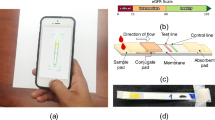
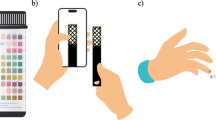
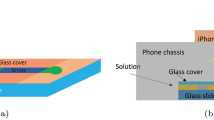
Abstract
This study presents a groundbreaking approach for the early detection of chronic kidney disease (CKD) and other urological disorders through an image-label-free, multi-dipstick identification method, eliminating the need for complex machinery, label libraries, or preset coordinates. Our research successfully identified reaction pads on 187 multi-dipsticks, each with 11 pads, leveraging machine learning algorithms trained on human urine data. This technique aims to surpass traditional colourimetric methods and concentration-colour curve fitting, offering more robust and precise community screening and home monitoring capabilities. The developed algorithms enhance the generalizability of machine learning models by extracting primary colours and correcting urine colours on each reaction pad. This method’s cost-effectiveness and portability are significant, as it requires no additional equipment beyond a standard smartphone. The system’s performance rivals professional medical equipment without auxiliary lighting or flash under regular indoor light conditions, effectively managing false positives and negatives across various categories with remarkable accuracy. In a controlled experimental setting, we found that random forest algorithms, based on a Bagging strategy and applied in the HSV colour space, showed optimal results in smartphone-assisted urinalysis. This study also introduces a novel urine colour correction method, significantly improving machine learning model performance. Additionally, ISO parameters were identified as crucial factors influencing the accuracy of smartphone-based urinalysis in the absence of additional lighting or optical configurations, highlighting the potential of this technology in low-resource settings.
Graphical Abstract





Similar content being viewed by others
References
Tummalapalli SL, Shlipak MG, Damster S, Jha V, Malik C, Levin A, et al. Availability and affordability of kidney health laboratory tests around the globe. Am J Nephrol. 2021;51(12):959–65.
Webster AC, Nagler EV, Morton RL, Masson P. Chronic kidney disease. Lancet. 2017;389(10075):1238–52.
Yamagata K, Iseki K, Nitta K, Imai H, Iino Y, Matsuo S, et al. Chronic kidney disease perspectives in Japan and the importance of urinalysis screening. Clin Exp Nephrol. 2008;12(1):1–8.
Hwang C, Lee WJ, Kim SD, Park S, Kim JH. Recent advances in biosensor technologies for point-of-care urinalysis. Biosens-Basel. 2022;12(11):1020.
Sritong N, de Medeiros MS, Basing LA, Linnes JC. Promise and perils of paper-based point-of-care nucleic acid detection for endemic and pandemic pathogens. Lab Chip. 2023;23(5):888–912.
van Delft S, Goedhart A, Spigt M, van Pinxteren B, de Wit N, Hopstaken R. Prospective, observational study comparing automated and visual point-of-care urinalysis in general practice. BMJ Open. 2016;6(8):e011230.
Yang Z, Cai G, Zhao J, Feng S. An Optical POCT device for colorimetric detection of urine test strips based on Raspberry Pi imaging. Photonics. 2022;9(10):784.
Kap Ö, Kılıç V, Hardy JG, Horzum N. Smartphone-based colorimetric detection systems for glucose monitoring in the diagnosis and management of diabetes. Analyst. 2021;146(9):2784–806.
Kumar S, Nehra M, Khurana S, Dilbaghi N, Kumar V, Kaushik A, et al. Aspects of point-of-care diagnostics for personalized health wellness. Int J Nanomedicine. 2021;16:383–402.
Abel G. Current status and future prospects of point-of-care testing around the globe. Expert Rev Mol Diagn. 2015;15(7):853–5.
Lewandrowski EL, Yeh S, Baron J, Benjamin Crocker J, Lewandrowski K. Implementation of point-of-care testing in a general internal medicine practice: a confirmation study. Clin Chim Acta Int J Clin Chem. 2017;473:71–4.
Mahoney E, Kun J, Smieja M, Fang Q. Review-point-of-care urinalysis with emerging sensing and imaging technologies. J Electrochem Soc. 2020;167(3):037518.
Lei R, Huo R, Mohan C. Current and emerging trends in point-of-care urinalysis tests. Expert Rev Mol Diagn. 2020;20(1):69–84.
Xu Z, Liu Z, Xiao M, Jiang L, Yi C. A smartphone-based quantitative point-of-care testing (POCT) system for simultaneous detection of multiple heavy metal ions. Chem Eng J. 2020;394:124966.
Xie M, Chen T, Cai Z, Lei B, Dong C. A digital microfluidic platform coupled with colorimetric loop-mediated isothermal amplification for on-site visual diagnosis of multiple diseases. Lab Chip. 2023;23:2778–88.
Kavuru V, Vu T, Karageorge L, Choudhury D, Senger R, Robertson J. Dipstick analysis of urine chemistry: benefits and limitations of dry chemistry-based assays. Postgrad Med. 2020;132(3):225–33.
Ohta S, Hiraoka R, Hiruta Y, Citterio D. Traffic light type paper-based analytical device for intuitive and semi-quantitative naked-eye signal readout. Lab Chip. 2022;22(4):717–26.
Liu G, Hu N, Ma Z, Li R. A portable analyzer based on a novel optical structure for urine dry-chemistry analysis. J Instrum. 2018;13:T07002.
Liu G, Ma Z. Study on a novel portable urine analyzer based on optical fiber bundles. Measurement. 2018;130:412–21.
Woodstock TK, Karlicek RF. RGB Color sensors for occupant detection: an alternative to PIR sensors. Ieee Sens J. 2020;20(20):12364–73.
de Carvalho OG, Machado CCS, Inacio DK, da SilveiraPetruci JF, Silva SG. RGB color sensor for colorimetric determinations: evaluation and quantitative analysis of colored liquid samples. Talanta. 2022;241:123244.
Ra M, Muhammad MS, Lim C, Han S, Jung C, Kim WY. Smartphone-based point-of-care urinalysis under variable illumination. Ieee J Transl Eng Health Med. 2018;6:2800111.
Burke AE, Thaler KM, Geva M, Adiri Y. Feasibility and acceptability of home use of a smartphone-based urine testing application among women in prenatal care. Am J Obstet Gynecol [Internet]. 2019 Nov 1;221(5):527–8, [cited 2023 Jun 1]. Available from: https://www.ajog.org/article/S0002-9378(19)30779-3/fulltext.
Balbach S, Jiang N, Moreddu R, Dong X, Kurz W, Wang C, et al. Smartphone-based colorimetric detection system for portable health tracking. Anal Methods. 2021;13(38):4361–9.
Alawsi T, Mattia GP, Al-Bawi Z, Beraldi R. Smartphone-based colorimetric sensor application for measuring biochemical material concentration. Sens Bio-Sens Res. 2021;32:100404.
Biswas SK, Chatterjee S, Laha S, Pakira V, Som NK, Saha S, et al. Instrument-free single-step direct estimation of the plasma glucose level from one drop of blood using smartphone-interfaced analytics on a paper strip. Lab Chip. 2022;22(23):4666–79.
Kim NK, Cho YS, Chil KS. Effect of illuminance on color-based analysis of diabetes-related urine fusion analytes on dipstick using a smartphone camera. J Korea Converg Soc. 2021;12(5):93–9.
Woodburn EV, Long KD, Cunningham BT, Fellow IEEE. Analysis of paper-based colorimetric assays with a smartphone spectrometer. IEEE Sens J. 2019;19(2):508–14.
Dong G, Gen L, Jia-qi M, Ya-jing S. A smartphone-based calibration-free portable urinalysis device. J Cent South Univ. 2021;28(12):3829–37.
Tong L, Hutcheson JD. A surface-based calibration approach to enable dynamic and accurate quantification of colorimetric assay systems. Anal Methods. 2021;13(37):4290–7.
Qin F, Yuan J. Research status and trend of artificial intelligence in the diagnosis of urinary diseases. J Biomed Eng. 2020;37:230–5.
Yoo WS, Kim JG, Kang K, Yoo Y. Development of static and dynamic colorimetric analysis techniques using image sensors and novel image processing software for chemical, biological and medical applications. Technologies. 2023;11(1):23.
Duan S, Cai T, Zhu J, Yang X, Lim EG, Huang K, et al. Deep learning-assisted ultra-accurate smartphone testing of paper-based colorimetric ELISA assays. Anal Chim Acta. 2023;1248:340868.
Solmaz ME, Mutlu AY, Alankus G, Kılıç V, Bayram A, Horzum N. Quantifying colorimetric tests using a smartphone app based on machine learning classifiers. Sens Actuators B Chem. 2018;255:1967–73.
Kim SC, Cho YS. Predictive system implementation to improve the accuracy of urine self-diagnosis with smartphones: application of a confusion matrix-based learning model through RGB semiquantitative analysis. Sensors. 2022;22(14):5445.
Smith GT, Dwork N, Khan SA, Millet M, Magar K, Javanmard M, et al. Robust dipstick urinalysis using a low-cost, micro-volume slipping manifold and mobile phone platform. Lab Chip. 2016;16(11):2069–78.
Yang R, Cheng W, Chen X, Qian Q, Zhang Q, Pan Y, et al. Color space transformation-based smartphone algorithm for colorimetric urinalysis. ACS Omega. 2018;3(9):12141–6.
Xiang J, Zhang Y, Cai Z, Wang W, Wang C. A 3D printed centrifugal microfluidic platform for automated colorimetric urinalysis. Microsyst Technol-Micro- Nanosyst-Inf Storage Process Syst. 2020;26(2):291–9.
Rahman MM, Uddin MJ, Hong JH, Bhuiyan NH, Shim JS. Lab-in-a-cup (LiC): an autonomous fluidic device for daily urinalysis using smartphone. Sens Actuators B-Chem. 2022;355:131336.
Tania MH, Lwin KT, Shabut AM, Najlah M, Chin J, Hossain MA. Intelligent image-based colourimetric tests using machine learning framework for lateral flow assays. Expert Syst Appl. 2020;139:112843.
Flaucher M, Nissen M, Jaeger KM, Titzmann A, Pontones C, Huebner H, et al. Smartphone-based colorimetric analysis of urine test strips for at-home prenatal care. Ieee J Transl Eng Health Med. 2022;10:2800109.
Ning Q, Zheng W, Xu H, Zhu A, Li T, Cheng Y, et al. Rapid segmentation and sensitive analysis of CRP with paper-based microfluidic device using machine learning. Anal Bioanal Chem. 2022;414(13):3959–70.
Thakur R, Maheshwari P, Datta SK, Dubey SK, Shakher C. Machine learning-based rapid diagnostic-test reader for albuminuria using smartphone. Ieee Sens J. 2021;21(13):14011–26.
Kibria IE, Ali H, Khan SA. Smartphone-based point-of-care urinalysis assessment. Annu Int Conf IEEE Eng Med Biol Soc. 2022;2022:3374–7.
Geng Z, Miao Y, Zhang G, Liang X. Colorimetric biosensor based on smartphone: state-of-art. Sens Actuators -Phys. 2023;349:114056.
Mutlu AY, Kılıç V, Özdemir GK, Bayram A, Horzum N, Solmaz ME. Smartphone-based colorimetric detection via machine learning. Analyst. 2017;142(13):2434–41.
Thakur R, Maheshwari P, Datta SK, Dubey SK. Smartphone-based, automated detection of urine albumin using deep learning approach. Measurement. 2022;194: 110948.
Kirillov A, Mintun E, Ravi N, Mao H, Rolland C, Gustafson L, et al. Segment Anything. 2023. arXiv:230402643.
Delanghe J, Speeckaert M. Preanalytical requirements of urinalysis. Biochem Medica. 2014;24(1):89–104.
Acknowledgements
The authors would like to acknowledge Yanming He and Xuanhe Zhao for figuring suggestions and Sijie Chen and Yuhao Zhang for algorithmic guidance.
Funding
The authors would like to acknowledge the financial support by Shanghai Engineering Research Centre of Interventional Medical Device (No.18DZ2250900), the financial support by National Natural Science Foundation of China (No.62373253), and the financial support from the School of Health Science and Engineering, University of Shanghai for Science and Technology.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical statement
The study was approved by the Medical Ethics Committee of Hainan Third People’s Hospital and underwent an ethical review.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, Q., Yan, R., Gui, X. et al. Machine learning-assisted image label-free smartphone platform for rapid segmentation and robust multi-urinalysis. Anal Bioanal Chem 416, 1443–1455 (2024). https://doi.org/10.1007/s00216-024-05147-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-024-05147-6