Abstract
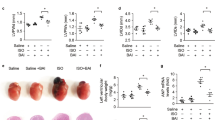
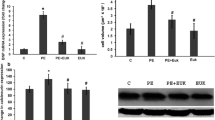
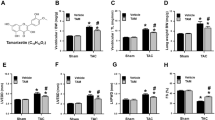
Oxidative stress, characterized by the accumulation of reactive oxygen species (ROS), is implicated in the pathogenesis of several diseases, including cardiac hypertrophy. The flavonoid quercetin is a potent ROS scavenger, with several beneficial effects for the cardiovascular system, including antihypertrophic effects. Oxidative imbalance has been implicated in cardiac hypertrophy and heart failure. In this work, we tested whether quercetin could attenuate cardiac hypertrophy by improving redox balance and mitochondrial homeostasis. To test this hypothesis, we treated a group of mice with isoproterenol (30 mg/kg/day) for 4 or 8 consecutive days. Another group received quercetin (10 mg/kg/day) from day 5th of isoproterenol treatment. We carried out the following assays in cardiac tissue: measurement of cardiac hypertrophy, protein sulfhydryl, catalase, Cu/Zn and Mn-superoxide dismutase (SOD) activity, detection of H2O2, and opening of the mitochondrial permeability transition pore. The animals treated with isoproterenol for the initial 4 days showed increased cardiac weight/tibia length ratio, decreased protein sulfhydryl content, compromised SOD and catalase activity, and high H2O2 levels. Quercetin was able to attenuate cardiac hypertrophy, restore protein sulfhydryl, and antioxidant activity, in addition to efficiently blocking the H2O2. We also observed that isoproterenol decreases mitochondrial SOD activity, while quercetin reverses it. Strikingly, quercetin also protects mitochondria against the opening of mitochondrial permeability transition pore. Taken together, these results suggest that quercetin is capable of reversing established isoproterenol-induced cardiac hypertrophy through the restoration of cellular redox balance and protecting mitochondria.





Similar content being viewed by others
References
Ballmann C, Hollinger K, Selsby JT, Amin R, Quindry JC (2015) Histological and biochemical outcomes of cardiac pathology in mdx mice with dietary quercetin enrichment. Exp Physiol 100:12–22. https://doi.org/10.1113/expphysiol.2014.083360
Boots AW, Haenen GRMM, Bast A (2008) Health effects of quercetin: from antioxidant to nutraceutical. Eur J Pharmacol 585:325–337
Chen K, Rekep M, Wei W, Wu Q, Xue Q, Li S, Tian J, Yi Q, Zhang G, Zhang G, Xiao Q, Luo J, Liu Y (2018) Quercetin prevents in vivo and in vitro myocardial hypertrophy through the proteasome-GSK-3 pathway. Cardiovasc Drugs Ther 32:5–21. https://doi.org/10.1007/s10557-018-6771-4
Dai DF, Santana LF, Vermulst M, Tomazela DM, Emond MJ, MacCoss MJ, Gollahon K, Martin GM, Loeb LA, Ladiges WC, Rabinovitch PS (2009) Overexpression of catalase targeted to mitochondria attenuates murine cardiac aging. Circulation 119:2789–2797. https://doi.org/10.1161/CIRCULATIONAHA.108.822403
David CEB, Lucas AMB, Araújo MTS, Coelho BN, Neto JBS, Portela BRC, Varela ALN, Kowaltowski AJ, Facundo HT (2018) Calorie restriction attenuates hypertrophy-induced redox imbalance and mitochondrial ATP-sensitive K+ channel repression. J Nutr Biochem 62:87–94. https://doi.org/10.1016/j.jnutbio.2018.08.008
Devienne KF, Cálgaro-Helena AF, Dorta DJ, Prado IMR, Raddi MSG, Vilegas W, Uyemura SA, Santos AC, Curti C (2007) Antioxidant activity of isocoumarins isolated from Paepalanthus bromelioides on mitochondria. Phytochemistry 68:1075–1080. https://doi.org/10.1016/j.phytochem.2007.01.014
Dorta DJ, Pigoso AA, Mingatto FE, Rodrigues T, Pestana CR, Uyemura SA, Santos AC, Curti C (2008) Antioxidant activity of flavonoids in isolated mitochondria. Phyther Res 22:1213–1218. https://doi.org/10.1002/ptr.2441
Duarte J, Pérez-Palencia R, Vargas F, Angeles Ocete M, Pérez-Vizcaino F, Zarzuelo A, Tamargo J (2001) Antihypertensive effects of the flavonoid quercetin in spontaneously hypertensive rats. Br J Pharmacol 133:117–124. https://doi.org/10.1038/sj.bjp.0704064
Frey N, Olson EN (2003) Cardiac hypertrophy: the good, the bad, and the ugly. Annu Rev Physiol 65:45–79. https://doi.org/10.1146/annurev.physiol.65.092101.142243
Han JJ, Hao J, Kim CH et al (2009) Quercetin prevents cardiac hypertrophy induced by pressure overload in rats. J Vet Med Sci 71:737–743. https://doi.org/10.1292/jvms.71.737
Izem-Meziane M, Djerdjouri B, Rimbaud S, Caffin F, Fortin D, Garnier A, Veksler V, Joubert F, Ventura-Clapier R (2012) Catecholamine-induced cardiac mitochondrial dysfunction and mPTP opening: protective effect of curcumin. Am J Physiol Heart Circ Physiol 302:H665–H674. https://doi.org/10.1152/ajpheart.00467.2011
Jalili T, Carlstrom J, Kim S, Freeman D, Jin H, Wu TC, Litwin SE, David Symons J (2006) Quercetin-supplemented diets lower blood pressure and attenuate cardiac hypertrophy in rats with aortic constriction. J Cardiovasc Pharmacol 47:531–541. https://doi.org/10.1097/01.fjc.0000211746.78454.50
Javadov S, Karmazyn M (2007) Mitochondrial permeability transition pore opening as an endpoint to initiate cell death and as a putative target for cardioprotection. Cell Physiol Biochem 20:1–22. https://doi.org/10.1159/000103747
Kwon SH, Pimentel DR, Remondino A et al (2003) H(2)O(2) regulates cardiac myocyte phenotype via concentration-dependent activation of distinct kinase pathways. J Mol Cell Cardiol 35:615–621
Liu H, Zhang L, Lu S (2012) Evaluation of antioxidant and immunity activities of quercetin in isoproterenol-treated rats. Molecules 17:4281–4291. https://doi.org/10.3390/molecules17044281
Lucas AM, Caldas FR, da Silva AP, Ventura MM, Leite IM, Filgueiras AB, Silva CGL, Kowaltowski AJ, Facundo HT (2017) Diazoxide prevents reactive oxygen species and mitochondrial damage, leading to anti-hypertrophic effects. Chem Biol Interact 261:50–55. https://doi.org/10.1016/j.cbi.2016.11.012
Marcil M, Ascah A, Matas J et al (2006) Compensated volume overload increases the vulnerability of heart mitochondria without affecting their functions in the absence of stress. J Mol Cell Cardiol 41:998–1009. https://doi.org/10.1016/j.yjmcc.2006.08.117
Metodiewa D, Jaiswal AK, Cenas N, Dickancaité E, Segura-Aguilar J (1999) Quercetin may act as a cytotoxic prooxidant after its metabolic activation to semiquinone and quinoidal product. Free Radic Biol Med 26:107–116. https://doi.org/10.1016/S0891-5849(98)00167-1
Prince PSM, Sathya B (2010) Pretreatment with quercetin ameliorates lipids, lipoproteins and marker enzymes of lipid metabolism in isoproterenol treated cardiotoxic male Wistar rats. Eur J Pharmacol 635:142–148. https://doi.org/10.1016/j.ejphar.2010.02.019
Punithavathi VR, Stanely Mainzen Prince P (2010) Protective effects of combination of quercetin and α-tocopherol on mitochondrial dysfunction and myocardial infarct size in isoproterenol-treated myocardial infarcted rats: biochemical, transmission electron microscopic, and macroscopic enzyme mapping evidences. J Biochem Mol Toxicol 24:303–312. https://doi.org/10.1002/jbt.20339
Schwarz K, Siddiqi N, Singh S, Neil CJ, Dawson DK, Frenneaux MP (2014) The breathing heart - mitochondrial respiratory chain dysfunction in cardiac disease. Int J Cardiol 171:134–143
Sestili P, Guidarelli A, Dacha M, Cantoni O (1998) Quercetin prevents DNA single strand breakage and cytotoxicity caused by tert-butylhydroperoxide: free radical scavenging versus iron chelating mechanism. Free Radic Biol Med 25:196–200. https://doi.org/10.1016/S0891-5849(98)00040-9
Shahbaz AU, Kamalov G, Zhao W, Zhao T, Johnson PL, Sun Y, Bhattacharya SK, Ahokas RA, Gerling IC, Weber KT (2011) Mitochondria-targeted cardioprotection in aldosteronism. J Cardiovasc Pharmacol 57:37–43. https://doi.org/10.1097/FJC.0b013e3181fe1250
Siwik DA, Tzortzis JD, Pimental DR, Chang DLF, Pagano PJ, Singh K, Sawyer DB, Colucci WS (1999) Inhibition of copper-zinc superoxide dismutase induces cell growth, hypertrophic phenotype, and apoptosis in neonatal rat cardiac myocytes in vitro. Circ Res 85:147–153
Stanley WC, Chandler MP (2002) Energy metabolism in the normal and failing heart: potential for therapeutic interventions. Heart Fail Rev 7:115–130
Sverdlov AL, Elezaby A, Qin F, Behring JB, Luptak I, Calamaras TD, Siwik DA, Miller EJ, Liesa M, Shirihai OS, Pimentel DR, Cohen RA, Bachschmid MM, Colucci WS (2016) Mitochondrial reactive oxygen species mediate cardiac structural, functional, and mitochondrial consequences of diet-induced metabolic heart disease. J Am Heart Assoc 5:1–13. https://doi.org/10.1161/JAHA.115.002555
Ulasova E, Perez J, Hill BG, Bradley WE, Garber DW, Landar A, Barnes S, Prasain J, Parks DA, Dell’Italia LJ, Darley-Usmar VM (2013) Quercetin prevents left ventricular hypertrophy in the Apo E knockout mouse. Redox Biol 1:381–386. https://doi.org/10.1016/j.redox.2013.07.001
Vakili BA, Okin PM, Devereux RB (2001) Prognostic implications of left ventricular hypertrophy. Am Heart J 141:334–341. https://doi.org/10.1067/mhj.2001.113218
Vercesi AE, Castilho RF, Kowaltowski AJ, de Oliveira HCF, de Souza-Pinto NC, Figueira TR, Busanello ENB (2018) Mitochondrial calcium transport and the redox nature of the calcium-induced membrane permeability transition. Free Radic Biol Med 129:1–24
Yan L, Zhang JD, Wang B, Lv YJ, Jiang H, Liu GL, Qiao Y, Ren M, Guo XF (2013) Quercetin inhibits left ventricular hypertrophy in spontaneously hypertensive rats and inhibits angiotensin II-induced H9C2 cells hypertrophy by enhancing PPAR-γ expression and suppressing AP-1 activity. PLoS One 8:e72548. https://doi.org/10.1371/journal.pone.0072548
Acknowledgment
We acknowledge the technical assistance of Antônio F. R. Santos.
Funding
Amanda Albuquerque Cabral and Yuana Ivia Ponte Viana are recipients of research scholarships from UFCA. Joana Varlla de L. Alexandre is a scholarship holder from CAPES. This research was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico (FUNCAP—grant number 88887.166577/2018-01), by the Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq to Heberty Tarso Facundo (Grant number 409489/2018-2) and by UFCA (Edital ConsolidaPG). Alicia J. Kowaltowski is supported by the Centro de Pesquisa, Inovação e Difusão de Processos Redox em Biomedicina (FAPESP 13/07937-8) and CNPq.
Author information
Authors and Affiliations
Contributions
We declare that all authors meet the criteria for authorship according to ICMJE. JVLA, YIPV, ALNV, and HTF designed methods, performed the experiments, analyzed, and interpreted the results. JVLA, YIPV, CEBD, PLOC, ACA, and HTF co-designed and worked together mitochondrial and H2O2 experiments. AJK contributed to design the work, drafting the work, and revising it critically. All authors read and approved the final version of the manuscript. All data were generated in-house and no paper mill was used.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 4757 kb)
Rights and permissions
About this article
Cite this article
de Lacerda Alexandre, J.V., Viana, Y.I.P., David, C.E.B. et al. Quercetin treatment increases H2O2 removal by restoration of endogenous antioxidant activity and blocks isoproterenol-induced cardiac hypertrophy. Naunyn-Schmiedeberg's Arch Pharmacol 394, 217–226 (2021). https://doi.org/10.1007/s00210-020-01953-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-01953-8