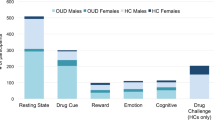
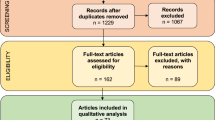
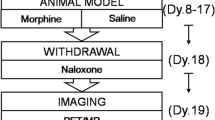
Abstract
Opioid use results in thousands of overdose deaths each year. To address this crisis, we need a better understanding of the neurobiological mechanisms that drive opioid abuse. The noninvasive imaging tools positron emission tomography (PET), functional magnetic resonance imaging (fMRI), and manganese-enhanced magnetic resonance imaging (MEMRI) can be used to identify how brain activity responds to acute opioid exposure and adapts to chronic drug treatment. These techniques can be performed in humans and animal models, and brain networks identified in animals closely map to the human brain. Animal models have the advantage of being able to systematically examine the independent effects of opioid exposure in a controlled environment accounting for the complex factors that drive opioid misuse in humans. This review synthesizes literature that utilized noninvasive neuroimaging tools (PET, fMRI, and MEMRI) measuring brain activity correlates in animals to understand the neurobiological consequences of exposure to abused opioids. A PubMed search in September 2023 identified 25 publications. These manuscripts were divided into 4 categories based on the route and duration of drug exposure (acute/chronic, active/passive administration). Within each category, the results were generally consistent across drug and imaging protocols. These papers cover a 20-year range and highlight the advancements in neuroimaging methodology during that time. These advances have enabled researchers to achieve greater resolution of brain regions altered by opioid exposure and to identify patterns of brain activation across regions (i.e., functional connectivity) and within subregions of structures. After describing the existing literature, we suggest areas where additional research is needed.





Similar content being viewed by others
Data Availability
No data was used for the research described in the article.
Abbreviations
- BOLD:
-
Blood oxygenation level-dependent
- CPP:
-
Conditioned place preference
- FDG:
-
2-Deoxy-2-[18F]fluoro-D-glucose
- fMRI:
-
Functional magnetic resonance imaging
- MEMRI:
-
Manganese-enhanced magnetic resonance imaging
- PET:
-
Positron emission tomography
- SPECT:
-
Single-photo emission computerized tomography
References
Alstrup AKO, Smith DF (2013) Anaesthesia for positron emission tomography scanning of animal brains. Lab Anim 47:12–18. https://doi.org/10.1258/la.2012.011173
Aramjoo H, Riahi-Zanjani B, Farkhondeh T et al (2021) Modulatory effect of opioid administration on the activity of cholinesterase enzyme: a systematic review of mice/rat models. Environ Sci Pollut Res Int 28:52675–52688. https://doi.org/10.1007/s11356-021-16044-1
Auvity S, Goutal S, Thézé B et al (2017) Evaluation of TSPO PET imaging, a marker of glial activation, to study the neuroimmune footprints of morphine exposure and withdrawal. Drug Alcohol Depend 170:43–50. https://doi.org/10.1016/j.drugalcdep.2016.10.037
Back SE, Lawson KM, Singleton LM, Brady KT (2011) Characteristics and correlates of men and women with prescription opioid dependence. Addict Behav 36:829–834. https://doi.org/10.1016/j.addbeh.2011.03.013
Belknap JK, Crabbe JC, Riggan J, O’Toole LA (1993) Voluntary consumption of morphine in 15 inbred mouse strains. Psychopharmacology (Berl) 112:352–358. https://doi.org/10.1007/BF02244932
Belknap JK, Riggan J, Cross S et al (1998) Genetic determinants of morphine activity and thermal responses in 15 inbred mouse strains. Pharmacol Biochem Behav 59:353–360. https://doi.org/10.1016/s0091-3057(97)00421-8
Browne CJ, Godino A, Salery M, Nestler EJ (2020) Epigenetic mechanisms of opioid addiction. Biol Psychiatry 87:22–33. https://doi.org/10.1016/j.biopsych.2019.06.027
Carmack SA, Keeley RJ, Vendruscolo JCM et al (2019) Heroin addiction engages negative emotional learning brain circuits in rats. J Clin Invest 129:2480–2484. https://doi.org/10.1172/JCI125534
Centers for Disease Control and Prevention (2022) Death Rate Maps & Graphs. https://www.cdc.gov/drugoverdose/deaths/index.html. Accessed 6 Feb 2023
Cerdá M, Santaella J, Marshall BDL et al (2015) Nonmedical prescription opioid use in childhood and early adolescence predicts transitions to heroin use in young adulthood: a national study. J Pediatr 167:605–612.e2. https://doi.org/10.1016/j.jpeds.2015.04.071
Chen Q, Hou H, Feng J et al (2018) PET imaging reveals brain metabolic changes in adolescent rats following chronic escalating morphine administration. Mol Imaging Biol 20:993–1000. https://doi.org/10.1007/s11307-018-1188-9
Cherry SR, Gambhir SS (2001) Use of positron emission tomography in animal research. ILAR J 42:219–232. https://doi.org/10.1093/ilar.42.3.219
Chow MSM, Wu SL, Webb SE et al (2017) Functional magnetic resonance imaging and the brain: a brief review. World J Radiol 9:5–9. https://doi.org/10.4329/wjr.v9.i1.5
Cosgrove KP, Mazure CM, Staley JK (2007) Evolving knowledge of sex differences in brain structure, function, and chemistry. Biol Psychiatry 62:847–855. https://doi.org/10.1016/j.biopsych.2007.03.001
Deak JD, Zhou H, Galimberti M et al (2022) Genome-wide association study in individuals of European and African ancestry and multi-trait analysis of opioid use disorder identifies 19 independent genome-wide significant risk loci. Mol Psychiatry 27:3970–3979. https://doi.org/10.1038/s41380-022-01709-1
Doyle MA, Mazei-Robison MS (2021) Opioid-Induced molecular and cellular plasticity of ventral tegmental area dopamine neurons. Cold Spring Harb Perspect Med 11:a039362. https://doi.org/10.1101/cshperspect.a039362
Drewes AM, Jensen RD, Nielsen LM et al (2013) Differences between opioids: pharmacological, experimental, clinical and economical perspectives. Br J Clin Pharmacol 75:60–78. https://doi.org/10.1111/j.1365-2125.2012.04317.x
Dvořáková L, Stenroos P, Paasonen E et al (2022) Light sedation with short habituation time for large-scale functional magnetic resonance imaging studies in rats. NMR Biomed 35:e4679. https://doi.org/10.1002/nbm.4679
Fang W (2020) Deciphering functional brain circuitry during morphine withdrawal with dynamic manganese-enhanced MRI. Neurosci Lett 716:134655. https://doi.org/10.1016/j.neulet.2019.134655
Fredriksson I, Tsai P-J, Shekara A et al (2021) Orbitofrontal cortex and dorsal striatum functional connectivity predicts incubation of opioid craving after voluntary abstinence. Proc Natl Acad Sci U S A 118:e2106624118. https://doi.org/10.1073/pnas.2106624118
Fredriksson I, Tsai P-J, Shekara A et al (2023) Role of ventral subiculum neuronal ensembles in incubation of oxycodone craving after electric barrier-induced voluntary abstinence. Sci Adv 9:eadd8687. https://doi.org/10.1126/sciadv.add8687
Gao Y-R, Ma Y, Zhang Q et al (2017) Time to wake up: Studying neurovascular coupling and brain-wide circuit function in the un-anesthetized animal. NeuroImage 153:382–398. https://doi.org/10.1016/j.neuroimage.2016.11.069
Gelernter J, Kranzler HR, Sherva R et al (2014) Genome-wide association study of opioid dependence: multiple associations mapped to calcium and potassium pathways. Biol Psychiatry 76:66–74. https://doi.org/10.1016/j.biopsych.2013.08.034
Giacomuzzi SM, Riemer Y, Ertl M et al (2005) Gender differences in health-related quality of life on admission to a maintenance treatment program. Eur Addict Res 11:69–75. https://doi.org/10.1159/000083035
Grandjean J, Desrosiers-Gregoire G, Anckaerts C et al (2023) A consensus protocol for functional connectivity analysis in the rat brain. Nat Neurosci 26:673–681. https://doi.org/10.1038/s41593-023-01286-8
Green JM, Sundman MH, Chou Y-H (2022) Opioid-induced microglia reactivity modulates opioid reward, analgesia, and behavior. Neurosci Biobehav Rev 135:104544. https://doi.org/10.1016/j.neubiorev.2022.104544
Hansen TM, Lelic D, Olesen AE et al (2018) Differential effects of oxycodone and venlafaxine on resting state functional connectivity-a randomized placebo-controlled magnetic resonance imaging study. CNS Neurosci Ther 24:820–827. https://doi.org/10.1111/cns.12827
Hernandez-Avila CA, Rounsaville BJ, Kranzler HR (2004) Opioid-, cannabis- and alcohol-dependent women show more rapid progression to substance abuse treatment. Drug Alcohol Depend 74:265–272. https://doi.org/10.1016/j.drugalcdep.2004.02.001
Ieong HF-H, Yuan Z (2017) Resting-state neuroimaging and neuropsychological findings in opioid use disorder during abstinence: a review. Front Hum Neurosci 11:169. https://doi.org/10.3389/fnhum.2017.00169
Iriah SC, Borges C, Shalev U et al (2021) The utility of maraviroc, an antiretroviral agent used to treat HIV, as treatment for opioid abuse? Data from MRI and behavioural testing in rats. J Psychiatry Neurosci JPN 46:E548–E558. https://doi.org/10.1503/jpn.200191
Iriah SC, Trivedi M, Kenkel W et al (2019) Oxycodone exposure: a magnetic resonance imaging study in response to acute and chronic oxycodone treatment in rats. Neuroscience 398:88–101. https://doi.org/10.1016/j.neuroscience.2018.11.042
Jonckers E, Shah D, Hamaide J et al (2015) The power of using functional fMRI on small rodents to study brain pharmacology and disease. Front Pharmacol 6:231. https://doi.org/10.3389/fphar.2015.00231
Kamens HM, Miller CN, Caulfield JI et al (2021) Adolescent stress reduces adult morphine-induced behavioral sensitization in C57BL/6 J mice. Front Behav Neurosci 15:678102. https://doi.org/10.3389/fnbeh.2021.678102
Kaufman MJ, Janes AC, Frederick B deB, et al (2013) A method for conducting functional MRI studies in alert nonhuman primates: initial results with opioid agonists in male cynomolgus monkeys. Exp Clin Psychopharmacol 21:323–331. https://doi.org/10.1037/a0033062
Kirkpatrick SL, Bryant CD (2015) Behavioral architecture of opioid reward and aversion in C57BL/6 substrains. Front Behav Neurosci 8:450. https://doi.org/10.3389/fnbeh.2014.00450
Koob GF, Volkow ND (2016) Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry 3:760–773. https://doi.org/10.1016/S2215-0366(16)00104-8
Kosel M, Noss RS, Hämmig R et al (2008) Cerebral blood flow effects of acute intravenous heroin administration. Eur Neuropsychopharmacol J Eur Coll Neuropsychopharmacol 18:278–285. https://doi.org/10.1016/j.euroneuro.2007.11.007
Lamanna-Rama N, MacDowell KS, López G et al (2023) Neuroimaging revealed long-lasting glucose metabolism changes to morphine withdrawal in rats pretreated with the cannabinoid agonist CP-55,940 during periadolescence. Eur Neuropsychopharmacol J Eur Coll Neuropsychopharmacol 69:60–76. https://doi.org/10.1016/j.euroneuro.2023.01.005
Lancelot S, Zimmer L (2010) Small-animal positron emission tomography as a tool for neuropharmacology. Trends Pharmacol Sci 31:411–417. https://doi.org/10.1016/j.tips.2010.06.002
Le Merrer J, Becker JAJ, Befort K, Kieffer BL (2009) Reward processing by the opioid system in the brain. Physiol Rev 89:1379–1412. https://doi.org/10.1152/physrev.00005.2009
Leppä M, Korvenoja A, Carlson S et al (2006) Acute opioid effects on human brain as revealed by functional magnetic resonance imaging. NeuroImage 31:661–669. https://doi.org/10.1016/j.neuroimage.2005.12.019
Li J, Shao D, Jiang D et al (2021) Alteration of neuroinflammation detected by 18F-GE180 PET imaging in place-conditioned rats with morphine withdrawal. EJNMMI Res 11:103. https://doi.org/10.1186/s13550-021-00849-9
Li Q, Wang Y, Zhang Y et al (2012) Craving correlates with mesolimbic responses to heroin-related cues in short-term abstinence from heroin: an event-related fMRI study. Brain Res 1469:63–72. https://doi.org/10.1016/j.brainres.2012.06.024
Liu J, Dong K, Sun Y et al (2021) Progress of brain network studies on anesthesia and consciousness: framework and clinical applications. Engineering. https://doi.org/10.1016/j.eng.2021.11.013
Lou M, Wang E, Shen Y, Wang J (2012) Cue-elicited craving in heroin addicts at different abstinent time: an fMRI pilot study. Subst Use Misuse 47:631–639. https://doi.org/10.3109/10826084.2011.646381
Lowe AS, Williams SCR, Symms MR et al (2002) Functional magnetic resonance neuroimaging of drug dependence: naloxone-precipitated morphine withdrawal. NeuroImage 17:902–910. https://doi.org/10.1006/nimg.2002.1251
Lu H, Zou Q, Gu H et al (2012) Rat brains also have a default mode network. Proc Natl Acad Sci U S A 109:3979–3984. https://doi.org/10.1073/pnas.1200506109
Luo F, Xi Z-X, Wu G et al (2004) Attenuation of brain response to heroin correlates with the reinstatement of heroin-seeking in rats by fMRI. NeuroImage 22:1328–1335. https://doi.org/10.1016/j.neuroimage.2004.03.017
Mandeville JB, Choi J-K, Jarraya B et al (2011) fMRI of cocaine self-administration in macaques reveals functional inhibition of basal ganglia. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 36:1187–1198. https://doi.org/10.1038/npp.2011.1
Mantini D, Gerits A, Nelissen K et al (2011) Default mode of brain function in monkeys. J Neurosci Off J Soc Neurosci 31:12954–12962. https://doi.org/10.1523/JNEUROSCI.2318-11.2011
Massaad CA, Pautler RG (2011) Manganese-enhanced magnetic resonance imaging (MEMRI). Methods Mol Biol Clifton NJ 711:145–174. https://doi.org/10.1007/978-1-61737-992-5_7
Mattson CL, Tanz LJ, Quinn K et al (2021) Trends and geographic patterns in drug and synthetic opioid overdose deaths — United States, 2013–2019. MMWR Morb Mortal Wkly Rep:70. https://doi.org/10.15585/mmwr.mm7006a4
McHugh RK, Devito EE, Dodd D et al (2013) Gender differences in a clinical trial for prescription opioid dependence. J Subst Abuse Treat 45:38–43. https://doi.org/10.1016/j.jsat.2012.12.007
Miech R, Johnston L, O’Malley PM et al (2015) Prescription opioids in adolescence and future opioid misuse. Pediatrics 136:e1169–e1177. https://doi.org/10.1542/peds.2015-1364
Moningka H, Lichenstein S, Worhunsky PD et al (2019) Can neuroimaging help combat the opioid epidemic? A systematic review of clinical and pharmacological challenge fMRI studies with recommendations for future research. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 44:259–273. https://doi.org/10.1038/s41386-018-0232-4
Moore K, Madularu D, Iriah S et al (2016) BOLD imaging in awake wild-type and Mu-opioid receptor knock-out mice reveals on-target activation maps in response to oxycodone. Front Neurosci 10:471. https://doi.org/10.3389/fnins.2016.00471
Muelbl MJ, Glaeser BL, Shah AS et al (2022) Repeated blast mild traumatic brain injury and oxycodone self-administration produce interactive effects on neuroimaging outcomes. Addict Biol 27:e13134. https://doi.org/10.1111/adb.13134
Nasseef MT, Singh JP, Ehrlich AT et al (2019) Oxycodone-mediated activation of the Mu opioid receptor reduces whole brain functional connectivity in mice. ACS Pharmacol Transl Sci 2:264–274. https://doi.org/10.1021/acsptsci.9b00021
Negus SS, Schrode K, Stevenson GW (2008) Micro/kappa opioid interactions in rhesus monkeys: implications for analgesia and abuse liability. Exp Clin Psychopharmacol 16:386–399. https://doi.org/10.1037/a0013088
Niu H, Zhang G, Li H et al (2017) Multi-system state shifts and cognitive deficits induced by chronic morphine during abstinence. Neurosci Lett 640:144–151. https://doi.org/10.1016/j.neulet.2016.10.057
Park TY, Nishida KS, Wilson CM et al (2017) Effects of isoflurane anesthesia and intravenous morphine self-administration on regional glucose metabolism ([18F]FDG-PET) of male Sprague-Dawley rats. Eur J Neurosci 45:922–931. https://doi.org/10.1111/ejn.13542
Reitan JA, James OF, Martucci RW (1977) Cardiovascular effects of atropine sulfate preparations in vagotomized dogs under halothane anesthesia. Anesth Analg 56:338–345. https://doi.org/10.1213/00000539-197705000-00004
Sanchez-Roige S, Fontanillas P, Jennings MV et al (2021) Genome-wide association study of problematic opioid prescription use in 132,113 23andMe research participants of European ancestry. Mol Psychiatry 26:6209–6217. https://doi.org/10.1038/s41380-021-01335-3
Scarlata MJ, Keeley RJ, Carmack SA et al (2022) Cingulate circuits are associated with escalation of heroin use and naloxone-induced increases in heroin self-administration. Addict Neurosci 1:100002. https://doi.org/10.1016/j.addicn.2021.100002
Shah YB, Haynes L, Prior MJW et al (2005) Functional magnetic resonance imaging studies of opioid receptor-mediated modulation of noxious-evoked BOLD contrast in rats. Psychopharmacology (Berl) 180:761–773. https://doi.org/10.1007/s00213-005-2214-6
Shao D, Jiang D, Huang Q et al (2023) Brain glucose metabolism and dopamine transporter changes in rats with morphine-induced conditioned place preference. Addict Biol 28:e13277. https://doi.org/10.1111/adb.13277
Soares JM, Magalhães R, Moreira PS et al (2016) A hitchhiker’s guide to functional magnetic resonance imaging. Front Neurosci 10:515. https://doi.org/10.3389/fnins.2016.00515
Soto-Montenegro ML, García-Vázquez V, Lamanna-Rama N et al (2022) Neuroimaging reveals distinct brain glucose metabolism patterns associated with morphine consumption in Lewis and Fischer 344 rat strains. Sci Rep 12:4643. https://doi.org/10.1038/s41598-022-08698-9
Stewart JL, May AC, Aupperle RL, Bodurka J (2019) Forging neuroimaging targets for recovery in opioid use disorder. Front Psychiatry 10:117. https://doi.org/10.3389/fpsyt.2019.00117
Tsai P-J, Keeley RJ, Carmack SA et al (2020) Converging structural and functional evidence for a rat salience network. Biol Psychiatry 88:867–878. https://doi.org/10.1016/j.biopsych.2020.06.023
Volpe DA, McMahon Tobin GA, Mellon RD et al (2011) Uniform assessment and ranking of opioid μ receptor binding constants for selected opioid drugs. Regul Toxicol Pharmacol RTP 59:385–390. https://doi.org/10.1016/j.yrtph.2010.12.007
Withey SL, Cao L, de Moura FB et al (2022) Fentanyl-induced changes in brain activity in awake nonhuman primates at 9.4 Tesla. Brain Imaging Behav 16:1684–1694. https://doi.org/10.1007/s11682-022-00639-4
Xi Z-X, Wu G, Stein EA, Li S-J (2002) GABAergic mechanisms of heroin-induced brain activation assessed with functional MRI. Magn Reson Med 48:838–843. https://doi.org/10.1002/mrm.10282
Xi Z-X, Wu G, Stein EA, Li S-J (2004) Opiate tolerance by heroin self-administration: an fMRI study in rat. Magn Reson Med 52:108–114. https://doi.org/10.1002/mrm.20119
Xu H, Li S-J, Bodurka J et al (2000) Heroin-induced neuronal activation in rat brain assessed by functional MRI. NeuroReport 11:1085
Yang J, Li Q (2020) Manganese-enhanced magnetic resonance imaging: application in central nervous system diseases. Front Neurol 11:143. https://doi.org/10.3389/fneur.2020.00143
Funding
This research was funded by internal support from Penn State University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kamens, H.M., Cramer, S., Hanley, R.N. et al. Neuroimaging of opioid exposure: a review of preclinical animal models to inform addiction research. Psychopharmacology 240, 2459–2482 (2023). https://doi.org/10.1007/s00213-023-06477-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-023-06477-6