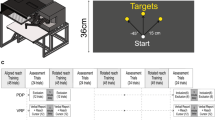
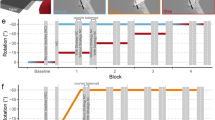
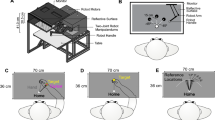
Abstract
Explicit (strategic) and implicit (unconscious) processes play a role in visuomotor adaptation (Bond and Taylor, J Neurophysiol 113:3836–3849, https://doi.org/10.1152/jn.00009.2015, 2015; Werner et al., PLoS ONE 10:1–18, https://doi.org/10.1371/journal.pone.0123321, 2015). We investigated the contributions of explicit and implicit processes to visuomotor adaptation when awareness was manipulated directly vs. indirectly, and asked how these contributions changed over time. Participants were assigned to a Strategy or No-Strategy group. Those in the Strategy group were made aware of the visuomotor distortion directly. Participants were further subdivided into groups to train with a large (60°), medium (40°) or small (20°) visuomotor distortion, providing the potential for awareness to develop indirectly. Participants reached with their respective distorted cursor, followed by a series of no-cursor reaches to assess the contributions of explicit and implicit processes to visuomotor adaptation after every 30 reach training trials. Within the no-cursor reaching trials, participants reached (1) with any strategies they had gained during training (explicit + implicit processes), and (2) as accurately to the target as possible (implicit processes). Results showed that implicit contributions were greatest in the No-Strategy group, took time to develop, and were transient, as partial decay was seen following a 5-min rest. As well, implicit contributions were similar (i.e., plateaued), regardless of the rotation size participants trained with. In contrast, explicit contributions were greatest in the Strategy group, increased with rotation size, and remained consistent over time. Taken together, results reveal that there are notable differences in the stability of explicit and implicit processes and their potential to contribute to visuomotor adaptation depending on if awareness is provided directly.




Similar content being viewed by others
References
Baraduc P, Wolpert DM (2002) Adaptation to a visuomotor shift depends on the starting posture. J Neurophysiol 88:973–981
Benson BL, Anguera JA, Seidler RD (2011) A spatial explicit strategy reduces error but interferes with sensorimotor adaptation. J Neurophysiol 105:2843–2851. https://doi.org/10.1152/jn.00002.2011
Bond KM, Taylor JA (2015) Flexible explicit but rigid implicit learning in a visuomotor adaptation task. J Neurophysiol 113:3836–3849. https://doi.org/10.1152/jn.00009.2015
Buch ER, Young S, Contreras-Vidal JL (2003) Visuomotor adaptation in normal aging. Learn Mem 10:55–63. https://doi.org/10.1101/lm.50303
Cressman EK, Henriques DYP (2009) Sensory recalibration of hand position following visuomotor adaptation. J Neurophysiol 102:3505–3518. https://doi.org/10.1152/jn.00514.2009
Day KA, Roemmich RT, Taylor JA, Bastian AJ (2016) Visuomotor learning generalizes around the intended movement. Cogn Behav. https://doi.org/10.1523/ENEURO.0005-16.2016
Flanagan JR, Nakano E, Imamizu H et al (1999) Composition and decomposition of internal models in motor learning under altered kinematic and dynamic environments. J Neurosci 19:1–5
Hagura N, Takei T, Hirose S et al (2007) Activity in the posterior parietal cortex mediates visual dominance over kinesthesia. J Neurosci 27:7047–7053
Heuer H, Hegele M (2008) Adaptation to visuomotor rotations in younger and older adults. Psychol Aging 23:190–202. https://doi.org/10.1037/0882-7974.23.1.190
Hwang EJ, Smith MA, Shadmehr R (2006) Dissociable effects of the implicit and explicit memory systems on learning control of reaching. Exp Brain Res 173:425–437. https://doi.org/10.1007/s00221-006-0391-0
Jacoby LL (1991) A process dissociation framework: separating automatic from intentional uses of memory. J Mem Lang 30:513–541
Jeannerod M (1999) To act or not to act: perspectives on the representation of actions. Q J Exp Psychol 1999:1–29
Kawato M (1999) Internal models for motor control and trajectory planning. Curr Opin Neurobiol 9:718–727
Mazzoni P, Krakauer JW (2006) An implicit plan overrides an explicit strategy during yisuomotor adaptation. J Neurosci 26:3642–3645. https://doi.org/10.1523/JNEUROSCI.5317-05.2006
McDougle SD, Bond KM, Taylor JA (2015) Explicit and implicit processes constitute the fast and slow processes of sensorimotor learning. J Neurosci 35:9568–9579. https://doi.org/10.1523/JNEUROSCI.5061-14.2015
McDougle SD, Bond KM, Taylor JA (2017) Implications of plan-based generalization in sensorimotor adaptation. J Neurophysiol 118:383–393. https://doi.org/10.1152/jn.00974.2016
Mon-Williams M, Wann JP, Jenkinson M, Rushton K (1997) Synaesthesia in the normal limb. Proc Biol Sci 264:1007–1010
Morehead JR, Taylor JA, Parvin DE, Ivry RB (2017) Characteristics of implicit sensorimotor adaptation revealed by task-irrelevant clamped feedback. J Cogn Neurosci 29:1061–1074. https://doi.org/10.1162/jocn_a_01108
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113
Redding GM, Wallace B (1996) Adaptive spatial alignment and strategic perceptual-motor control. J Exp Psychol Hum Percept Perform 22:379
Redding GM, Wallace B (2001) Calibration and alignment are separable: Evidence from prism adaptation. J Mot Behav 33:401–412. https://doi.org/10.1080/00222890109601923
Redding GM, Wallace B (2002) Strategic calibration and spatial alignment: A model from prism adaptation. J Mot Behav 34:126–138. https://doi.org/10.1080/00222890209601935
Rock I, Victor J (1964) Vision and touch: An experimentally created conflict between the two senses. Science 143:594–596
Sing GC, Smith MA (2010) Reduction in learning rates associated with anterograde interference results from interactions between different timescales in motor adaptation. Plos Comput Biol 6:1–14
Smith MA, Ghazizadeh A, Shadmehr R (2006) Interacting adaptive processes with different timescales underlie short-term motor learning. PLoS Biol 4:e179. https://doi.org/10.1371/journal.pbio.0040179
Taylor JA, Krakauer JW, Ivry RB (2014) Explicit and implicit contributions to learning in a sensorimotor adaptation task. J Neurosci 34:3023–3032. https://doi.org/10.1523/JNEUROSCI.3619-13.2014
Thomas M, Bock O (2012) Concurrent adaptation to four different visual rotations. Exp Brain Res 221:85–91. https://doi.org/10.1007/s00221-012-3150-4
Werner S, van Aken BC, Hulst T et al (2015) Awareness of sensorimotor adaptation to visual rotations of different size. PLoS ONE 10:1–18. https://doi.org/10.1371/journal.pone.0123321
Wolpert DM, Kawato M (1998) Multiple paired forward and inverse models for motor control. Neural Netw 11:1317–1329
Zbib B, Henriques DYP, Cressman EK (2016) Proprioceptive recalibration arises slowly compared to reach adaptation. Exp Brain Res 234:2201–2213. https://doi.org/10.1007/s00221-016-4624-6
Acknowledgements
This work was supported by a Discovery Grand provided by the Natural Sciences and Engineering Research Council of Canada (E. K. Cressman).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest, financial or otherwise, are declared by the author(s).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Neville, KM., Cressman, E.K. The influence of awareness on explicit and implicit contributions to visuomotor adaptation over time. Exp Brain Res 236, 2047–2059 (2018). https://doi.org/10.1007/s00221-018-5282-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-018-5282-7