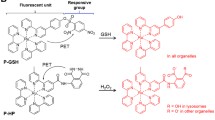
Abstract
Isoniazid (INH) and pyrazinamide (PZA) are both the first-line anti-tuberculosis drugs in clinical treatment. It is notable that there are serious side effects of the drugs along with upregulation of reactive nitrogen species, mainly including peripheral neuritis, gastrointestinal reactions, and acute drug-induced liver injury (DILI). Among them, DILI is the most common clinical symptom as well as the basic reason of treatment interruption, protocol change, and drug resistance. As vital reactive nitrogen species (RNS), peroxynitrite (ONOO-) has been demonstrated as a biomarker for evaluation and pre-diagnosis of drug-induced liver injury (DILI). In this work, we developed a red-emitting D-π-A type fluorescence probe DIC-NP which was based on 4′-hydroxy-4-biphenylcarbonitrile modified with dicyanoisophorone as a fluorescent reporter and diphenyl phosphinic chloride group as the reaction site for highly selective and sensitive sensing ONOO-. Probe DIC-NP displayed a low detection limit (14.9 nM) and 60-fold fluorescent enhancement at 669 nm in the sensing of ONOO-. Probe DIC-NP was successfully applied to monitor exogenous and endogenous ONOO- in living HeLa cells and zebrafish. Furthermore, we verified the toxicity of isoniazid (INH) and pyrazinamide (PZA) by taking the oxidative stress induced by APAP as a reference, and successfully imaged anti-tuberculosis drug-induced endogenous ONOO- in HepG2 cells. More importantly, we developed a series of mice models of liver injury and investigated the hepatotoxicity caused by the treatment of anti-tuberculosis drugs. At the same time, H&E of mice organs (heart, liver, spleen, lung, kidney) further confirmed the competence of probe DIC-NP for estimating the degree of drug-induced liver injury, which laid a solid foundation for medical research.
Graphical abstract






Similar content being viewed by others
References
Gong WP, Wu XQ. Differential diagnosis of latent tuberculosis infection and active tuberculosis: a key to a successful tuberculosis control strategy. Front Microbiol. 2021;12:745592.
Dartois VA, Rubin EJ. Anti-tuberculosis treatment strategies and drug development: challenges and priorities. Nat Rev Microbiol. 2022;20(11):685–701.
Jiang FR, Yan HD, Liang LL, Du JY, Jin SS, Yang SQ, et al. Incidence and risk factors of anti-tuberculosis drug induced liver injury (DILI): large cohort study involving 4652 Chinese adult tuberculosis patients. Liver INT. 2021;41(7):1565–75.
Wang PC, Pradhan K, Zhong XB, Ma XC. Isoniazid metabolism and hepatotoxicity. Acta Pharm Sin B. 2016;6(5):384–92.
Albanna AS, Smith BM, Cowan D, Menzies D. Fixed-dose combination antituberculosis therapy: a systematic review and meta-analysis. Eur Respir J. 2013;42(3):21–32.
Werf MJ, Langendam MW, Huitric E, Manissero D. Knowledge of tuberculosis-treatment prescription of health workers: a systematic review. Eur Respir J. 2012;39(5):1248–55.
Wu LL, Tian X, Lee DJ, Yoon JY, Lim CS, Kim HM, et al. Two-photon ESIPT-based fluorescent probe using 4-hydroxyisoindoline-1,3-dione for the detection of peroxynitrite. Chem Commun. 2021;57(84):11084–7.
Geng YJ, Wang Z, Zhou ZY, Zhu MG, Liu J, James TD. Recent progress in the development of fluorescent probes for imaging pathological oxidative stress. Chem Soc Rev. 2023;52(11):3873–926.
Guo BP, Shu W, Liu WJ, Wang H, Xing S, Chen JB, et al. Mitochondria-specific ultrasensitive ratiometric AIE probe for imaging endogenous peroxynitrite. Sens Actuators B Chem. 2021;344:130206.
Luo P, Zhao XJ. A sensitive and selective fluorescent probe for real-time detection and imaging of hypochlorous acid in living cells. ACS Omega. 2021;6(18):12287–92.
Wang MM, Wang C, Song WW, Zhong WT, Sun TM, Zhu JL, et al. A novel borate fluorescent probe for rapid selective intracellular peroxynitrite imaging. Spectrochim Acta A Mol Biomol Spectrosc. 2021;251: 119398.
Wang C, Shu W, Chen QQ, Yang CL, Su S, Gao MX, et al. A simple dual-response fluorescent probe for imaging of viscosity and ONOO- through different fluorescence signals in living cells and zebrafish. Spectrochim Acta A Mol Biomol Spectrosc. 2021;260:119990.
Xia QF, Feng SM, Hong JX, Feng GQ. One probe for multiple targets: a NIR fluorescent rhodamine-based probe for ONOO- and lysosomal pH detection in live cells. Sens Actuators B Chem. 2021;337:129732.
Chung JW, Kim HJ, Li HD, Yoon JY. Reasonably constructed NIR fluorescent probes based on dicyanoisophorone skeleton for imaging ONOO- in living cells. Dyes Pigment. 2021;195: 109665.
Wang NN, Wang H, Zhang J, Ji X, Su HH, Liu JY, et al. Endogenous peroxynitrite activated fluorescent probe for revealing anti-tuberculosis drug induced hepatotoxicity. Chin Chem Lett. 2022;33(3):1584–8.
Wang Z, Zhang F, Xiong JH, Mao ZQ, Liu ZH. Investigations of drug-induced liver injury by a peroxynitrite activatable two-photon fluorescence probe. Spectrochim Acta A Mol Biomol Spectrosc. 2021;246:118960.
Qin SC, Lu HJ, Zhang J, Ji X, Wang NN, Liu JY, et al. An activatable reporter for fluorescence imaging drug-induced liver injury in diverse cell lines and in vivo. Dyes Pigment. 2022;203:110345.
Fu ML, Wang K, Xue JY, Li Y, Bian ML, Zhu Q. A dual-response fluorescent probe for N2H4 and viscosity in living cells and zebrafish to evaluate liver injury. Org Biomol Chem. 2022;20(16):3359–64.
Liu XB, He C, Li Q, Li ZT, Liu LK, Chen S, Hou P. HClO imaging in vivo and drug-damaged liver tissues by a large stokes shift fluorescent probe. Spectrochim Acta A Mol Biomol Spectrosc. 2023;302:123081.
Wang YZ, Liu Y, Song XRY, Feng CL, Jing GL, Zhang YW, Huang W.S. Liu. Dual-targetable fluorescent probe for mapping the fluctuation of peroxynitrite in drug-induced liver injury model. Spectrochim Acta A Mol Biomol Spectrosc. 2023;286:121892.
Xu ZX, Xu ZC, Zhang D. A near infrared fluorescent probe for rapid sensing of peroxynitrite in living cells and breast cancer mice. RSC Adv. 2023;13:8262–9.
Wu LL, Liu JH, Tian X, Groleau RR, Feng BD, Yang YG, et al. Dual-channel fluorescent probe for the simultaneous monitoring of peroxynitrite and adenosine-5′-triphosphate in cellular applications. J Am Chem Soc. 2022;144(1):174–83.
Dai LX, Zhang Q, Ma QQ, Lin WY. Emerging near infrared fluorophore: dicyanoisophorone-based small-molecule fluorescent probes with large stokes shifts for bioimaging. Coord Chem Rev. 2023;489:215193.
Wang Z, Wang WW, Wang PZ, Song XJ, Mao ZQ, Liu ZH. Highly sensitive near-infrared imaging of peroxynitrite fluxes in inflammation progress. Anal Chem. 2021;93(5):3035–41.
Gong XY, Cheng D, Li W, Shen Y, Peng R, Shi L, et al. Highly selective ratiometric molecular probe for imaging peroxynitrite during drug-induced acute liver injury. J Name. 2021;9(39):8246–52.
Jin C, Wu PF, Yang YS, He ZX, Zhu HL, Li Z. A novel fluorescent probe for the detection of peroxynitrite and its application in acute liver injury model. Redox Biol. 2021;46:102068.
Su HH, Wang NN, Zhang J, Lu XY, Qin SC, Wang JM, et al. An activatable fluorescent probe for monitoring the up-regulation of peroxynitrite in drug-induced hepatotoxicity model. Dyes Pigm. 2022;203:110341.
Wu LL, Liu JH, Tian X, Groleau RR, Bull SD, Li P, et al. Fluorescent probe for the imaging of superoxide and peroxynitrite during drug-induced liver injury. Chem Sci. 2021;12(11):3921–8.
Fan XP, Ren TB, Yang W, Zhang XB, Yuan L. Activatable photoacoustic/fluorescent dual-modal probe for monitoring of drug-induced liver hypoxia in vivo. Chem Commun. 2021;57(69):8644–7.
Zhang YB, Ma DG. Selective detection of peroxynitrite in living cells by a near-infrared diphenyl phosphinate-based dicyanoisophorone probe. Spectrochim Acta A Mol Biomol Spectrosc. 2021;244:118890.
Song W, Dong B, Lu Y, Li Z, Zhang W, Lin W. Two-photon fluorescent sensors for visual detection of abnormal superoxide anion in diabetes mice. Sens Actuators B: Chem. 2021;332:129537.
Peng T, Wong NK, Chen X, Chan YK, Sun Z, Hu JJ. Molecular imaging of peroxynitrite with HKGreen-4 in live cells and tissues. J Am Chem Soc. 2014;136(33):11728–34.
Jiao S, Zhai J, Yang S, Meng X. A highly responsive, sensitive NIR fluorescent probe for imaging of superoxide anion in mitochondria of oral cancer cells. Talanta. 2021;222:121566.
He XJ, Zheng ZM, Zhang FF, Xu CC, Xu W, Ye LS, et al. Mitochondria-targeted chemosensor to discriminately and continuously visualize HClO and H2S with multiresponse fluorescence signals for in vitro and in vivo bioimaging. ACS Appl Bio Mater. 2020;3(11):7886–97.
Cui J, Zang SP, Nie HL, Shen TJ, Su S, Jing J, et al. An ICT-based fluorescent probe for ratiometric monitoring the fluctuations of peroxynitrite in mitochondria. Sens Actuators B Chem. 2021;328:129069.
Fan L, Yang QQ, Zan Q, Zhao KY, Lu WJ, Wang X, et al. Multifunctional fluorescent probe for simultaneous detection of ONOO-, viscosity, and polarity and its application in ferroptosis and cancer models. Anal Chem. 2023;95(13):5780–7.
Su HH, Wang NN, Wang JM, Wang H, Zhang J, Zhao WL. A resorufin-based red-emitting fluorescent probe with high selectivity for tracking endogenous peroxynitrite in living cells and inflammatory mice. Spectrochim Acta A Mol Biomol Spectrosc. 2021;252:119502.
Wang BD, Wei R, Gao MJ, Wang YH, Zhang CF, Guo XH, et al. Development of peroxynitrite-responsive fluorescence probe for recognition of drug-induced liver injury. Spectrochim Acta A Mol Biomol Spectrosc. 2022;283:121755.
Funding
This work received the financial support provided by the Research Project of Basic Scientific Research Operating Expenses of Provincial Colleges and Universities in Heilongjiang Province (No. 2022-KYYWF-0788).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
All animal studies were conducted in accordance with the guidelines of Ethics Care Committee of Animal Experiments of Qiqihar Medical University (QMU-AECC-2020-63).
Source of biological material
Kunming mice (20–25 g) were obtained from the Liaoning Changsheng Biotechnology Co., Ltd. of Liaoning Province, China.
Statement on animal welfare
The animal care and experimental procedures were carried out in accordance with the Guidelines of the Institutional Animal Care and Use Committee of Qiqihar Medical University of Health Guide for the Care and Use of Laboratory Animals.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, X., Ma, Y., Liu, Y. et al. Near-infrared molecular sensor for visualizing and tracking ONOO- during the process of anti-tuberculosis drug-induced liver damage. Anal Bioanal Chem 415, 7187–7196 (2023). https://doi.org/10.1007/s00216-023-04985-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-023-04985-0