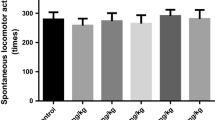
Abstract
Considering the involvement of the opioid system in major depressive disorder (MDD), mainly concerning refractory MDD, and the evidence that ascorbic acid may exert a beneficial effect for the treatment of this disorder, this study investigated the involvement of the opioid system in the antidepressant-like effect of ascorbic acid in the tail suspension test (TST). Treatment of Swiss mice with the non-selective opioid receptor antagonist naloxone (1 mg/kg, i.p.) prevented the reduced immobility time caused by ascorbic acid (1 mg/kg, p.o.) in the TST. Additionally, administration of the selective μ1-opioid receptor antagonist, naloxonazine (10 mg/kg, i.p.), also abolished the antidepressant-like action of the same dose of ascorbic acid in the TST. We also investigated the possible relationship between the opioid system and NMDA receptors in the mechanism of action of ascorbic acid or ketamine (0.1 mg/kg, i.p.) in the TST. Treatment of mice with naloxone (1 mg/kg, i.p.) blocked the synergistic antidepressant-like effect of ascorbic acid (0.1 mg/kg. p.o.) and MK-801 (0.001 mg/kg, p.o., a non-competitive NMDA receptor antagonist) in the TST. Combined administration of ketamine and MK-801 induced a synergistic antidepressant-like action, and naloxone partially abolished this effect. Our results indicate that the antidepressant-like effect of ascorbic acid in the TST appears to be dependent on the activation of the opioid system, especially μ1-opioid receptors, which might be an indirect consequence of NMDA receptor inhibition elicited by ascorbic acid administration.




Similar content being viewed by others
References
Aburawi SG, Ghambirlou FA, Attumi AA, Altubuly RA, Kara AA (2014) Effect of ascorbic acid on mental depression drug therapy: clinical study. J Psychol Psychother 4:131
Amr M, El-Mogy A, Shams T, Vieira K, Lakhan SE (2013) Efficacy of vitamin C as an adjunct to fluoxetine therapy in pediatric major depressive disorder: a randomized, double-blind, placebo-controlled pilot study. Nutr J 12(1):31. https://doi.org/10.1186/1475-2891-12-31
Berton O, Nestler EJ (2006) New approaches to antidepressant drug discovery: beyond monoamines. Nat Rev Neurosci 7(2):137–151. https://doi.org/10.1038/nrn1846
Binfare RW, Rosa AO, Lobato KR, Santos AR, Rodrigues ALS (2009) Ascorbic acid administration produces an antidepressant-like effect: evidence for the involvement of monoaminergic neurotransmission. Prog Neuro-Psychopharmacol Biol Psychiatry 33(3):530–540. https://doi.org/10.1016/j.pnpbp.2009.02.003
Bobo WV, Voort JL, Croarkin PE, Leung JG, Tye SJ, Frye MA (2016) Ketamine for treatment-resistant unipolar and bipolar major depression: critical review and implications for clinical practice. Depress Anxiety 33(8):698–710. https://doi.org/10.1002/da.22505
Bodnar RJ, Klein GE (2006) Endogenous opiates and behavior: 2005. Peptides 27(12):3391–3478. https://doi.org/10.1016/j.peptides.2006.07.011
Bourin M, Fiocco AJ, Clenet F (2001) How valuable are animal models in defining antidepressant activity? Hum Psychopharmacol 16(1):9–21. https://doi.org/10.1002/hup.178
Brocardo Pde S, Budni J, Lobato KR, Kaster MP, Rodrigues ALS (2008) Antidepressant-like effect of folic acid: involvement of NMDA receptors and L-arginine-nitric oxide-cyclic guanosine monophosphate pathway. Eur J Pharmacol 598(1–3):37–42. https://doi.org/10.1016/j.ejphar.2008.08.033
Brocardo PS, Budni J, Lobato KR, Santos AR, Rodrigues AL (2009) Evidence for the involvement of the opioid system in the antidepressant-like effect of folic acid in the mouse forced swimming test. Behav Brain Res 200(1):122–127. https://doi.org/10.1016/j.bbr.2009.01.004
Brody S (2002) High-dose ascorbic acid increases intercourse frequency and improves mood: a randomized controlled clinical trial. Biol Psychiatry 52(4):371–374. https://doi.org/10.1016/S0006-3223(02)01329-X
Bruning CA, Souza AC, Gai BM, Zeni G, Nogueira CW (2011) Antidepressant-like effect of m-trifluoromethyl-diphenyl diselenide in the mouse forced swimming test involves opioid and serotonergic systems. Eur J Pharmacol 658(2–3):145–149. https://doi.org/10.1016/j.ejphar.2011.02.039
Curtis AL, Bethea T, Valentino RJ (2006) Sexually dimorphic responses of the brain norepinephrine system to stress and corticotropin-releasing factor. Neuropsychopharmacology 31(3):544–554. https://doi.org/10.1038/sj.npp.1300875
Gautam M, Agrawal M, Gautam M, Sharma P, Gautam AS, Gautam S (2012) Role of antioxidants in generalised anxiety disorder and depression. Indian J Psychiatry 54(3):244–247. https://doi.org/10.4103/0019-5545.102424
Harrison FE, May JM (2009) Vitamin C function in the brain: vital role of the ascorbate transporter SVCT2. Free Radic Biol Med 46(6):719–730. https://doi.org/10.1016/j.freeradbiomed.2008.12.018
Hsu DT, Sanford BJ, Meyers KK, Love TM, Hazlett KE, Walker SJ, Mickey BJ, Koeppe RA, Langenecker SA, Zubieta JK (2015) It still hurts: altered endogenous opioid activity in the brain during social rejection and acceptance in major depressive disorder. Mol Psychiatry 20(2):193–200. https://doi.org/10.1038/mp.2014.185
Kaster MP, Budni J, Santos AR, Rodrigues AL (2007) Pharmacological evidence for the involvement of the opioid system in the antidepressant-like effect of adenosine in the mouse forced swimming test. Eur J Pharmacol 576(1–3):91–98. https://doi.org/10.1016/j.ejphar.2007.08.026
Kaster MP, Machado DG, Santos AR, Rodrigues AL (2012) Involvement of NMDA receptors in the antidepressant-like action of adenosine. Pharmacol Rep 64(3):706–713. https://doi.org/10.1016/S1734-1140(12)70865-4
Khanzode SD, Dakhale GN, Khanzode SS, Saoji A, Palasodkar R (2003) Oxidative damage and major depression: the potential antioxidant action of selective serotonin re-uptake inhibitors. Redox Rep 8(6):365–370. https://doi.org/10.1179/135100003225003393
Le Mevel JC, Abitbol S, Beraud G, Maniey J (1979) Temporal changes in plasma adrenocorticotropin concentration after repeated neurotropic stress in male and female rats. Endocrinology 105(3):812–817. https://doi.org/10.1210/endo-105-3-812
Mansour A, Khachaturian H, Lewis ME, Akil H, Watson SJ (1988) Anatomy of CNS opioid receptors. Trends Neurosci 11(7):308–314. https://doi.org/10.1016/0166-2236(88)90093-8
Mao J (1999) NMDA and opioid receptors: their interactions in antinociception, tolerance and neuroplasticity. Brain research. Brain Res Rev 30(3):289–304. https://doi.org/10.1016/S0165-0173(99)00020-X
Maze M, Fujinaga M (2000) Recent advances in understanding the actions and toxicity of nitrous oxide. Anaesthesia 55(4):311–314. https://doi.org/10.1046/j.1365-2044.2000.01463.x
Moretti M, Budni J, Dos Santos DB, Antunes A, Daufenbach JF, Manosso LM, Farina M, Rodrigues ALS (2013) Protective effects of ascorbic acid on behavior and oxidative status of restraint-stressed mice. J Mol Neurosci 49(1):68–79. https://doi.org/10.1007/s12031-012-9892-4
Moretti M, Budni J, Freitas AE, Rosa PB, Rodrigues ALS (2014) Antidepressant-like effect of ascorbic acid is associated with the modulation of mammalian target of rapamycin pathway. J Psychiatr Res 48(1):16–24. https://doi.org/10.1016/j.jpsychires.2013.10.014
Moretti M, Budni J, Freitas AE, Neis VB, Ribeiro CM, de Oliveira Balen G, Rieger DK, Leal RB, Rodrigues ALS (2015) TNF-α-induced depressive-like phenotype and p38(MAPK) activation are abolished by ascorbic acid treatment. Eur Neuropsychopharmacol 25(6):902–912. https://doi.org/10.1016/j.euroneuro.2015.03.006
Moretti M, Budni J, Ribeiro CM, Rieger DK, Leal RB, Rodrigues ALS (2016) Subchronic administration of ascorbic acid elicits antidepressant-like effect and modulates cell survival signaling pathways in mice. J Nutr Biochem 38:50–56. https://doi.org/10.1016/j.jnutbio.2016.09.004
Moretti M, Budni J, Ribeiro CM, Rodrigues ALS (2012a) Involvement of different types of potassium channels in the antidepressant-like effect of ascorbic acid in the mouse tail suspension test. Eur J Pharmacol 687(1–3):21–27. https://doi.org/10.1016/j.ejphar.2012.04.041
Moretti M, Colla A, de Oliveira Balen G, dos Santos DB, Budni J, de Freitas AE, Farina M, Rodrigues ALS (2012b) Ascorbic acid treatment, similarly to fluoxetine, reverses depressive-like behavior and brain oxidative damage induced by chronic unpredictable stress. J Psychiatr Res 46(3):331–340. https://doi.org/10.1016/j.jpsychires.2011.11.009
Moretti M, Freitas AE, Budni J, Fernandes SC, Balen Gde O, Rodrigues ALS (2011) Involvement of nitric oxide-cGMP pathway in the antidepressant-like effect of ascorbic acid in the tail suspension test. Behav Brain Res 225(1):328–333. https://doi.org/10.1016/j.bbr.2011.07.024
Neis VB, Manosso LM, Moretti M, Freitas AE, Daufenbach J, Rodrigues ALS (2014) Depressive-like behavior induced by tumor necrosis factor-alpha is abolished by agmatine administration. Behav Brain Res 261:336–344. https://doi.org/10.1016/j.bbr.2013.12.038
Neis VB, Moretti M, Manosso LM, Lopes MW, Leal RB, Rodrigues ALS (2015) Agmatine enhances antidepressant potency of MK-801 and conventional antidepressants in mice. Pharmacol Biochem Behav 130:9–14. https://doi.org/10.1016/j.pbb.2014.12.009
Nestler EJ, Carlezon WA Jr (2006) The mesolimbic dopamine reward circuit in depression. Biol Psychiatry 59(12):1151–1159. https://doi.org/10.1016/j.biopsych.2005.09.018
Pickar D, Cutler NR, Naber D, Post RM, Pert CB, Bunney WE Jr (1980) Plasma opioid activity in manic-depressive illness. Lancet 1(8174):937
Rebec GV, Pierce RC (1994) A vitamin as neuromodulator—ascorbate release into the extracellular fluid of the brain regulates dopaminergic and glutamatergic transmission. Prog Neurobiol 43(6):537–565. https://doi.org/10.1016/0301-0082(94)90052-3
Rice ME (2000) Ascorbate regulation and its neuroprotective role in the brain. Trends Neurosci 23(5):209–216. https://doi.org/10.1016/S0166-2236(99)01543-X
Rosa PB, Neis VB, Ribeiro CM, Moretti M, Rodrigues ALS (2016) Antidepressant-like effects of ascorbic acid and ketamine involve modulation of GABAA and GABAB receptors. Pharmacol Rep 68(5):996–1001. https://doi.org/10.1016/j.pharep.2016.05.010
Sahraian A, Ghanizadeh A, Kazemeini F (2015) Vitamin C as an adjuvant for treating major depressive disorder and suicidal behavior, a randomized placebo-controlled clinical trial. Trials 16(1):94–102. https://doi.org/10.1186/s13063-015-0609-1
Schreiber S, Bleich A, Pick CG (2002) Venlafaxine and mirtazapine: different mechanisms of antidepressant action, common opioid-mediated antinociceptive effects—a possible opioid involvement in severe depression? J Mol Neurosci 18(1–2):143–149. https://doi.org/10.1385/JMN:18:1-2:143
Seale JV, Wood SA, Atkinson HC, Bate E, Lightman SL, Ingram CD, Jessop DS, Harbuz MS (2004) Gonadectomy reverses the sexually diergic patterns of circadian and stress-induced hypothalamic-pituitary-adrenal axis activity in male and female rats. J Neuroendocrinol 16(6):516–524. https://doi.org/10.1111/j.1365-2826.2004.01195.x
Stanciu CN, Glass OM, Penders TM (2017) Use of buprenorphine in treatment of refractory depression—a review of current literature. Asian J Psychiatr 26:94–98. https://doi.org/10.1016/j.ajp.2017.01.015
Steru L, Chermat R, Thierry B, Simon P (1985) The tail suspension test—a new method for screening antidepressants in mice. Psychopharmacology 85(3):367–370. https://doi.org/10.1007/Bf00428203
Stoll AL, Rueter S (1999) Treatment augmentation with opiates in severe and refractory major depression. Am J Psychiatry 156(12):2017. https://doi.org/10.1176/ajp.156.12.2017
Wong ML, Licinio J (2001) Research and treatment approaches to depression. Nat Rev Neurosci 2(5):343–351. https://doi.org/10.1038/35072566
Yang C, Li WY, Yu HY, Gao ZQ, Liu XL, Zhou ZQ, Yang JJ (2012a) Tramadol pretreatment enhances ketamine-induced antidepressant effects and increases mammalian target of rapamycin in rat hippocampus and prefrontal cortex. J Biomed Biotechnol 2012:175619–175616. https://doi.org/10.1155/2012/175619
Yang C, Li X, Wang N, Xu S, Yang J, Zhou Z (2012b) Tramadol reinforces antidepressant effects of ketamine with increased levels of brain-derived neurotrophic factor and tropomyosin-related kinase B in rat hippocampus. Front Med 6(4):411–415. https://doi.org/10.1007/s11684-012-0226-2
Zomkowski AD, Santos AR, Rodrigues ALS (2005) Evidence for the involvement of the opioid system in the agmatine antidepressant-like effect in the forced swimming test. Neurosci Lett 381(3):279–283. https://doi.org/10.1016/j.neulet.2005.02.026
Acknowledgements
This study was supported by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, #308723/2013-9), Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior (CAPES), and NENASC Project (PRONEX-FAPESC/CNPq, #1262/2012-9). ALSR is a CNPq Research Fellow. LEBB is an International Brain Research Organization (IBRO) postdoctoral fellow.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Animals were used according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals, and the protocol and experiments were approved by the Ethical Committee of Animal Research of Federal University of Santa Catarina.
Rights and permissions
About this article
Cite this article
Moretti, M., Ribeiro, C.M., Neis, V.B. et al. Evidence for the involvement of opioid system in the antidepressant-like effect of ascorbic acid. Naunyn-Schmiedeberg's Arch Pharmacol 391, 169–176 (2018). https://doi.org/10.1007/s00210-017-1446-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-017-1446-4