Abstract
Rationale
Glutamate receptor antagonists can improve the symptoms of Parkinson’s disease (PD) and reduce l-3,4-dihydroxyphenylalanine (l-DOPA)-induced dyskinesia (LID) in both animal models and humans, but usually produce intolerable side effects. Recent evidence suggests that upregulation of the major glutamate transporter, GLT-1, by the β-lactam antibiotic, ceftriaxone, can increase the removal of synaptic glutamate without producing noticeable side effects, and may provide an effective alternative to receptor antagonists for several neurodegenerative diseases.
Objectives
We examined whether repeated i.p. injections of ceftriaxone would, like glutamate antagonists, reduce the deficits in contralateral forepaw stepping produced by unilateral injections of 6-OHDA into the medial forebrain bundle of rats and reduce LID (as measured by abnormal involuntary movements).
Methods and results
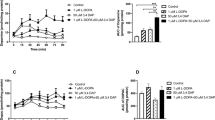
In Experiment 1, daily injections of 100 mg/kg ceftriaxone improved contralateral forepaw stepping by 44 %, and these therapeutic effects were still apparent 29 days following the cessation of treatment. In Experiment 2, daily injections of 50 mg/kg ceftriaxone were as effective as daily injections of 10 mg/kg l-DOPA in increasing contralateral forepaw stepping by 40 %. These therapeutic effects of ceftriaxone were decreased by an injection of 10 mg/kg of the selective GLT-1 antagonist, dihydrokainate (DHK), and were still evident 69 days after the cessation of ceftriaxone injections. Furthermore, ceftriaxone did not produce dyskinesia by itself and reduced the development, but not the expression, of LID.
Conclusions
These data suggest that ceftriaxone, by producing a long-term increase in GLT-1 function and increasing the removal of synaptic glutamate, may offer several advantages over l-DOPA as therapy for PD.




Similar content being viewed by others
References
Anderson CM, Swanson RA (2000) Astrocyte glutamate transport: review of properties, regulation, and physiological functions. Glia 32:1–14
Berry JD, Shefner JM, Conwit R, Schoenfeld D, Keroack M, Felsenstein D, Krivickas L, David WS, Vriesendorp F, Pestronk A, Caress JB, Katz J, Simpson E, Rosenfeld J, Pascuzzi R, Glass J, Rezania K, Rothstein JD, Greenblatt DJ, Cudkowicz ME, Northeast ALS Consortium (2013) Design and initial results of a multi-phase randomized trial of ceftriaxone in amyotrophic lateral sclerosis. PLoS One 8:e61177. doi:10.1371/journal.pone.0061177;10.1371/journal.pone.0061177
Blanchet PJ, Konitsiotis S, Chase TN (1998) Amantadine reduces levodopa-induced dyskinesias in parkinsonian monkeys. Mov Disord 13:798–802. doi:10.1002/mds.870130507
Chang JW, Wachtel SR, Young D, Kang UJ (1999) Biochemical and anatomical characterization of forepaw adjusting steps in rat models of Parkinson's disease: studies on medial forebrain bundle and striatal lesions. Neuroscience 88:617–628
Chu K, Lee ST, Sinn DI, Ko SY, Kim EH, Kim JM, Kim SJ, Park DK, Jung KH, Song EC, Lee SK, Kim M, Roh JK (2007) Pharmacological induction of ischemic tolerance by glutamate transporter-1 (EAAT2) upregulation. Stroke 38:177–182. doi:10.1161/01.STR.0000252091.36912.65
Cui C, Cui Y, Gao J, Sun L, Wang Y, Wang K, Li R, Tian Y, Song S, Cui J (2013) Neuroprotective effect of ceftriaxone in a rat model of traumatic brain injury. Neurol Sci. doi:10.1007/s10072-013-1585-4
Duty S (2012) Targeting glutamate receptors to tackle the pathogenesis, clinical symptoms and levodopa-induced dyskinesia associated with Parkinson's disease. CNS Drugs 26:1017–1032. doi:10.1007/s40263-012-0016-z;10.1007/s40263-012-0016-z
Elahi B, Phielipp N, Chen R (2012) N-Methyl-d-aspartate antagonists in levodopa induced dyskinesia: a meta-analysis. Can J Neurol Sci 39:465–472
Kelsey JE, Harris O, Cassin J (2009a) The CB1 antagonist rimonabant is adjunctively therapeutic as well as monotherapeutic in an animal model of Parkinson's disease. Behav Brain Res 203:304–307. doi:10.1016/j.bbr.2009.04.035
Kelsey JE, Langelier NA, Oriel BS, Reedy C (2009b) The effects of systemic, intrastriatal, and intrapallidal injections of caffeine and systemic injections of A2A and A1 antagonists on forepaw stepping in the unilateral 6-OHDA-lesioned rat. Psychopharmacology 201:529–539. doi:10.1007/s00213-008-1319-0
Kelsey JE, Mague SD, Pijanowski RS, Harris RC, Kleckner NW, Matthews RT (2004) NMDA receptor antagonists ameliorate the stepping deficits produced by unilateral medial forebrain bundle injections of 6-OHDA in rats. Psychopharmacology 175:179–188. doi:10.1007/s00213-004-1799-5
Kobylecki C, Cenci MA, Crossman AR, Ravenscroft P (2010) Calcium-permeable AMPA receptors are involved in the induction and expression of l-DOPA-induced dyskinesia in Parkinson's disease. J Neurochem 114:499–511. doi:10.1111/j.1471-4159.2010.06776.x;10.1111/j.1471-4159.2010.06776.x
Lamb HM, Ormrod D, Scott LJ, Figgitt DP (2002) Ceftriaxone: an update of its use in the management of community-acquired and nosocomial infections. Drugs 62:1041–1089
Leung TC, Lui CN, Chen LW, Yung WH, Chan YS, Yung KK (2012) Ceftriaxone ameliorates motor deficits and protects dopaminergic neurons in 6-hydroxydopamine-lesioned rats. ACS Chem Neurosci 3:22–30. doi:10.1021/cn200072h;10.1021/cn200072h
Löschmann PA, Lange KW, Kunow M, Rettig KJ, Jahnig P, Honore T, Turski L, Wachtel H, Jenner P, Marsden CD (1991) Synergism of the AMPA-antagonist NBQX and the NMDA-antagonist CPP with l-dopa in models of Parkinson's disease. J Neural Transm Parkinsons Dis Dementia Sect 3:203–213
Lundblad M, Andersson M, Winkler C, Kirik D, Wierup N, Cenci MA (2002) Pharmacological validation of behavioural measures of akinesia and dyskinesia in a rat model of Parkinson's disease. Eur J Neurosci 15:120–132
Miller BR, Dorner JL, Shou M, Sari Y, Barton SJ, Sengelaub DR, Kennedy RT, Rebec GV (2008) Up-regulation of GLT1 expression increases glutamate uptake and attenuates the Huntington's disease phenotype in the R6/2 mouse. Neuroscience 153:329–337. doi:10.1016/j. neuroscience .2008.02.004
Mouradian MM, Heuser IJ, Baronti F, Chase TN (1990) Modification of central dopaminergic mechanisms by continuous levodopa therapy for advanced Parkinson's disease. Ann Neurol 27:18–23. doi:10.1002/ana.410270105
Nash JE, Fox SH, Henry B, Hill MP, Peggs D, McGuire S, Maneuf Y, Hille C, Brotchie JM, Crossman AR (2000) Antiparkinsonian actions of ifenprodil in the MPTP-lesioned marmoset model of Parkinson's disease. Exp Neurol 165:136–142. doi:10.1006/exnr.2000.7444
Oh JD, Russell DS, Vaughan CL, Chase TN (1998) Enhanced tyrosine phosphorylation of striatal NMDA receptor subunits: effect of dopaminergic denervation and l-DOPA administration. Brain Res 813:150–159
Papa SM, Chase TN (1996) Levodopa-induced dyskinesias improved by a glutamate antagonist in Parkinsonian monkeys. Ann Neurol 39:574–578. doi:10.1002/ana.410390505
Paxinos G, Watson C (2006) The rat brain in stereotaxic coordinates. Academic Press, San Diego
Pines G, Danbolt NC, Bjoras M, Zhang Y, Bendahan A, Eide L, Koepsell H, Storm-Mathisen J, Seeberg E, Kanner BI (1992) Cloning and expression of a rat brain l-glutamate transporter. Nature 360:464–467. doi:10.1038/360464a0
Rabey JM, Nissipeanu P, Korczyn AD (1992) Efficacy of memantine, an NMDA receptor antagonist, in the treatment of Parkinson's disease. J Neural Transm Park Dis Dement Sect 4:277–282
Rasmussen BA, Baron DA, Kim JK, Unterwald EM, Rawls SM (2011) β-Lactam antibiotic produces a sustained reduction in extracellular glutamate in the nucleus accumbens of rats. Amino Acids 40:761–764. doi:10.1007/s00726-010-0589-0;10.1007/s00726-010-0589-0
Rebec GV (2013) Dysregulation of corticostriatal ascorbate release and glutamate uptake in transgenic models of Huntington's disease. Antioxid Redox Signal. doi:10.1089/ars.2013.5387
Rothstein JD, Patel S, Regan MR, Haenggeli C, Huang YH, Bergles DE, Jin L, Dykes Hoberg M, Vidensky S, Chung DS, Toan SV, Bruijn LI, Su ZZ, Gupta P, Fisher PB (2005) β-Lactam antibiotics offer neuroprotection by increasing glutamate transporter expression. Nature 433:73–77. doi:10.1038/nature03180
Rothstein JD, Dykes-Hoberg M, Pardo CA, Bristol LA, Jin L, Kuncl RW, Kanai Y, Hediger MA, Wang Y, Schielke JP, Welty DF (1996) Knockout of glutamate transporters reveals a major role for astroglial transport in excitotoxicity and clearance of glutamate. Neuron 16:675–686
Sgambato-Faure V, Cenci MA (2012) Glutamatergic mechanisms in the dyskinesias induced by pharmacological dopamine replacement and deep brain stimulation for the treatment of Parkinson’ disease. Prog Neuro 96:69–86. doi:10.1016/j.pneurobio.2011.10.005
Snow BJ, Macdonald L, Mcauley D, Wallis W (2000) The effect of amantadine on levodopa-induced dyskinesias in Parkinson's disease: a double-blind, placebo-controlled study. Clin Neuropharmacol 23:82–85
Tanaka K (2005) Antibiotics rescue neurons from glutamate attack. Trends Mol Med 11:259–262. doi:10.1016/j.molmed.2005.04.002
Thone-Reineke C, Neumann C, Namsolleck P, Schmerbach K, Krikov M, Schefe JH, Lucht K, Hortnagl H, Godes M, Muller S, Rumschussel K, Funke-Kaiser H, Villringer A, Steckelings UM, Unger T (2008) The β-lactam antibiotic, ceftriaxone, dramatically improves survival, increases glutamate uptake and induces neurotrophins in stroke. J Hypertens 26:2426–2435. doi:10.1097/HJH.0b013e328313e40
Verma R, Mishra V, Sasmal D, Raghubir R (2010) Pharmacological evaluation of glutamate transporter 1 (GLT-1) mediated neuroprotection following cerebral ischemia/reperfusion injury. Eur J Pharmacol 638:65–71. doi:10.1016/j.ejphar.2010.04.021
Weiss JL, Ng LK, Chase TN (1971) Long-lasting dyskinesia induced by levodopa. Lancet 1:1016–1017
Winkler C, Kirik D, Bjorklund A, Cenci MA (2002) l-DOPA-induced dyskinesia in the intrastriatal 6-hydroxydopamine model of Parkinson's disease: relation to motor and cellular parameters of nigrostriatal function. Neurobiol Dis 10:165–186
Acknowledgements
This study was based on research submitted by C. Neville as partial fulfillment for a Bachelor of Science degree in Neuroscience at Bates College. Some of these data were presented at the Society for Neuroscience Convention in New Orleans, LA in 2012. We would like to thank George Rebec for advice and encouragement during the design of this study and for comments on an earlier draft and Will Ash of the Imaging Center at Bates College for his help in preparing Fig. 1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kelsey, J.E., Neville, C. The effects of the β-lactam antibiotic, ceftriaxone, on forepaw stepping and l-DOPA-induced dyskinesia in a rodent model of Parkinson’s disease. Psychopharmacology 231, 2405–2415 (2014). https://doi.org/10.1007/s00213-013-3400-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-013-3400-6