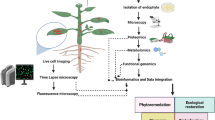
Abstract
In situ separation and visualization of synthetic and naturally occurring isomers from heterogeneous plant tissues, especially when they share similar molecular structures, are a challenging task. In this study, we combined the ion mobility separation with desorption electrospray ionization mass spectrometry imaging (DESI-IM-MSI) to achieve a direct separation and visualization of two synthetic auxin derivatives, auxinole and its structural isomer 4pTb-MeIAA, as well as endogenous auxins from Arabidopsis samples. Distinct distribution of these synthetic isomers and endogenous auxins in Arabidopsis primary roots and hypocotyls was achieved in the same imaging analysis from both individually treated and cotreated samples. We also observed putative metabolites of synthetic auxin derivatives, i.e. auxinole amino acid conjugates and hydrolysed 4pTb-MeIAA product — 4pTb-IAA, based on their unique drifting ion intensity patterns. Furthermore, DESI-IM-MSI-revealed abundance of endogenous auxins and synthetic isomers was validated by liquid chromatography–mass spectrometry (LC-MS). Our results demonstrate that DESI-IM-MSI could be used as a robust technique for detecting endogenous and exogenous isomers and provide a spatiotemporal evaluation of hormonomics profiles in plants.
Graphical abstract










Similar content being viewed by others
Data availability
The data that supports the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Davies PJ. The plant hormones: their nature, occurrence, and functions. In: Davies PJ, editor. Plant hormones: biosynthesis, signal transduction, action! Dordrecht: Springer Netherlands; 2010. p. 1-15.
Jiang K, Asami T. Chemical regulators of plant hormones and their applications in basic research and agriculture. Biosci Biotechnol Biochem. 2018;82(8):1265–300. https://doi.org/10.1080/09168451.2018.1462693.
Prusinska J, Uzunova V, Schmitzer P, Weimer M, Bell J, Napier RM. The differential binding and biological efficacy of auxin herbicides. Pest Manag Sci. 2023;79(4):1305–15. https://doi.org/10.1002/ps.7294.
Nambara E, Van Wees SCM. Plant hormone functions and interactions in biological systems. Plant J. 2021;105(2):287–9. https://doi.org/10.1111/tpj.15151.
Pokorná E, Hluska T, Galuszka P, Hallmark HT, Dobrev PI, Záveská Drábková L, et al. Cytokinin N-glucosides: occurrence, metabolism and biological activities in plants. Biomolecules. 2021;11(1):24. https://doi.org/10.3390/biom11010024.
Balarynová J, Klčová B, Tarkowská D, Turečková V, Trněný O, Špundová M, et al. Domestication has altered the ABA and gibberellin profiles in developing pea seeds. Planta. 2023;258(2):25. https://doi.org/10.1007/s00425-023-04184-2.
Hayashi K-i, Arai K, Aoi Y, Tanaka Y, Hira H, Guo R, et al. The main oxidative inactivation pathway of the plant hormone auxin. Nat Commun. 2021;12(1):6752. https://doi.org/10.1038/s41467-021-27020-1.
Brunoni F, Pěnčík A, Žukauskaitė A, Ament A, Kopečná M, Collani S, et al. Amino acid conjugation of oxIAA is a secondary metabolic regulation involved in auxin homeostasis. New Phytol. 2023;238(6):2264–70. https://doi.org/10.1111/nph.18887.
Hayashi K, Neve J, Hirose M, Kuboki A, Shimada Y, Kepinski S, et al. Rational design of an auxin antagonist of the SCF(TIR1) auxin receptor complex. ACS Chem Biol. 2012;7(3):590–8. https://doi.org/10.1021/cb200404c.
Isoda R, Yoshinari A, Ishikawa Y, Sadoine M, Simon R, Frommer WB, et al. Sensors for the quantification, localization and analysis of the dynamics of plant hormones. Plant J. 2021;105(2):542–57. https://doi.org/10.1111/tpj.15096.
Wells DM, Laplaze L, Bennett MJ, Vernoux T. Biosensors for phytohormone quantification: challenges, solutions, and opportunities. Trends Plant Sci. 2013;18(5):244–9. https://doi.org/10.1016/j.tplants.2012.12.005.
Gika HG, Theodoridis GA, Plumb RS, Wilson ID. Current practice of liquid chromatography–mass spectrometry in metabolomics and metabonomics. J Pharm Biomed Anal. 2014;87:12–25. https://doi.org/10.1016/j.jpba.2013.06.032.
Durgbanshi A, Arbona V, Pozo O, Miersch O, Sancho JV, Gómez-Cadenas A. Simultaneous determination of multiple phytohormones in plant extracts by liquid chromatography−electrospray tandem mass spectrometry. J Agric Food Chem. 2005;53(22):8437–42. https://doi.org/10.1021/jf050884b.
Skalický V, Antoniadi I, Pěnčík A, Chamrád I, Lenobel R, Kubeš MF, et al. Fluorescence-activated multi-organelle mapping of subcellular plant hormone distribution. Plant J. 2023;n/a(n/a). https://doi.org/10.1111/tpj.16456.
Široká J, Brunoni F, Pěnčík A, Mik V, Žukauskaitė A, Strnad M, et al. High-throughput interspecies profiling of acidic plant hormones using miniaturised sample processing. Plant Methods. 2022;18(1):122. https://doi.org/10.1186/s13007-022-00954-3.
Urbanová T, Tarkowská D, Novák O, Hedden P, Strnad M. Analysis of gibberellins as free acids by ultra performance liquid chromatography–tandem mass spectrometry. Talanta. 2013;112:85–94. https://doi.org/10.1016/j.talanta.2013.03.068.
Šimura J, Antoniadi I, Široká J, Tarkowská De, Strnad M, Ljung K, et al. Plant hormonomics: multiple phytohormone profiling by targeted metabolomics. Plant Physiol. 2018;177(2):476–89. https://doi.org/10.1104/pp.18.00293.
Včelařová L, Skalický V, Chamrád I, Lenobel R, Kubeš MF, Pěnčík A, et al. Auxin metabolome profiling in the Arabidopsis endoplasmic reticulum using an optimised organelle isolation protocol. Int J Mol Sci. 2021;22(17):9370. https://doi.org/10.3390/ijms22179370.
Yan Z, Maher N, Torres R, Cotto C, Hastings B, Dasgupta M, et al. Isobaric metabolite interferences and the requirement for close examination of raw data in addition to stringent chromatographic separations in liquid chromatography/tandem mass spectrometric analysis of drugs in biological matrix. Rapid Commun Mass Spectrom RCM. 2008;22(13):2021–8. https://doi.org/10.1002/rcm.3577.
Boughton BA, Thinagaran D, Sarabia D, Bacic A, Roessner U. Mass spectrometry imaging for plant biology: a review. Phytochem Rev. 2016;15(3):445–88. https://doi.org/10.1007/s11101-015-9440-2.
Bjarnholt N, Li B, D’Alvise J, Janfelt C. Mass spectrometry imaging of plant metabolites – principles and possibilities. Nat Prod Rep. 2014;31(6):818–37. https://doi.org/10.1039/C3NP70100J.
Parrot D, Papazian S, Foil D, Tasdemir D. Imaging the unimaginable: desorption electrospray ionization – imaging mass spectrometry (DESI-IMS) in natural product research. Planta Med. 2018;84(09/10):584–93. https://doi.org/10.1055/s-0044-100188.
Žukauskaitė A, Saiz-Fernández I, Bieleszová K, Iškauskienė M, Zhang C, Smýkalová I, et al. New PEO-IAA-inspired anti-auxins: synthesis, biological activity, and possible application in hemp (Cannabis sativa L.) Micropropagation. J Plant Growth Regul. 2023. https://doi.org/10.1007/s00344-023-11031-x.
Kanu AB, Dwivedi P, Tam M, Matz L, Hill HH Jr. Ion mobility–mass spectrometry. J Mass Spectrom. 2008;43(1):1–22. https://doi.org/10.1002/jms.1383.
Hou J, Zhang Z, Zhang L, Wu W, Huang Y, Jia Z, et al. Spatial lipidomics of eight edible nuts by desorption electrospray ionization with ion mobility mass spectrometry imaging. Food Chem. 2022;371: 130893. https://doi.org/10.1016/j.foodchem.2021.130893.
Hinnenkamp V, Klein J, Meckelmann SW, Balsaa P, Schmidt TC, Schmitz OJ. Comparison of CCS values determined by traveling wave ion mobility mass spectrometry and drift tube ion mobility mass spectrometry. Anal Chem. 2018;90(20):12042–50. https://doi.org/10.1021/acs.analchem.8b02711.
Pěnčík A, Casanova-Sáez R, Pilařová V, Žukauskaitė A, Pinto R, Micol JL, et al. Ultra-rapid auxin metabolite profiling for high-throughput mutant screening in Arabidopsis. J Exp Bot. 2018;69(10):2569–79. https://doi.org/10.1093/jxb/ery084.
Hladík P, Petřík I, Žukauskaitė A, Novák O, Pěnčík A. Metabolic profiles of 2-oxindole-3-acetyl-amino acid conjugates differ in various plant species. Front Plant Sci. 2023;14:1217421. https://doi.org/10.3389/fpls.2023.1217421.
Guo R, Zhou L, Chen X. Desorption electrospray ionization (DESI) source coupling ion mobility mass spectrometry for imaging fluoropezil (DC20) distribution in rat brain. Anal Bioanal Chem. 2021;413(23):5835–47. https://doi.org/10.1007/s00216-021-03563-6.
Hahsler M, Piekenbrock M, Doran D. dbscan: fast density-based clustering with R. J Stat Softw. 2019;91(1):1–30. https://doi.org/10.18637/jss.v091.i01.
Yu B, Zheng W, Persson S, Zhao Y. Protocol for analyzing root halotropism using split-agar system in Arabidopsis thaliana. STAR Protoc. 2023;4(2): 102157. https://doi.org/10.1016/j.xpro.2023.102157.
Saiz-Fernández I, Černý M, Skalák J, Brzobohatý B. Split-root systems: detailed methodology, alternative applications, and implications at leaf proteome level. Plant Methods. 2021;17:1–19. https://doi.org/10.1186/s13007-020-00706-1.
Fukui K, Arai K, Tanaka Y, Aoi Y, Kukshal V, Jez JM, et al. Chemical inhibition of the auxin inactivation pathway uncovers the roles of metabolic turnover in auxin homeostasis. Proc Natl Acad Sci. 2022;119(32):e2206869119. https://doi.org/10.1073/pnas.2206869119.
Acknowledgements
The authors are thankful to Tomáš Pospíšil for valuable scientific discussions and Alexander Muck from Analytical Professional Services Group (Waters Corporation) for technical solutions.
Funding
This work was supported by the Internal Grant Agency of Palacký University Olomouc (IGA_PrF_2023_016, IGA_PrF_2023_031).
Author information
Authors and Affiliations
Contributions
C.Z.: conceptualization; methodology; investigation; software; visualization; and writing, original draft. K.B.: investigation and formal analysis. A.Ž.: investigation; formal analysis; visualization; and writing, review and editing. P.H.: investigation and formal analysis. J.G.: resources. O.N.: funding acquisition; supervision; and writing, review and editing. K.D.: funding acquisition; supervision; and writing, review and editing.
Corresponding authors
Ethics declarations
Ethics approval
Not applicable.
Statement on animal welfare
Not applicable.
Source of biological material
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, C., Bieleszová, K., Žukauskaitė, A. et al. In situ separation and visualization of isomeric auxin derivatives in Arabidopsis by ion mobility mass spectrometry imaging. Anal Bioanal Chem 416, 125–139 (2024). https://doi.org/10.1007/s00216-023-04996-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-023-04996-x