y, Xiao J, Swanson PC, Lappe J, Xiao P, Cullen D, Akhter M, Recker R, Xiao GG (2012) Smoke-induced signal molecules in bone marrow cells from altered low-density lipoprotein receptor-related protein 5 mice. J Proteome Res 11(7):3548–3560
Palmer VL, Kassmeier MD, Willcockson J, Akhter MP, Cullen DM, Swanson PC (2011) N-acetylcysteine increases the frequency of bone marrow pro-B/pre-B cells, but does not reverse cigarette smoking-induced loss of this subset. PLoS One 6(9):e24804
Schweitzer KS, Johnstone BH, Garrison J, Rush NI, Cooper S, Traktuev DO, Feng D, Adamowicz JJ, Van Demark M, Fisher AJ, Kamocki K, Brown MB, Presson RG Jr, Broxmeyer HE, March KL, Petrache I (2011) Adipose stem cell treatment in mice attenuates lung and systemic injury induced by cigarette smoking. Am J Respir Crit Care Med 183(2):215–225
Akhter MP, Lund AD, Gairola CG (2005) Bone biomechanical property deterioration due to tobacco smoke exposure. Calcif Tissue Int 77(5):319–326
Wang D, Nasto LA, Roughley P, Leme AS, Houghton AM, Usas A, Sowa G, Lee J, Niedernhofer L, Shapiro S, Kang J, Vo N (2012) Spine degeneration in a murine model of chronic human tobacco smokers. Osteoarthr Cartil 20(8):896–905
Wong HP, Li ZJ, Shin VY, Tai EK, Wu WK, Yu L, Cho CH (2009) Effects of cigarette smoking and restraint stress on human colon tumor growth in mice. Digestion 80(4):209–214
Liu ES, Ye YN, Shin VY, Yuen ST, Leung SY, Wong BC, Cho CH (2003) Cigarette smoke exposure increases ulcerative colitis-associated colonic adenoma formation in mice. Carcinogenesis 24(8):1407–1413
Chow JY, Ma L, Cho CH (1996) An experimental model for studying passive cigarette smoking effects on gastric ulceration. Life Sci 58(26):2415–2422
Adam T, Mitschke S, Streibel T, Baker RR, Zimmermann R (2006) Quantitative puff-by-puff-resolved characterization of selected toxic compounds in cigarette mainstream smoke. Chem Res Toxicol 19(4):511–520
Ye YN, Liu ES, Shin VY, Wu WK, Cho CH (2004) Contributory role of 5-lipoxygenase and its association with angiogenesis in the promotion of inflammation-associated colonic tumorigenesis by cigarette smoking. Toxicology 203(1–3):179–188
Benowitz NL, Jacob P 3rd, Kozlowski LT, Yu L (1986) Influence of smoking fewer cigarettes on exposure to tar, nicotine, and carbon monoxide. N Engl J Med 315(21):1310–1313
Haufroid V, Lison D (1998) Urinary cotinine as a tobacco-smoke exposure index: a minireview. Int Arch Occup Environ Health 71(3):162–168
Turner RT, Maran A, Lotinun S, Hefferan T, Evans GL, Zhang M, Sibonga JD (2001) Animal models for osteoporosis. Rev Endocr Metab Disord 2(1):117–127
Willinghamm MD, Brodt MD, Lee KL, Stephens AL, Ye J, Silva MJ (2010) Age-related changes in bone structure and strength in female and male BALB/c mice. Calcif Tissue Int 86(6):470–483
Bouxsein ML, Myers KS, Shultz KL, Donahue LR, Rosen CJ, Beamer WG (2005) Ovariectomy-induced bone loss varies among inbred strains of mice. J Bone Miner Res 20(7):1085–1092
Yang M, Kunugita N, Kitagawa K, Kang SH, Coles B, Kadlubar FF, Katoh T, Matsuno K, Kawamoto T (2001) Individual differences in urinary cotinine levels in Japanese smokers: relation to genetic polymorphism of drug-metabolizing enzymes. Cancer Epidemiol Biomark Prev 10(6):589–593
Liu XS, Sajda P, Saha PK, Wehrli FW, Guo XE (2006) Quantification of the roles of trabecular microarchitecture and trabecular type in determining the elastic modulus of human trabecular bone. J Bone Miner Res 21(10):1608–1617
Tamaki J, Iki M, Sato Y, Kajita E, Kagamimori S, Kagawa Y, Yoneshima H (2010) Smoking among premenopausal women is associated with increased risk of low bone status: the JPOS Study. J Bone Miner Metab 28(3):320–327
Kiel DP, Zhang Y, Hannan MT, Anderson JJ, Baron JA, Felson DT (1996) The effect of smoking at different life stages on bone mineral density in elderly men and women. Osteoporos Int 6(3):240–248
Wong PK, Christie JJ, Wark JD (2007) The effects of smoking on bone health. Clin Sci (Lond) 113(5):233–241
Landis WJ, Jacquet R (2013) Association of calcium and phosphate ions with collagen in the mineralization of vertebrate tissues. Calcif Tissue Int 93(4):329–337
Iki M, Akiba T, Matsumoto T, Nishino H, Kagamimori S, Kagawa Y, Yoneshima H (2004) Reference database of biochemical markers of bone turnover for the Japanese female population. Japanese Population-based Osteoporosis (JPOS) Study. Osteoporos Int 15(12):981–991
Gao SG, Li KH, Xu M, Jiang W, Shen H, Luo W, Xu WS, Tian J, Lei GH (2011) Bone turnover in passive smoking female rat: relationships to change in bone mineral density. BMC Musculoskelet Disord 12:131
Iwama H, Amagaya S, Ogihara Y (1987) Effect of shosaikoto, a Japanese and Chinese traditional herbal medicinal mixture, on the mitogenic activity of lipopolysaccharide: a new pharmacological testing method. J Ethnopharmacol 21(1):45–53
Liu X, Kohyama T, Kobayashi T, Abe S, Kim HJ, Reed EC, Rennard SI (2003) Cigarette smoke extract inhibits chemotaxis and collagen gel contraction mediated by human bone marrow osteoprogenitor cells and osteoblast-like cells. Osteoporos Int 14(3):235–242
Islam S, Hassan F, Tumurkhuu G, Dagvadorj J, Koide N, Naiki Y, Yoshida T, Yokochi T (2008) Receptor activator of nuclear factor-kappa B ligand induces osteoclast formation in RAW 264.7 macrophage cells via augmented production of macrophage-colony-stimulating factor. Microbiol Immunol 52(12):585–590
Phan TC, Xu J, Zheng MH (2004) Interaction between osteoblast and osteoclast: impact in bone disease. Histol Histopathol 19(4):1325–1344
Acknowledgments
We would like to thank Mr. K. M. Chan for his technical assistance and the financial support from the Hong Kong Jockey Club Osteoporosis Fund, The Chinese University of Hong Kong.
Conflict of interest
Chun Hay Ko, Ruby Lok Yi Chan, Wing Sum Siu, Wai Ting Shum, Ping Chung Leung, Lin Zhang, and Chi Hin Cho declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
All the experimental procedures had been approved by the Animal Experimentation Ethics Committee (CUHK) in accordance to the Department of Health (HKSAR) guidelines in Care and Use of Animals.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ko, C.H., Chan, R.L.Y., Siu, W.S. et al. Deteriorating Effect on Bone Metabolism and Microstructure by Passive Cigarette Smoking Through Dual Actions on Osteoblast and Osteoclast. Calcif Tissue Int 96, 389–400 (2015). https://doi.org/10.1007/s00223-015-9966-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-015-9966-8