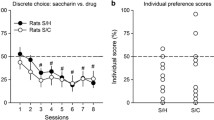
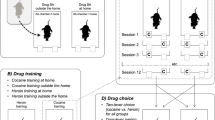
Abstract
Rationale
Cocaine use not only depends on the reinforcing properties of the drug, but also on its pharmacological effects on alternative nondrug activities. In animal models investigating choice between cocaine and alternative sweet rewards, the latter influence can have a dramatic impact on choice outcomes. When choosing under cocaine influence is prevented by imposing sufficiently long intervals between choice trials, animals typically prefer the sweet reward. However, when choosing under the drug influence is permitted, animals shift their preference in favor of cocaine.
Objectives
We previously hypothesized that this preference shift is mainly due to a direct suppression of responding for sweet reward by cocaine pharmacological effects. Here we tested this hypothesis by making rats tolerant to this drug-induced behavioral suppression.
Results
Contrary to our expectation, tolerance did not prevent rats from shifting their preference to cocaine when choosing under the influence.
Conclusion
Thus, other mechanisms must be invoked to explain the influence of cocaine intoxication on choice outcomes.






Similar content being viewed by others
References
Ahmed SH, Cador M (2006) Dissociation of psychomotor sensitization from compulsive cocaine consumption. Neuropsychopharmacology 31:563–571. https://doi.org/10.1038/sj.npp.1300834
Ahmed SH, Lin D, Koob GF, Parsons LH (2003) Escalation of cocaine self-administration does not depend on altered cocaine-induced nucleus accumbens dopamine levels. J Neurochem 86(1):102–113. https://doi.org/10.1046/j.1471-4159.2003.01833.x
American Psychiatric Association (2013) Diagnostic and Statistical Manual of Mental Disorders, 5th ed. American Psychiatric Association. https://doi.org/10.1176/appi.books.9780890425596
Augier E, Vouillac C, Ahmed SH (2012) Diazepam promotes choice of abstinence in cocaine self-administering rats. Addict Biol 17:378–391. https://doi.org/10.1111/j.1369-1600.2011.00368.x
Balopole DC, Hansult CD, Dorph D (1979) Effect of cocaine on food intake in rats. Psychopharmacology 64:121–122. https://doi.org/10.1007/BF00427356
Berridge KC, Robinson TE (2016) Liking, wanting, and the incentive-sensitization theory of addiction. Am Psychol 71:670–679. https://doi.org/10.1037/amp0000059
Bozarth MA, Wise RA (1985) Toxicity associated with long-term intravenous heroin and cocaine self-administration in the rat. JAMA J Am Med Assoc 254:81–83. https://doi.org/10.1001/jama.1985.03360010087032
Branch MN (2011) Drug addiction. Is it a disease or is it based on choice? A review of Gene Heyman’s Addiction: a disorder of choice. J Exp Anal Behav 95:263–267. https://doi.org/10.1901/jeab.2011.95-263
Cantin L, Lenoir M, Augier E, Vanhille N, Dubreucq S, Serre F, Vouillac C, Ahmed SH (2010) Cocaine is low on the value ladder of rats: possible evidence for resilience to addiction. PLoS ONE 5. https://doi.org/10.1371/journal.pone.0011592
Caprioli D, Zeric T, Thorndike EB, Venniro M (2015) Persistent palatable food preference in rats with a history of limited and extended access to methamphetamine self-administration. Addict Biol 20:913–926. https://doi.org/10.1111/adb.12220
Chow JJ, Beckmann JS (2021) Remifentanil-food choice follows predictions of relative subjective value. Drug Alcohol Depend 218:108369. https://doi.org/10.1016/j.drugalcdep.2020.108369
Cooper SJ, Francis J (1993) A microstructural analysis of the effects of presatiation on feeding behavior in the rat. Physiol Behav 53:413–416. https://doi.org/10.1016/0031-9384(93)90227-7
Cooper SJ, Jackson A, Morgan R, Carter R (1985) Evidence for opiate receptor involvement in the consumption of a high palatability diet in nondeprived rats. Neuropeptides 5:345–348
Corre J, van Zessen R, Loureiro M, Patriarchi T, Tian L, Pascoli V, Lüscher C (2018) Dopamine neurons projecting to medial shell of the nucleus accumbens drive heroin reinforcement. Elife 7:1–22. https://doi.org/10.7554/eLife.39945
de Wit H, Stewart J (1981) Reinstatement of cocaine-reinforced responding in the rat. Psychopharmacology 75:134–143. https://doi.org/10.1007/BF00432175
de Wit H, Stewart J (1983) Drug reinstatement of heroin-reinforced responding in the rat. Psychopharmacology 79:29–31. https://doi.org/10.1007/BF00433012
Ettenberg A (2004) Opponent process properties of self-administered cocaine. Neurosci Biobehav Rev 27:721–728. https://doi.org/10.1016/j.neubiorev.2003.11.009
Freese L, Durand A, Guillem K, Ahmed SH (2018) Pre-trial cocaine biases choice toward cocaine through suppression of the nondrug option. Pharmacol Biochem Behav 173:65–73. https://doi.org/10.1016/j.pbb.2018.07.010
Goldstein RZ, Volkow ND (2012) Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nat Rev Neurosci 12:652–669. https://doi.org/10.1038/nrn3119.Dysfunction
Guillem K, Ahmed SH (2018) Preference for cocaine is represented in the orbitofrontal cortex by an increased proportion of cocaine use-coding neurons. Cereb Cortex 28:819–832. https://doi.org/10.1093/cercor/bhw398
Heyman GM (2010) Addiction: a disorder of choice. Havard University Press
Heyman GM (2013) Addiction and choice: theory and new data. Front Psychiatry 4:1–5. https://doi.org/10.3389/fpsyt.2013.00031
Huynh C, Fam J, Ahmed SH, Clemens KJ (2017) Rats quit nicotine for a sweet reward following an extensive history of nicotine use. Addict Biol 22:142–151. https://doi.org/10.1111/adb.12306
Kearns DN, Kim JS, Tunstall BJ, Silberberg A (2017) Essential values of cocaine and non-drug alternatives predict the choice between them. Addict Biol 22:1501–1514. https://doi.org/10.1111/adb.12450
Kerstetter KA, Ballis MA, Duffin-Lutgen S, Carr AE, Behrens AM, Kippin TE (2012) Sex differences in selecting between food and cocaine reinforcement are mediated by estrogen. Neuropsychopharmacology 37:2605–2614. https://doi.org/10.1038/npp.2012.99
Koob G, Le Moal M (2001) Drug addiction, dysregulation of reward, and allostasis. Neuropsychopharmacology 24:97–129. https://doi.org/10.1016/S0893-133X(00)00195-0
Lenoir M, Ahmed SH (2008) Supply of a nondrug substitute reduces escalated heroin consumption. Neuropsychopharmacology 33:2272–2282. https://doi.org/10.1038/sj.npp.1301602
Lenoir M, Serre F, Cantin L, Ahmed SH (2007) Intense sweetness surpasses cocaine reward. PLoS ONE 2. https://doi.org/10.1371/journal.pone.0000698
Lenoir M, Augier E, Vouillac C, Ahmed SH (2013a) A choice-based screening method for compulsive drug users in rats. Curr Protoc Neurosci 1:1–17. https://doi.org/10.1002/0471142301.ns0944s64
Lenoir M, Cantin L, Vanhille N, Serre F, Ahmed SH (2013b) Extended heroin access increases heroin choices over a potent nondrug alternative. Neuropsychopharmacology 38:1209–1220. https://doi.org/10.1038/npp.2013.17
Madsen HB, Ahmed SH (2015) Drug versus sweet reward: greater attraction to and preference for sweet versus drug cues. Addict Biol 20:433–444. https://doi.org/10.1111/adb.12134
Müller CP (2020) Drug instrumentalization. Behav Brain Res 390:112672. https://doi.org/10.1016/j.bbr.2020.112672
Müller CP, Schumann G (2011) Drugs as instruments: a new framework for non-addictive psychoactive drug use. Behav Brain Sci 34:293–310. https://doi.org/10.1017/S0140525X11000057
Norman AB, Tsibulsky VL (2006) The compulsion zone: a pharmacological theory of acquired cocaine self-administration. Brain Res 1116:143–152. https://doi.org/10.1016/j.brainres.2006.07.092
Parker LA, Maier S, Rennie M, Crebolder J (1992) Morphine- and naltrexone-induced modification of palatability: analysis by the taste reactivity test. Behav Neurosci 106:999–1010. https://doi.org/10.1037//0735-7044.106.6.999
Pascoli V, Terrier J, Hiver A, Lüscher C (2015) Sufficiency of mesolimbic dopamine neuron stimulation for the progression to addiction. Neuron 88:1054–1066. https://doi.org/10.1016/j.neuron.2015.10.017
Pelloux Y, Baunez C (2017) Targeting the subthalamic nucleus in a preclinical model of alcohol use disorder. Psychopharmacology 234:2127–2137. https://doi.org/10.1007/s00213-017-4618-5
Pickard H (2020) What we’re not talking about when we talk about addiction. Hastings Cent Rep 50:37–46. https://doi.org/10.1002/hast.1172
Robinson TE, Berridge KC (1993) The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res Brain Res Rev 18:247–291
Russo M, Funk D, Loughlin A, Coen K, Lê AD (2018) Effects of alcohol dependence on discrete choice between alcohol and saccharin. Neuropsychopharmacology 43:1859–1866. https://doi.org/10.1038/s41386-018-0101-1
Schwartz LP, Kim JS, Silberberg A, Kearns DN (2017) Heroin and saccharin demand and preference in rats. Drug Alcohol Depend 178:87–93. https://doi.org/10.1016/j.drugalcdep.2017.04.031
Shaham Y, Shalev U, Lu L, de Wit H, Stewart J (2003) The reinstatement model of drug relapse: history, methodology and major findings. Psychopharmacology 168:3–20. https://doi.org/10.1007/s00213-002-1224-x
Thomsen M, Fink-Jensen A, Woldbye DPD, Wörtwein G, Sager TN, Holm R, Pepe LM, Barak CS (2008) Effects of acute and chronic aripiprazole treatment on choice between cocaine self-administration and food under a concurrent schedule of reinforcement in rats. Psychopharmacology 201:43–53. https://doi.org/10.1007/s00213-008-1245-1
Thomsen M, Barrett AC, Negus SS, Caine SB (2013) Cocaine versus food choice procedure in rats: environmental manipulations and effects of amphetamine. J Exp Anal Behav 99:211–233. https://doi.org/10.1002/jeab.15
Townsend EA, Schwienteck KL, Robinson HL, Lawson ST, Banks ML (2021) A drug-vs-food “choice” self-administration procedure in rats to investigate pharmacological and environmental mechanisms of substance use disorders. J Neurosci Methods 354:109110. https://doi.org/10.1016/j.jneumeth.2021.109110
Vandaele Y, Ahmed SH (2021) Choosing between cocaine and sucrose under the influence: testing the effect of cocaine tolerance. bioRxiv 2021.04.02.438165. https://doi.org/10.1101/2021.04.02.438165
Vandaele Y, Cantin L, Serre F, Vouillac-Mendoza C, Ahmed SH (2016) Choosing under the influence: a drug-specific mechanism by which the setting controls drug choices in rats. Neuropsychopharmacology 41:646–657. https://doi.org/10.1038/npp.2015.195
Venniro M, Zhang M, Caprioli D, Hoots JK, Golden SA, Heins C, Morales M, Epstein DH, Shaham Y (2018) Volitional social interaction prevents drug addiction in rat models. Nat Neurosci 21:1520–1529
Wise RA, Leeb K, Pocock D, Newton P, Burnette B, Justice JB (1995) Fluctuations in nucleus accumbens dopamine concentration during intravenous cocaine self-administration in rats. Psychopharmacology 120(1):10–20. https://doi.org/10.1007/BF02246140
Wolgin DL (2000) Contingent tolerance to amphetamine hypophagia: new insights into the role of environmental context in the expression of stereotypy. Neurosci Biobehav Rev 24:279–294. https://doi.org/10.1016/S0149-7634(99)00070-6
Wolgin DL, Hertz JM (1995) Effects of acute and chronic cocaine on milk intake, body weight, and activity in bottle- and cannula-fed rats. Behav Pharmacol 6:746–753. https://doi.org/10.1097/00008877-199511000-00010
Wolgin DL, Hughes KM (1997) Role of behavioral and pharmacological variables in the loss of tolerance to amphetamine hypophagia. Psychopharmacology 132:342–349. https://doi.org/10.1007/s002130050354
Wolgin DL, Jakubow JJ (2004) Tolerance to amphetamine hypophagia: a real-time depiction of learning to suppress stereotyped movements in the rat. Behav Neurosci 118:470–478. https://doi.org/10.1037/0735-7044.118.3.470
Woolverton WL, Kandel D, Schuster CR (1978) Tolerance and cross-tolerance to cocaine and d-amphetamine. J Pharmacol Exp Ther 205:525–535
Funding
This work was supported by the French Research Council (CNRS), the Université de Bordeaux, the French National Agency (ANR-2010-BLAN-1404–01), the Ministère de l’Enseignement Supérieur et de la Recherche (MESR), the Fondation pour la Recherche Médicale (FRM DPA20140629788), and the Peter und Traudl Engelhorn foundation.
Author information
Authors and Affiliations
Contributions
Conceptualization, SHA and YV; methodology, SHA and YV; investigation, YV; formal analysis, YV; supervision, SHA; visualization, YV; writing (original draft), YV; writing (review and editing), SHA and YV.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author comments
This manuscript has been posted as a preprint on bioRχiv: https://www.biorxiv.org/content/10.1101/2021.04.02.438165v1 (Vandaele and Ahmed 2021).
This article belongs to a Special Issue on Nature vs. Nurture in Addiction Research
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vandaele, Y., Ahmed, S.H. Choosing between cocaine and sucrose under the influence: testing the effect of cocaine tolerance. Psychopharmacology 239, 1053–1063 (2022). https://doi.org/10.1007/s00213-021-05987-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-021-05987-5