Abstract
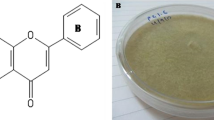
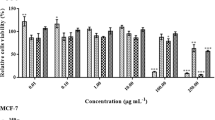
Endophytic fungi were isolated from the marine green alga Chaetomorpha antennina and identified as Clonostachys rosea through molecular analysis. C. rosea was grown in a tryptophan medium for 21 days and after that, the metabolites were extracted by ethyl acetate. The ethyl acetate extract showed a high cytotoxic effect on MCF-7 cells. GC–MS analysis of the ethyl acetate extract revealed the presence of many compounds, and chrysin was one of the major compounds among them. Hence, further studies were concentrated on chrysin, as it was assumed to be the major attributor to the potent cytotoxicity, based on its high anticancer efficacies reported earlier. The fungal ethyl acetate extract had been analysed for chrysin using HPTLC and compared its Rf value with authentic chrysin and it was matched. Further, the purified fungal chrysin was structurally elucidated using techniques like LC–MS and NMR analyses. Quantification revealed that C. rosea produced 1050 mg/L of chrysin. This surplus production of chrysin was the major significance of the study. The purified fungal chrysin was found to be highly cytotoxic to MCF-7 cells with a low IC50 value 35.5 ± 0.6 µM. Furthermore, DNA fragmentation and apoptosis analysis indicated the selective inhibition of MCF-7 by DNA damage. Thus, the present study implies that C. rosea is an alternative source and new method for surplus production of chrysin in the tryptophan medium. All results indicate that the marine algae endophytic C. rosa produces chrysin, and for the first time, an excess amount of production was revealed by the study.












Similar content being viewed by others
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abotaleb M, Samuel SM, Varghese E, Varghese S, Kubatka P, Liskova A, Büsselberg D (2019) Flavonoids in cancer and apoptosis. Cancers 11(1):28. https://doi.org/10.3390/cancers11010028
Al-Ishaq RK, Abotaleb M, Kubatka P, Kajo K, Büsselberg D (2019) Flavonoids and their anti-diabetic effects: cellular mechanisms and effects to improve blood sugar levels. Biomolecules 9(9):430. https://doi.org/10.3390/biom9090430
Anand KV, Mohamed Jaabir MS, Thomas PA, Geraldine P (2012) Protective role of chrysin against oxidative stress in d-galactose-induced aging in an experimental rat model. Geriatr Gerontol Int 12(4):741–750. https://doi.org/10.1111/j.1447-0594.2012.00843.x
Arnold AE, Maynard Z, Gilbert GS, Coley PD, Kursar TA (2000) Are tropical fungal endophytes hyper-diverse? Ecol Lett 3:267–274. https://doi.org/10.1046/j.1461-0248.2000.00159.x.
Beg F, Bhaita AM, Baig MU (2014) Biological activity of modified chrysin. Int J Multidiscip Curr Res 18:2555
Chahar MK, Sharma N, Dobhal MP, Joshi YC (2011) Flavonoids: a versatile source of anticancer drugs. Pharmacogn Rev 5(9):1. https://doi.org/10.4103/0973-7847.79093
Crowley LC, Marfell BJ, Scott AP, Waterhouse NJ (2016) Quantitation of apoptosis and necrosis by annexin V binding, propidium iodide uptake, and flow cytometry. Cold Spring Harbor Protoc 11:087288. https://doi.org/10.1101/pdb.prot087288
Debbab A, Aly AH, Proksch P (2011) Bioactive secondary metabolites from endophytes and associated marine derived fungi. Fungal Divers 49:1–12. https://doi.org/10.1007/s13225-011-0114-0
Faulkner DJ (2000) Marine natural products. Nat Prod Rep 17(1):7–55. https://doi.org/10.1039/a809395d
Gresa-Arribas N, Serratosa J, Saura J, Solà C (2010) Inhibition of CCAAT/enhancer binding protein δ expression by chrysin in microglial cells results in anti-inflammatory and neuroprotective effects. J Neurochem 115(2):526–536. https://doi.org/10.1111/j.1471-4159.2010.06952.x
Hadjmohammadi MR, Nazari SS (2017) Separation optimization of quercetin, hesperetin and chrysin in honey by micellar liquid chromatography and experimental design. J Sep Sci 33:3144–3151. https://doi.org/10.1002/jssc.201000326
Kamat S, Kumari M, Sajna KV, Jayabaskaran C (2020) Endophytic fungus, Chaetomium globosum, associated with marine green alga, a new source of chrysin. Sci Rep 10(1):1–7. https://doi.org/10.1038/s41598-020-72497-3
Kaul S, Gupta S, Ahmed M, Dhar MK (2012) Endophytic fungi from medicinal plants: a treasure hunt for bioactive metabolites. Phytochem Rev 11:487–505. https://doi.org/10.1007/s11101-012-9260-6
Kumar A, Patil D, Rajamohanan PR, Ahmad A (2013) Isolation, purification and characterization of vinblastine and vincristine from endophytic fungus Fusarium oxysporum isolated from Catharanthus roseus. PLoS ONE 8(9):71805. https://doi.org/10.1371/journal.pone.0071805
Kumari M, Taritla S, Sharma A, Jayabaskaran C (2018) Antiproliferative and antioxidative bioactive compounds in extracts of marine-derived endophytic fungus Talaromyces purpureogenus. Front Microbiol 9:1777. https://doi.org/10.3389/fmicb.2018.01777
Larit F, Benyahia S, Benayache S, Benayache F, Léon F, Brouard I et al (2012) Flavonoïds from Calycotome spinosa (L.) Lamk. Int J Med Arom Plants 2(1):34–37
Liu IX, Durham DG, Richards RM (2001) Vancomycin resistance reversal in enterococci by flavonoids. J Pharm Pharmacol 53(1):129–132. https://doi.org/10.1211/0022357011775118
Mani R, Natesan V (2018) Chrysin: Sources, beneficial pharmacological activities, and molecular mechanism of action. Phytochemistry 145:187–196. https://doi.org/10.1016/j.phytochem.2017.09.016
Marris E (2006) Drugs from the deep. Nature 443:904–905. https://doi.org/10.1038/443904a
Medić-Šarić M, Jasprica I, Smolčić-Bubalo A, Mornar A (2004) Optimization of chromatographic conditions in thin layer chromatography of flavonoids and phenolic acids. Croat Chem Acta 77(1–2):361–366
Mousa WK, Raizada MN (2013) The diversity of anti-microbial secondary metabolites produced by fungal endophytes: an interdisciplinary perspective. Front Microbiol 4:65. https://doi.org/10.3389/fmicb.2013.00065
Parthasarathy R, Chandrika M, Yashavantha Rao HC, Kamalraj S, Jayabaskaran C, Pugazhendhi A (2020a) Molecular profiling of marine endophytic fungi from green algae: assessment of antibacterial and anticancer activities. Process Biochem 1(96):11–20. https://doi.org/10.1016/j.procbio.2020.05.012
Parthasarathy R, Shanmuganathan R, Pugazhendhi A (2020b) Vinblastine production by the endophytic fungus Curvularia verruculosa from the leaves of Catharanthus roseus and it’s in vitro cytotoxicity against HeLa cell line. Anal Biochem 593:113530. https://doi.org/10.1016/j.ab.2019.113530
Pietra F (1997) Secondary metabolites from marine microorganisms: bacteria, protozoa, algae and fungi. Achiev Prospect Nat Prod Rep 14:453–464. https://doi.org/10.1039/np9971400453
Qin L, Dixon RA, Mabry TJ (1993) Additional flavonoids from elicitor-treated cell cultures of Cephalocereus senilis. Phytochemistry 34(1):167–170. https://doi.org/10.1016/S0031-9422(00)90800-8
Rasouli S, Zarghami N (2018) Synergistic growth inhibitory effects of chrysin and metformin combination on breast cancer cells through hTERT and cyclin D1 suppression. Asian Pac J Cancer Prev 19(4):977. https://doi.org/10.22034/APJCP.2018.19.4.977
Samarghandian S, Azimi-Nezhad M, Borji A, Hasanzadeh M, Jabbari F, Farkhondeh T et al (2016) Inhibitory and cytotoxic activities of chrysin on human breast adenocarcinoma cells by induction of apoptosis. Pharmacogn Mag 12(Suppl 4):S436–S440. https://doi.org/10.4103/0973-1296.191453
Sarasan M, Puthumana J, Job N, Han J, Lee JS, Philip R (2017) Marine algicolous endophytic fungi-a promising drug resource of the era. J Microbiol Biotechnol 27(6):1039–1052. https://doi.org/10.4014/jmb.1701.01036
Sathishkumar G, Bharti R, Jha PK, Selvakumar M, Dey G, Jha R et al (2015) Dietary flavone chrysin (5, 7-dihydroxyflavone ChR) functionalized highly-stable metal nanoformulations for improved anticancer applications. RSC Adv 5(109):89869–89878. https://doi.org/10.1039/C5RA15060D
Schulz B, Boyle C (2005) The endophytic continuum. Mycol Res 109(6):661–686. https://doi.org/10.1017/s095375620500273x
Seetharaman P, Gnanasekar S, Chandrasekaran R, Chandrakasan G, Kadarkarai M, Sivaperumal S (2017) Isolation and characterization of anticancer flavone chrysin (5, 7-dihydroxy flavone)-producing endophytic fungi from Passiflora incarnata L. leaves. Ann Microbiol 67(4):321–331. https://doi.org/10.1007/s13213-017-1263-5
Strobel G, Daisy B (2003) Bioprospecting for microbial endophytes and their natural products. Microbiol Mol Biol Rev 67:491–502. https://doi.org/10.1128/mmbr.67.4.491-502.2003
Sturz AV, Christie BR, Nowak J (2000) Bacterial endophytes: potential role in developing sustainable systems of crop production. Crit Rev Plant Sci 19(1):1–30. https://doi.org/10.1080/07352680091139169
Taritla S, Kumari M, Kamat S, Bhat SG, Jayabaskaran C (2021) Optimization of physicochemical parameters for production of cytotoxic secondary metabolites and apoptosis induction activities in the culture extract of a marine algal-derived endophytic fungus Aspergillus sp. Front Pharmacol 12:542891. https://doi.org/10.3389/fphar.2021.542891
Torres-Piedra M, Ortiz-Andrade R, Villalobos-Molina R, Singh N, Webster M-F, SP, et al (2010) comparative study of flavonoid analogues on streptozotocin–nicotinamide induced diabetic rats: Quercetin as a potential antidiabetic agent acting via 11β-hydroxysteroid dehydrogenase type 1 inhibition. Eur J Med Chem 45(6):2606–2612. https://doi.org/10.1016/j.ejmech.2010.02.049
Venkatraman P (2010) Specificity in the interaction of natural products with their target proteins—a biochemical and structural insight. Mini Rev Med Chem 10(6):540–549. https://doi.org/10.2174/138955710791384090
Wang J, Qiu J, Dong J, Li H, Luo M, Dai X et al (2011) Chrysin protects mice from Staphylococcus aureus pneumonia. J App Microbiol 111(6):1551–1558. https://doi.org/10.1111/j.1365-2672.2011.05170.x
Xiang W, Ke Z, Zhang Y, Cheng GH-Y, Irwan ID, Sulochana KN, Potturi P, Wang Z, Yang H, Wang J, Zhuo L (2011) Isthmin is a novel secreted angiogenesis inhibitor that inhibits tumour growth in mice. J Cell Mol Med 2:359–374. https://doi.org/10.1111/j.1582-4934.2009.00961.x
Xue C, Chen Y, Hu DN, Iacob C, Lu C, Huang Z (2016) Chrysin induces cell apoptosis in human uveal melanoma cells via intrinsic poptosis. Oncol Lett 12(6):4813–4820. https://doi.org/10.3892/ol.2016.5251
Yang F, Jin H, Pi J, Jiang JH, Liu L, Bai HH, Yang PH, Cai JY (2013) Anti-tumor activity evaluation of novel chrysin-organogermanium(IV) complex in MCF-7 cells. Bioorg Med Chem Lett 23:5544–5555
Zaveri M, Jain S (2007) Gastroprotective effects of root bark of Oroxylum indicum, vent. J Nat Rem 7(2):269–277. https://doi.org/10.18311/jnr/2007/319
Funding
Dr. Ramalingam Parthasarathy thanks to funding from Department of Science and Technology and Science and Engineering Research Board (DST-SERB), New Delhi for the National Post-Doctoral fellowship (Ref. no. PDF/2017/001184), Indian Council Medical Research-Research Associate (ICMR-RA) (Ref. no. 45/1/2020-DDI-BMS), New Delhi, for the research associate fellowship Dr. Chandrika Manjegowda, thanks to Indian Council Medical Research (ICMR) for providing financial support (File no.45/36-2018-PHA/BMS Dated 23/6/2018). Dr. D. Sruthi thanks Department of Health Research (DHR), Government of India, New Delhi, for her award of Young scientist -HRD Scheme (YSS/2019/000035/PRCYSS). Authors thank FACS and confocal microscopy facility, Department of Biochemistry, Division of Biological Sciences, Indian Institute of Science (IISc), Bangalore. Dr. H.C. Yashavantha Rao acknowledges Young Scientist grant from DHR-HRD Indian Council of Medical Research (YSS/2020/0000054/PRCYSS) and University Grants Commission—DSKPDF (No.F.4-2/2006 (BSR)/BL/17-18/0234) Government of India, New Delhi.
Author information
Authors and Affiliations
Contributions
RP—investigation and methodology, writing review and editing original draft, conceptualization; MC—investigation, review; DS& HCYR—review and editing draft; CJ—supervision.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Additional information
Communicated by Dr. El-Sayed R. El-Sayed.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Parthasarathy, R., Chandrika, M., Sruthi, D. et al. Clonostachys rosea, a marine algal endophyte, as an alternative source of chrysin and its anticancer effect. Arch Microbiol 205, 275 (2023). https://doi.org/10.1007/s00203-023-03615-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03615-8