Abstract
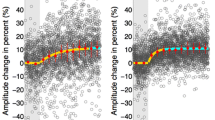
Saccadic latency is composed of separate sensory and motor processing delays. Therefore, any alteration in the sensory processing should effect the saccadic latency. Because the highest density of benzodiazepine (Bz) binding sites is located in cerebral cortex, sensory processing of stimuli in this cortical area is expected to be substantially effected by administration of Bzs. It is well known that sensory processing of binocular disparity occurs in the cerebral cortical areas and therefore the latency of saccades to stimuli defined by binocular disparity should be substantially affected by Bz intake. In this study, we tested this prediction by comparing the latency of saccadic eye movements for binocular disparity defined stimuli (stereo stimuli) with those for luminance contrast defined stimuli (luminance stimuli), after diazepam or placebo. Eye movements were mainly recorded by use of the magnetic search coil technique, and the study was performed in a randomized, double-blind way. Although diazepam prolonged the latency of saccades for stereo and luminance stimuli, the percentage increases in saccadic latency for the stereo stimuli were significantly larger than those for the luminance stimuli. Saccadic peak velocity, and saccadic amplitude, also significantly decreased after diazepam under conditions of stereo and luminance stimuli. However, there was no significant difference for either saccadic peak velocity or amplitude between the two types of target. The results suggest that the latency of saccades to binocular disparity defined random-dot stimuli could more sensitively reflect the pharmacodynamic effects of Bzs on the cerebral cortex.


Similar content being viewed by others
References
Ball DM, Glue P, Wilson S, Nutt DJ (1991) Pharmacology of saccadic eye movements in man. 1. Effects of the benzodiazepine receptor ligands midazolam and flumazenil. Psychopharmacology 105:361–367
Blackwell B (1973) Psychotropic drugs in use today. The role of diazepam in medical practice. JAMA 225:1637–1641
Braestrup C, Albrechtsen R, Squires RF (1977) High densities of benzodiazepine receptors in human cortical areas. Nature 269:702–704
Carlson MD, Penney JB Jr, Young AB (1993) NMDA, AMPA, and benzodiazepine binding site changes in Alzheimer’s disease visual cortex. Neurobiol Aging 14:343–352
Collewijn H, van der Mark F, Jansen TC (1975) Precise recording of human eye movements. Vision Res 15:447–450
DeAngelis GC (2000) Seeing in three dimensions: the neurophysiology of stereopsis. Trends Cogn Sci 4:80–90
Erkelens CJ, Collewijn H (1985a) Motion perception during dichoptic viewing of moving random-dot stereograms. Vision Res 25:583–588
Erkelens CJ, Collewijn H (1985b) Eye movements and stereopsis during dichoptic viewing of random-dot stereograms. Vision Res 25:1689–1700
Fafrowicz M, Unrug A, Marek T, van Luijtelaar G, Noworol C, Coenen A (1995) Effects of diazepam and buspirone on reaction time of saccadic eye movements. Neuropsychobiology 32:156–160
Ferraina S, Paré M, Wurtz RH (2000) Disparity sensitivity of frontal eye field neurons. J Neurophysiol 83:625–629
Fraser AD (1998) Use and abuse of the benzodiazepines. Ther Drug Monit 20:481–489
Gaymard B, Ploner CJ, Rivaud S, Vermersch AI, Pierrot-Deseilligny C (1998) Cortical control of saccades. Exp Brain Res 123:159–163
Gnadt JW, Mays LE (1995) Neurons in monkey parietal area LIP are turned for eye-movement parameters in three-dimensional space. J Neurophysiol 73:280–297
Gonzalez F, Perez R (1998) Neural mechanisms underlying stereoscopic vision. Prog Neurobiol (England) 55:191–224
Heron G, Dholakia S, Collins DE, McLaughlan H (1985) Stereoscopic threshold in children and adults. Am J Optom Physiol Opt 62:505–515
Hopfenbeck JR, Cowley DS, Radant A, Greenblatt DJ, Roy-Byrne PP (1995) Effects of diphenhydramine on human eye movements. Psychopharmacology 118:280–286
Hubel DH, Wiesel TN (1970) Stereoscopic vision in macaque monkey. Cells sensitive to binocular depth in area 18 of the macaque monkey cortex. Nature 225:41–42
Isa T, Kobayashi Y (2004) Switching between cortical and subcortical sensorimotor pathways. In: Mori S, Stuart DG, Wiesendanger M (eds) Brain Mechanisms for the Integration of Posture and Movement. Prog Brain Res, vol 143. Elsevier, Amsterdam, pp 299–305
Julesz B (1960) Binocular depth perception of computer generated patterns. Bell System Tech J 39:1125–1162
Julesz B (1971) Foundations of cyclopean perception. University of Chicago Press, Chicago
Keller EL (1974) Participation of medial pontine reticular formation in eye movement generation in monkey. J Neurophysiol 37:316–332
Leigh RJ, Zee DS (1999) The neurology of eye movements, 3rd edn. Oxford University Press, New York
Lipton L (1991) Selection devices for field-sequential stereoscopic displays: a brief history. Proc SPIE 1457:274–282
Luschei ES, Fuchs AF (1972) Activity of brain stem neurons during eye movements of alert monkeys. J Neurophysiol 35:445–461
Masson GS, Mestre DR, Martineau F, Soubrouillard C, Brefel C, Rascol O, Blin O (2000) Lorazepam-induced modifications of saccadic and smooth-pursuit eye movements in humans: attentional and motor factors. Behav Brain Res 108:169–180
Möhler H, Okada T, Heitz P, Ulrich J (1978) Biochemical identification of the site of action of benzodiazepines in human brain by 3H–diazepam binding. Life Sci 22:985–996
Munoz DP (2002) Commentary: Saccadic eye movements: overview of neural circuitry. In: Hyona J, Munoz DP, Heide W, Radach R (eds) The brain’s eye: neurobiological and clinical aspects of oculomotor research, Prog Brain Res, vol 140. Elsevier, Amsterdam, pp 89–96
Poggio GF, Motter BC, Squatrito S, Trotter Y (1985) Responses of neurons in visual cortex (V1 and V2) of the alert macaque to dynamic random-dot stereograms. Vision Res 25:397–406
Potokar J, Nash J, Sandford J, Rich A, Nutt D (2000) GABA A benzodiazepine receptor (GBzR) sensitivity: test–retest reliability in normal volunteers. Hum Psychopharmacol 15:281–286
Robinson DA (1963) A method of measuring eye movement using a scleral search coil in a magnetic field. IEEE Trans Biomed Eng 10:137–145
Robinson DL, McClurkin JW (1989) The visual superior colliculus and pulvinar. In: Wurtz RH, Goldberg ME (eds) The neurobiology of saccadic eye movements. Elsevier, Amsterdam, pp 337–360
Rothenberg SJ, Selkoe D (1981) Specific oculomotor deficit after diazepam. I. Saccadic eye movements. Psychopharmacology 74:232–236
Roy-Byrne PP, Cowley DS, Radant A, Hommer D, Greenblatt DJ (1993) Benzodiazepine pharmacodynamics: utility of eye movement measures. Psychopharmacology 110:85–91
Speth RC, Wastek GJ, Johnson PC, Yamamura HI (1978) Benzodiazepine binding in human brain: characterization using (3H) flunitrazepam. Life Sci 22:859–866
Tedeschi G, Quattrone A, Bonavita V (1986) Saccadic eye movements analysis as a measure of drug effect on central nervous system function. Ital J Neurol Sci 7:223–231
Tian J, Wei M, Liang P, Sun F (2003) Effects of diazepam on closed- and open-loop optokinetic nystagmus (OKN) in humans. Exp Brain Res 152:523–527
Tong J, Wang J, Sun F (2002) Dual-directional optokinetic nystagmus elicited by the intermittent display of gratings in primary open-angle glaucoma and normal eyes. Curr Eye Res 25:355–62
de Visser SJ, van der Post JP, de Waal PP, Cornet F, Cohen AF, van Gerven JMA (2003) Biomarkers for the effects of benzodiazepines in healthy volunteers. Br J Clin Pharmacol 55:39–50
Wei M, Sun F (1998) The alternation of optokinetic responses driven by moving stimuli in humans. Brain Res 813:406–410
Woods JH, Katz JL, Winger G (1992) Benzodiazepines: use, abuse and consequences. Pharmacol Rev 44:151–347
Yang Q, Wei M, Sun F, Tian J, Chen X, Lu C (2000) Open-loop and closed-loop optokinetic nystagmus (OKN) in myasthenia gravis and nonmyasthenic subjects. Exp Neurol 166:166–172
Yang Q, Bucci MP, Kapoula Z (2002) The latency of saccades, vergence, and combined eye movements in children and in adults. Invest Ophthalmol Vis Sci 43:2939–2949
Acknowledgements
This study was partially supported by the National Basic Research Program of China (G1999054000) and by the National Natural Science Foundation of China. The authors are grateful to Dr Saumil S. Patel for valuable comments and suggestion on the manuscript, Dr Qing Yang for help with statistical analysis of data, and Lingyu Chen, Xinzhen Zhao, and Zhi Li for help with experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, C., Tong, J. & Sun, F. Effects of diazepam on the latency of saccades for luminance and binocular disparity defined stimuli. Exp Brain Res 163, 246–251 (2005). https://doi.org/10.1007/s00221-005-2235-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-005-2235-8