Abstract
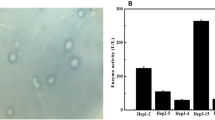
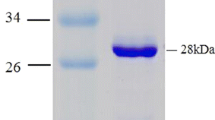
The enzymes are biological macromolecules that biocatalyze certain biochemical reactions without undergoing any modification or degradation at the end of the reaction. In this work, we constructed a recombinant novel Raoultella sp. NX-TZ-3-15 strain that produces heparinase with a maltose binding tag to enhance its production and activity. Additionally, MBP–heparinase was purified and its enzymatic capabilities are investigated to determine its industrial application. Moreover, the recombinant plasmid encoding the MBP–heparinase fusion protein was effectively generated and purified to a high purity. According to SDS-PAGE analysis, the MBP–heparinase has a molecular weight of around 70 kDa and the majority of it being soluble with a maximum activity of 5386 U/L. It has also been noted that the three ions of Ca2 + , Co2 + , and Mg2 + can have an effect on heparinase activities, with Mg2 + being the most noticeable, increasing by about 85%, while Cu2 + , Fe2 + , Zn2 + having an inhibitory effect on heparinase activities. Further investigations on the mechanistic action, structural features, and genomes of Raoultella sp. NX-TZ-3-15 heparinase synthesis are required for industrial-scale manufacturing.





Similar content being viewed by others
Data availability statement
The raw data will be available on reasonable request from corresponding author.
References
Ahsan A, Jeske W, Hoppensteadt D, Lormeau JC, Wolf H, Fareed J (1995) Molecular profiling and weight determination of heparins and depolymerized heparins. J Pharm Sci 84:724–727. https://doi.org/10.1002/jps.2600840612
Ansell JE, Laulicht BE, Bakhru SH, Hoffman M, Steiner SS, Costin JC (2016) Ciraparantag safely and completely reverses the anticoagulant effects of low molecular weight heparin. Thromb Res 146:113–118. https://doi.org/10.1016/j.thromres.2016.07.008
Atallah J, Khachfe HH, Berro J, Assi HI (2020) The use of heparin and heparin-like molecules in cancer treatment: a review. Cancer Treat Res Commun 24:100192. https://doi.org/10.1016/j.ctarc.2020.100192
Baytas SN, Linhardt RJ (2020) Advances in the preparation and synthesis of heparin and related products. Drug Discov Today 25:2095–2109. https://doi.org/10.1016/j.drudis.2020.09.011
Campos J, Mourão J, Pestana N, Peixe L, Novais C, Antunes P (2013) Microbiological quality of ready-to-eat salads: an underestimated vehicle of bacteria and clinically relevant antibiotic resistance genes. Int J Food Microbiol 166:464–470. https://doi.org/10.1016/j.ijfoodmicro.2013.08.005
Costa S, Almeida A, Castro A, Domingues L (2014) Fusion tags for protein solubility, purification and immunogenicity in Escherichia coli: the novel Fh8 system. Front Microbiol 5:63. https://doi.org/10.3389/fmicb.2014.00063
Du S, Yu Y, Xu C, Xiong H, Yang S, Yao J (2019) LMWH and its derivatives represent new rational for cancer therapy: construction strategies and combination therapy. Drug Discov Today 24:2096–2104. https://doi.org/10.1016/j.drudis.2019.06.011
Ejaz U, Akhtar F, Xue J, Wan X, Zhang T, He S (2021) Review: Inhibitory potential of low molecular weight Heparin in cell adhesion; emphasis on tumor metastasis. Eur J Pharmacol 892:173778. https://doi.org/10.1016/j.ejphar.2020.173778
Han L, Liu Q, Yang L, Ye T, He Z, Jia L (2017) Facile oriented immobilization of histidine-tagged proteins on nonfouling cobalt polyphenolic self-assembly surfaces. ACS Biomater Sci Eng 3:3328–3337. https://doi.org/10.1021/acsbiomaterials.7b00691
Huang Z et al (2013) Rational design of a tripartite fusion protein of heparinase I enables one-step affinity purification and real-time activity detection. J Biotechnol 163:30–37. https://doi.org/10.1016/j.jbiotec.2012.09.016
Jiang R et al (2020) Fluorescence detection of protamine, heparin and heparinase II based on a novel AIE molecule with four carboxyl. Int J Biol Macromol 156:1153–1159. https://doi.org/10.1016/j.ijbiomac.2019.11.150
Kuang Y et al (2006) Production of heparin oligosaccharides by fusion protein of MBP–heparinase I and the enzyme thermostability. J Mol Catal B Enzym 43:90–95. https://doi.org/10.1016/j.molcatb.2006.06.022
Li X, Lee KH, Shorkey S, Chen J, Chen M (2020) Different anomeric sugar bound states of maltose binding protein resolved by a cytolysin a nanopore tweezer. ACS Nano 14:1727–1737. https://doi.org/10.1021/acsnano.9b07385
Li Y, Lin Y, Jiang Y, Mehwish HM, Rajoka MSR, Zhao L (2022) Cloning and expression of heparinase gene from a novel strain Raoultella NX-TZ-3-15. Appl Biochem Biotechnol. https://doi.org/10.1007/s12010-022-03917-5
Liu Y et al (2020) Comparative genomics analysis of Raoultella planticola S25 isolated from duck in China, with florfenicol resistance. Comp Immunol Microbiol Infect Dis 68:101398. https://doi.org/10.1016/j.cimid.2019.101398
Onishi A, Ange KS, Dordick JS, Linhardt RJ (2016) Heparin and Anticoagulation. Front Biosci (landmark Ed) 21:1372–1392. https://doi.org/10.2741/4462
Reuten R et al (2016) Maltose-binding protein (MBP), a secretion-enhancing tag for mammalian protein expression systems. PLoS ONE 11:e0152386. https://doi.org/10.1371/journal.pone.0152386
Selmke B, Borbat PP, Nickolaus C, Varadarajan R, Freed JH, Trommer WE (2018) Open and closed form of maltose binding protein in its native and molten globule state as studied by electron paramagnetic resonance spectroscopy. Biochem 57:5507–5512. https://doi.org/10.1021/acs.biochem.8b00322
Seng P et al (2016) Emerging role of Raoultella ornithinolytica in human infections: a series of cases and review of the literature. Int J Infect Dis 45:65–71. https://doi.org/10.1016/j.ijid.2016.02.014
Wang T, Liu L, Voglmeir J (2020) Chemoenzymatic synthesis of ultralow and low-molecular weight heparins. Biochim Biophys Acta Proteins Proteom 1868:140301. https://doi.org/10.1016/j.bbapap.2019.140301
Wu J, Zhang C, Mei X, Li Y, Xing X-H (2014) Controllable production of low molecular weight heparins by combinations of heparinase I/II/III. Carbohydr Polym 101:484–492. https://doi.org/10.1016/j.carbpol.2013.09.052
Xu S, Qiu M, Zhang X, Chen J (2017) Expression and characterization of an enhanced recombinant heparinase I with chitin binding domain. Int J Biol Macromol 105:1250–1258. https://doi.org/10.1016/j.ijbiomac.2017.07.158
Yates EA, Rudd TR (2016) Recent innovations in the structural analysis of heparin. Int J Cardiol 212(Suppl 1):S5-9. https://doi.org/10.1016/s0167-5273(16)12002-9
Ye F, Kuang Y, Chen S, Zhang C, Chen Y, Xing X-H (2009) Characteristics of low molecular weight heparin production by an ultrafiltration membrane bioreactor using maltose binding protein fused heparinase I. Biochem Eng J 46:193–198. https://doi.org/10.1016/j.bej.2009.05.007
Yu P, Jia T, Chen Y, Wu Y, Zhang Y (2016) Improving the activity of heparinase I by the directed evolution, its enzymatic properties and optimal conditions for heparin degrading by recombinant cells. Biochem Eng J 114:237–243. https://doi.org/10.1016/j.bej.2016.07.011
Funding
This work was supported by the National Key R&D Program of China (2021YFA0910800), the Natural Science Foundation of Guangdong Province (Grant No. 2022A1515012043), and the Special Funds for Development of Strategic Emerging Industries in Shenzhen (JCYJ20190808145613154, KQJSCX20180328100801771), and Shenzhen Science and Technology Program (ZDSYS20210623100800001).
Author information
Authors and Affiliations
Contributions
YL Conceptualization, methodology, software. YL Conceptualization, methodology, software. YJ Visualization, investigation. MSRR Software, validation, writing—original draft preparation. HMM: Data curation, writing—original draft preparation. MU: Software and original draft preparation. LZ: Software, validation, Supervision. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors have no conflict of interest.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Y., Lin, Y., Jiang, Y. et al. Expression and characterization of heparinase II with MBP tag from a novel strain, Raoultella NX-TZ-3-15. Arch Microbiol 204, 551 (2022). https://doi.org/10.1007/s00203-022-03158-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-03158-4