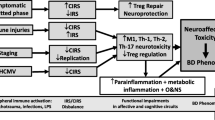
Abstract
Aim
To investigate whether circulating T cells including regulatory T cells (Treg) and derived cytokines contribute to the immune imbalance observed in schizophrenia.
Methods
Forty patients with schizophrenia and 40 age, sex, body mass index, education, and smoking status–matched healthy controls (HC) are included in the study. We stained cells with anti-CD14, anti-CD3, anti-CD4, anti-CD8, anti-CD19, anti-CD20, and anti-CD16/56. Peripheral blood mononuclear cells (PBMCs) were isolated and stained with the human FoxP3 kit containing anti-CD4/anti-CD25 and intracellular anti-Foxp3. PBMCs were cultured for 72 h and stimulated with anti-CD3/anti-CD28. Cytokines (IL-2, IL-4, IL-6, IL-10, IFN-γ, TNF-α, and IL-17A) were measured from the culture supernatant and plasma using the Th1/Th2/Th17 cytokine bead array kit.
Results
In comparison with HC, Treg percentages in schizophrenia were higher (1.17 ± 0.63 vs 0.81 ± 0.53, P = 0.005) in unstimulated but lower in the stimulated condition (0.73 ± 0.69 vs 0.97 ± 0.55, P = 0.011). Activated T cell percentages were higher in schizophrenia than HC in unstimulated (2.22 ± 0.78 vs 1.64 ± 0.89, P = 0.001) and stimulated (2.25 ± 1.01 vs 1.72 ± 1.00, P = 0.010) conditions. The culture supernatant levels of IL-6 (7505.17 ± 5170.07 vs 1787.81 ± 1363.32, P < 0.001), IL-17A (191.73 ± 212.49 vs 46.43 ± 23.99, P < 0.001), TNF-α (1557 ± 1059.69 vs 426.57 ± 174.62, P = 0.023), and IFN-γ (3204.13 ± 1397.06 vs 447.79 ± 270.13, P < 0.001); and plasma levels of IL-6 (3.83 ± 3.41vs 1.89 ± 1.14, P = 0.003) and IL-17A (1.20 ± 0.84 vs 0.83 ± 0.53, P = 0.033) were higher in patients with schizophrenia than HC.
Conclusion
Our explorative study shows reduced level of Foxp3 expressing Treg in a stimulated condition with induced levels of proinflammatory cytokines in patients with schizophrenia.

Similar content being viewed by others
References
Al-Diwani AAJ, Pollak TA, Irani SR, Lennox BR (2017) Psychosis: an autoimmune disease? Immunology 152:388–401. https://doi.org/10.1111/imm.12795
Altinoz MA, Ince B, Tek C, Srihari VH, Guloksuz S (2018) The NF-kappaB signaling pathway: an important therapeutic target in psychiatric disorders. Mol Psychiatry 23:490–491. https://doi.org/10.1038/mp.2016.207
Anderson G et al (2013) Immuno-inflammatory, oxidative and nitrosative stress, and neuroprogressive pathways in the etiology, course and treatment of schizophrenia. Prog Neuro-Psychopharmacol Biol Psychiatry 42:1–4. https://doi.org/10.1016/j.pnpbp.2012.10.008
Bender R, Lange S (2001) Adjusting for multiple testing--when and how? J Clin Epidemiol 54(4):343–349
Benros ME, Nielsen PR, Nordentoft M, Eaton WW, Dalton SO, Mortensen PB (2011) Autoimmune diseases and severe infections as risk factors for schizophrenia: a 30-year population-based register study. Am J Psychiatry 168:1303–1310. https://doi.org/10.1176/appi.ajp.2011.11030516
Borovcanin M, Jovanovic I, Radosavljevic G, Djukic Dejanovic S, Bankovic D, Arsenijevic N, Lukic ML (2012) Elevated serum level of type-2 cytokine and low IL-17 in first episode psychosis and schizophrenia in relapse. J Psychiatr Res 46:1421–1426. https://doi.org/10.1016/j.jpsychires.2012.08.016
Bowden NA, Weidenhofer J, Scott RJ, Schall U, Todd J, Michie PT, Tooney PA (2006) Preliminary investigation of gene expression profiles in peripheral blood lymphocytes in schizophrenia. Schizophr Res 82:175–183. https://doi.org/10.1016/j.schres.2005.11.012
Choi GB et al (2016) The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science (New York, NY) 351:933–939. https://doi.org/10.1126/science.aad0314
Contreras F et al (2016) Dopamine receptor D3 signaling on CD4+ T cells favors Th1- and Th17-mediated immunity. Journal of immunology (Baltimore, Md : 1950) 196:4143–4149. https://doi.org/10.4049/jimmunol.1502420
Corsi-Zuelli F et al (2019) Cytokine profile in first-episode psychosis, unaffected siblings and community-based controls: the effects of familial liability and childhood maltreatment. Psychol Med:1–9. https://doi.org/10.1017/s0033291719001016
Cosentino M, Ferrari M, Kustrimovic N, Rasini E, Marino F (2015) Influence of dopamine receptor gene polymorphisms on circulating T lymphocytes: a pilot study in healthy subjects. Hum Immunol 76:747–752. https://doi.org/10.1016/j.humimm.2015.09.032
Craddock RM, Lockstone HE, Rider DA, Wayland MT, Harris LJ, McKenna PJ, Bahn S (2007) Altered T-cell function in schizophrenia: a cellular model to investigate molecular disease mechanisms. PLoS One 2:e692. https://doi.org/10.1371/journal.pone.0000692
Debnath M, Berk M (2014) Th17 pathway–mediated immunopathogenesis of schizophrenia: mechanisms and implications. Schizophr Bull 40:1412–1421. https://doi.org/10.1093/schbul/sbu049
Dimitrov DH et al (2013) Differential correlations between inflammatory cytokines and psychopathology in veterans with schizophrenia: potential role for IL-17 pathway. Schizophr Res 151:29–35. https://doi.org/10.1016/j.schres.2013.10.019
Ding M et al (2014) Activation of Th17 cells in drug naive, first episode schizophrenia. Prog Neuro-Psychopharmacol Biol Psychiatry 51:78–82. https://doi.org/10.1016/j.pnpbp.2014.01.001
do Prado CH, Rizzo LB, Wieck A, Lopes RP, Teixeira AL, Grassi-Oliveira R, Bauer ME (2013) Reduced regulatory T cells are associated with higher levels of Th1/TH17 cytokines and activated MAPK in type 1 bipolar disorder. Psychoneuroendocrinology 38:667–676. https://doi.org/10.1016/j.psyneuen.2012.08.005
Drexhage R, Hoogenboezem T, Cohen D, Versnel M, Nolen W, van Beveren M, Drexhage H (2011) An activated set point of T-cell and monocyte inflammatory networks in recent-onset schizophrenia patients involves both pro- and anti-inflammatory forces. Int J Neuropsychoph 14:746–755. https://doi.org/10.1017/S1461145710001653
Fang X, Zhang Y, Fan W, Tang W, Zhang C (2018) Interleukin-17 alteration in first-episode psychosis: a meta-analysis. Mol Neuropsychiatry 3:135–140. https://doi.org/10.1159/000481661
Fernandez-Egea E et al (2016) Peripheral immune cell populations associated with cognitive deficits and negative symptoms of treatment-resistant schizophrenia. PLoS One 11:e0155631. https://doi.org/10.1371/journal.pone.0155631
First MB, Spitzer RL, Goibbon M, Williams JBW (1997) Structured clinical interview for DSM-IV axis I disorders (SCID-I), clinical version, Washington, DC
Frauwirth KA, Thompson CB (2002) Activation and inhibition of lymphocytes by costimulation. J Clin Invest 109:295–299. https://doi.org/10.1172/jci14941
Gendelman HE, Appel SH (2011) Neuroprotective activities of regulatory T cells. Trends Mol Med 17:687–688. https://doi.org/10.1016/j.molmed.2011.08.005
Goldsmith DR, Rapaport MH, Miller BJ (2016) A meta-analysis of blood cytokine network alterations in psychiatric patients: comparisons between schizophrenia, bipolar disorder and depression. Mol Psychiatry 21:1696–1709. https://doi.org/10.1038/mp.2016.3
Henneberg A, Riedl B, Dumke HO, Kornhuber HH (1990) T-lymphocyte subpopulations in schizophrenic patients. Eur Arch Psychiatr Neurol Sci 239:283–284
Herberth M et al (2010) Differential effects on T-cell function following exposure to serum from schizophrenia smokers. Mol Psychiatry 15:364–371. https://doi.org/10.1038/mp.2008.120
Herberth M et al (2011) Impaired glycolytic response in peripheral blood mononuclear cells of first-onset antipsychotic-naive schizophrenia patients. Mol Psychiatry 16:848–859. https://doi.org/10.1038/mp.2010.71
Himmerich H, Schonherr J, Fulda S, Sheldrick AJ, Bauer K, Sack U (2011) Impact of antipsychotics on cytokine production in-vitro. J Psychiatr Res 45:1358–1365. https://doi.org/10.1016/j.jpsychires.2011.04.009
Huang EE et al (2017) The maintenance of telomere length in CD28+ T cells during T lymphocyte stimulation. Sci Rep 7:6785. https://doi.org/10.1038/s41598-017-05174-7
Jarskog LF (2006) Apoptosis in schizophrenia: pathophysiologic and therapeutic considerations. Curr Opin Psychiatr 19:307–312. https://doi.org/10.1097/01.yco.0000218603.25346.8f
Kelly DL et al (2018) Increased circulating regulatory T cells in medicated people with schizophrenia. Psychiatry Res 269:517–523. https://doi.org/10.1016/j.psychres.2018.09.006
Koola MM (2016) Methodological issues in cytokine measurement in schizophrenia. Indian J Psychol Med 38:6–9. https://doi.org/10.4103/0253-7176.175086
Lal G et al (2009) Epigenetic regulation of Foxp3 expression in regulatory T cells by DNA methylation. Journal of immunology (Baltimore, Md : 1950) 182:259–273. https://doi.org/10.4049/jimmunol.182.1.259
Levite M, Marino F, Cosentino M (2017) Dopamine, T cells and multiple sclerosis (MS). J Neural Transm (Vienna, Austria 1996) 124:525–542. https://doi.org/10.1007/s00702-016-1640-4
Louveau A et al (2015) Structural and functional features of central nervous system lymphatic vessels. Nature 523:337–341. https://doi.org/10.1038/nature14432
Meyer U (2011) Anti-inflammatory signaling in schizophrenia. Brain Behav Immun 25:1507–1518. https://doi.org/10.1016/j.bbi.2011.05.014
Meyer U (2013) Developmental neuroinflammation and schizophrenia. Prog Neuro-Psychopharmacol Biol Psychiatry 42:20–34. https://doi.org/10.1016/j.pnpbp.2011.11.003
Miller BJ, Goldsmith DR (2017) Towards an immunophenotype of schizophrenia: progress, potential mechanisms, and future directions. Neuropsychopharmacology 42:299–317. https://doi.org/10.1038/npp.2016.211
Miller BJ, Buckley P, Seabolt W, Mellor A, Kirkpatrick B (2011) Meta-analysis of cytokine alterations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry 70:663–671. https://doi.org/10.1016/j.biopsych.2011.04.013
Miller BJ, Gassama B, Sebastian D, Buckley P, Mellor A (2013) Meta-analysis of lymphocytes in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry 73:993–999. https://doi.org/10.1016/j.biopsych.2012.09.007
Najjar S, Pearlman DM (2015) Neuroinflammation and white matter pathology in schizophrenia: systematic review. Schizophr Res 161:102–112. https://doi.org/10.1016/j.schres.2014.04.041
Noto C et al (2015) Effects of depression on the cytokine profile in drug naive first-episode psychosis. Schizophr Res 164:53–58. https://doi.org/10.1016/j.schres.2015.01.026
Oldenhove G et al (2009) Decrease of Foxp3+ Treg cell number and acquisition of effector cell phenotype during lethal infection. Immunity 31:772–786. https://doi.org/10.1016/j.immuni.2009.10.001
Osorio-Barrios F, Prado C, Contreras F, Pacheco R (2018) Dopamine receptor D5 signaling plays a dual role in experimental autoimmune encephalomyelitis potentiating Th17-mediated immunity and favoring suppressive activity of regulatory T-cells. Front Cell Neurosci 12:192. https://doi.org/10.3389/fncel.2018.00192
Prado C et al (2012) Stimulation of dopamine receptor D5 expressed on dendritic cells potentiates Th17-mediated immunity. J Immunol 188:3062–3070. https://doi.org/10.4049/jimmunol.1103096
Riedel M, Spellmann I, Schwarz MJ, Strassnig M, Sikorski C, Moller HJ, Muller N (2007) Decreased T cellular immune response in schizophrenic patients. J Psychiatr Res 41:3–7. https://doi.org/10.1016/j.jpsychires.2005.11.007
Rudolf S, Schlenke P, Broocks A, Peters M, Rothermundt M, Arolt V, Kirchner H (2004) Search for atypical lymphocytes in schizophrenia. World J Biol Psychiatr 5:33–37
Russo R, Ciminale M, Ditommaso S, Siliquini R, Zotti C, Ruggenini AM (1994) Hepatitis B vaccination in psychiatric patients. Lancet 343:356
Sakaguchi S, Yamaguchi T, Nomura T, Ono M (2008) Regulatory T cells and immune tolerance. Cell 133:775–787. https://doi.org/10.1016/j.cell.2008.05.009
Schizophrenia Working Group of the Psychiatric Genomics Consortium (2014) Biological insights from 108 schizophrenia-associated genetic loci. Nature 511:421–427. https://doi.org/10.1038/nature13595
Simsek S, Yildirim V, Cim A, Kaya S (2016) Serum IL-4 and IL-10 levels correlate with the symptoms of the drug-naive adolescents with first episode, early onset schizophrenia. J Child Adolesc Psychopharmacol 26:721–726. https://doi.org/10.1089/cap.2015.0220
Sperner-Unterweger B, Whitworth A, Kemmler G, Hilbe W, Thaler J, Weiss G, Fleischhacker WW (1999) T-cell subsets in schizophrenia: a comparison between drug-naive first episode patients and chronic schizophrenic patients. Schizophr Res 38:61–70
Steiner J et al (2010) Acute schizophrenia is accompanied by reduced T cell and increased B cell immunity. Eur Arch Psychiatry Clin Neurosci 260:509–518. https://doi.org/10.1007/s00406-010-0098-x
Sullivan PF, Fan C, Perou CM (2006) Evaluating the comparability of gene expression in blood and brain. 141b:261–268. https://doi.org/10.1002/ajmg.b.30272
Trickett A, Kwan YL (2003) T cell stimulation and expansion using anti-CD3/CD28 beads. J Immunol Methods 275:251–255. https://doi.org/10.1016/s0022-1759(03)00010-3
Vergaelen E et al (2018) A pilot study on immuno-psychiatry in the 22q11.2 deletion syndrome: a role for Th17 cells in psychosis? Brain Behav Immun 70:88–95. https://doi.org/10.1016/j.bbi.2018.03.022
Wang AK, Miller BJ (2018) Meta-analysis of cerebrospinal fluid cytokine and tryptophan catabolite alterations in psychiatric patients: comparisons between schizophrenia. Bipolar Disord Depress Schizophr Bull 44:75–83. https://doi.org/10.1093/schbul/sbx035
Wei J, Duramad O, Perng OA, Reiner SL, Liu YJ, Qin FX (2007) Antagonistic nature of T helper 1/2 developmental programs in opposing peripheral induction of Foxp3+ regulatory T cells. Proc Natl Acad Sci U S A 104:18169–18174. https://doi.org/10.1073/pnas.0703642104
Williams LM, Rudensky AY (2007) Maintenance of the Foxp3-dependent developmental program in mature regulatory T cells requires continued expression of Foxp3. Nat Immunol 8:277–284. https://doi.org/10.1038/ni1437
Yang XO et al (2008) Molecular antagonism and plasticity of regulatory and inflammatory T cell programs. Immunity 29:44–56. https://doi.org/10.1016/j.immuni.2008.05.007
Author information
Authors and Affiliations
Contributions
Cigdem Sahbaz, MD, managed the conceptualization, literature searches, statistical analyses, and designing figures and wrote the first draft of the manuscript; Tunc Akkoc, PhD, planned and managed experimental analysis; Sinan Guloksuz, MD, PhD, supervised interpreting the data and writing. All authors contributed to and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics declarations
The Medical Ethical Review Committee of the Bezmialem Vakif University approved the study, which was conducted according to the latest version of the Declaration of Helsinki. Written informed consent was obtained from all participants.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PPTX 49 kb)
Rights and permissions
About this article
Cite this article
Sahbaz, C., Zibandey, N., Kurtulmus, A. et al. Reduced regulatory T cells with increased proinflammatory response in patients with schizophrenia. Psychopharmacology 237, 1861–1871 (2020). https://doi.org/10.1007/s00213-020-05504-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05504-0