Abstract
Key message
A novel qualitative locus regulating the uppermost internode elongation of barley was identified and mapped on 6H, and the candidate gene mining was performed by employing various barley genomic resources.
Abstract
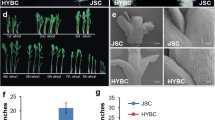
The stem of grass crops, such as barley and wheat, is composed of several interconnected internodes. The extent of elongation of these internodes determines stem height, and hence lodging, canopy architecture, and grain yield. The uppermost internode (UI) is the last internode to elongate. Its elongation contributes largely to stem height and facilitates spike exsertion, which is crucial for final grain yield. Despite the molecular mechanism underlying regulation of UI elongation was extensively investigated in rice, little is known in barley. In this study, we characterized a barley spontaneous mutant, Sheathed Spike 1 (SS1), showing significantly shortened UI and sheathed spike (SS). The extension of UI parenchyma cell in SS1 was significantly suppressed. Exogenous hormone treatments and RNA-seq analysis indicated that the suppression of UI elongation is possibly related to insufficient content of endogenous bioactive gibberellin. Genetic analysis showed that SS1 is possibly controlled by a qualitative dominant nuclear factor. Bulked segregant analysis and further molecular marker mapping identified a novel major locus, HvSS1, in a recombination cold spot expanding 173.44–396.33 Mb on chromosome 6H. The candidate gene mining was further conducted by analyzing sequence differences, spatiotemporal expression patterns, and variant distributions of genes in the candidate interval by employing various barley genomic resources of worldwide collections of barley accessions. This study made insight into genetic control of UI elongation in barley and laid a solid foundation for further gene cloning and functional characterization. The results obtained here also provided valuable information for similar research in wheat.






Similar content being viewed by others
References
Alvarez RV, Pongor LS, Marino-Ramirez L, Landsman D (2019) TPMCalculator: one-step software to quantify mRNA abundance of genomic features. Bioinformatics 35:1960–1962
Ashikari M, Wu JZ, Yano M, Sasaki T, Yoshimura A (1999) Rice gibberellin-insensitive dwarf mutant gene Dwarf 1 encodes the alpha-subunit of GTP-binding protein. Proc Natl Acad Sci USA 96:10284–10289
Bardenas EA (1965) The morphology and varietal characteristics of the rice plant. Technical Bulletin. International Rice Research Institute, Manila
Brinton J, Simmonds J, Minter F, Leverington-Waite M, Snape J, Uauy C (2017) Increased pericarp cell length underlies a major quantitative trait locus for grain weight in hexaploid wheat. New Phytol 215:1026–1038
Chen H-J, Wang S-J (2008) Molecular regulation of sink-source transition in rice leaf sheaths during the heading period. Acta Physiol Plant 30:639–649
Chiang C, Layer RM, Faust GG, Lindberg MR, Rose DB, Garrison EP, Marth GT, Quinlan AR, Hall IM (2015) SpeedSeq: ultra-fast personal genome analysis and interpretation. Nat Methods 12:966–968
Cho E, Zambryski PC (2011) ORGAN BOUNDARY1 defines a gene expressed at the junction between the shoot apical meristem and lateral organs. Proc Natl Acad Sci USA 108:2154–2159
Cingolani P, Platts A, Wang LL, Coon M, Tung N, Wang L, Land SJ, Lu X, Ruden DM (2012) A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w(1118); iso-2; iso-3. Fly 6:80–92
Deng Q, Kong Z, Wu X, Ma S, Yuan Y, Jia H, Ma Z (2019) Cloning of a COBL gene determining brittleness in diploid wheat using a MapRseq approach. Plant Sci 285:141–150
Faust GG, Hall IM (2014) SAMBLASTER: fast duplicate marking and structural variant read extraction. Bioinformatics 30:2503–2505
Gao S, Fang J, Xu F, Wang W, Chu C (2016) Rice HOX12 regulates panicle exsertion by directly modulating the expression of ELONGATED UPPERMOST INTERNODE1. Plant Cell 28:680–695
Garrison E, Marth G (2012) Haplotype-based variant detection from short-read sequencing. arXiv:1207.3907 [q-bio.GN]
Hao C, Jiao C, Hou J, Li T, Liu H, Wang Y, Zheng J, Liu H, Bi Z, Xu F, Zhao J, Ma L, Wang Y, Majeed U, Liu X, Appels R, Maccaferri M, Tuberosa R, Lu H, Zhang X (2020) Resequencing of 145 landmark cultivars reveals asymmetric sub-genome selection and strong founder genotype effects on wheat breeding in China. Mol Plant 13:1733–1751
Hedden P (2003) The genes of the Green Revolution. Trends Genet 19:5–9
Hempel FD, Feldman LJ (1994) Bi-directional in florescence development in Arabidopsis thaliana: acropetal initiation of flowers and basipetal initiation of paraclades. Planta 192:276–286
Jossier M, Bouly J-P, Meimoun P, Arjmand A, Lessard P, Hawley S, Grahame Hardie D, Thomas M (2009) SnRK1 (SNF1-related kinase 1) has a central role in sugar and ABA signalling in Arabidopsis thaliana. Plant J 59:316–328
Kim D, Langmead B, Salzberg SL (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12:357-U121
Kirby EJM, Appleyard M, Simpson NA (1994) Co-ordination of stem elongation and Zadoks growth stages with leaf emergence in wheat and barley. J Agric Sci 122:21–29
Kohorn BD, Kohorn SL (2012) The cell wall-associated kinases, WAKs, as pectin receptors. Front Plant Sci 3:88
Lally D, Ingmire P, Tong HY, He ZH (2001) Antisense expression of a cell wall-associated protein kinase, WAK4, inhibits cell elongation and alters morphology. Plant Cell 13:1317–1331
Lee M, Dong X, Song H, Yang JY, Kim S, Hur Y (2020) Molecular characterization of Arabidopsis thaliana LSH1and LSH2 genes. Genes & Genomics 42:1151–1162
Li C, Tang H, Luo W, Zhang X, Mu Y, Deng M, Liu Y, Jiang Q, Chen G, Wang J, Qi P, Pu Z, Jiang Y, Wei Y, Zheng Y, Lan X, Ma J (2020) A novel, validated, and plant height-independent QTL for spike extension length is associated with yield-related traits in wheat. Theor Appl Genet 133:3381–3393
Liang J, Chen X, Deng G, Pan Z, Zhang H, Li Q, Yang K, Long H, Yu M (2017) Dehydration induced transcriptomic responses in two Tibetan hulless barley (Hordeum vulgare var. nudum) accessions distinguished by drought tolerance. BMC Genomics 18:775
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550
Luo AD, Qian Q, Yin HF, Liu XQ, Yin CX, Lan Y, Tang JY, Tang ZS, Cao SY, Wang XJ, Xia K, Fu XD, Luo D, Chu CC (2006) EUI1, encoding a putative cytochrome P450 monooxygenase, regulates internode elongation by modulating gibberellin responses in rice. Plant Cell Physiol 47:181–191
Ma J, Sun M, Ding P, Luo W, Zhou X, Yang C, Zhang H, Qin N, Yang Y, Lan X (2017) Genetic identification of QTL for neck length of spike in wheat. Journal of Triticeae Crops 37:319–324
Magwene PM, Willis JH, Kelly JK (2011) The statistics of bulk segregant analysis using next generation sequencing. PLoS Comput Biol 7:e1002255
Mascher M, Gundlach H, Himmelbach A, Beier S, Twardziok SO, Wicker T, Radchuk V, Dockter C, Hedley PE, Russell J, Bayer M, Ramsay L, Liu H, Haberer G, Zhang X-Q, Zhang Q, Barrero RA, Li L, Taudien S, Groth M, Felder M, Hastie A, Simkova H, Stankova H, Vrana J, Chan S, Munoz-Amatrian M, Ounit R, Wanamaker S, Bolser D, Colmsee C, Schmutzer T, Aliyeva-Schnorr L, Grasso S, Tanskanen J, Chailyan A, Sampath D, Heavens D, Clissold L, Cao S, Chapman B, Dai F, Han Y, Li H, Li X, Lin C, McCooke JK, Tan C, Wang P, Wang S, Yin S, Zhou G, Poland JA, Bellgard MI, Borisjuk L, Houben A, Dolezel J, Ayling S, Lonardi S, Kersey P, Lagridge P, Muehlbauer GJ, Clark MD, Caccamo M, Schulman AH, Mayer KFX, Platzer M, Close TJ, Scholz U, Hansson M, Zhang G, Braumann I, Spannagl M, Li C, Waugh R, Stein N (2017) A chromosome conformation capture ordered sequence of the barley genome. Nature 544:426–433
McKim SM (2019) How plants grow up. J Integr Plant Biol 61:257–277
Milner SG, Jost M, Taketa S, Mazon ER, Himmelbach A, Oppermann M, Weise S, Knuepffer H, Basterrechea M, Koenig P, Schueler D, Sharma R, Pasam RK, Rutten T, Guo G, Xu D, Zhang J, Herren G, Müller T, Krattinger SG, Keller B, Jiang Y, Gonzalez MY, Zhao Y, Habekuss A, Faerber S, Ordon F, Lange M, Börner A, Graner A, Reif JC, Scholz U, Mascher M, Stein N (2019) Genebank genomics highlights the diversity of a global barley collection. Nat Genet 51:319–326
Monat C, Padmarasu S, Lux T, Wicker T, Gundlach H, Himmelbach A, Ens J, Li C, Muehlbauer GJ, Schulman AH, Waugh R, Braumann I, Pozniak C, Scholz U, Mayer KFX, Spannagl M, Stein N, Mascher M (2019) TRITEX: chromosome-scale sequence assembly of Triticeae genomes with open-source tools. Genome Biol 20:284
Neff MM, Turk E, Kalishman M (2002) Web-based primer design for single nucleotide polymorphism analysis. Trends Genet 18:613–615
Nicholls PB, MayLH, (1964) Studies on the growth of the barley apex. II. on the initiation of internode elongation in the in florescence. Aust J Biol Sci 17:619–630
Peng JR, Richards DE, Hartley NM, Murphy GP, Devos KM, Flintham JE, Beales J, Fish LJ, Worland AJ, Pelica F, Sudhakar D, Christou P, Snape JW, Gale MD, Harberd NP (1999) “Green revolution” genes encode mutant gibberellin response modulators. Nature 400:256–261
Qi LL, Gill BS (2001) High-density physical maps reveal that the dominant male-sterile gene Ms3 is located in a genomic region of low recombination in wheat and is not amenable to map-based cloning. Theor Appl Genet 103:998–1006
Rapazote-Flores P, Bayer M, Milne L, Mayer C-D, Fuller J, Guo W, Hedley PE, Morris J, Halpin C, Kam J, McKim SM, Zwirek M, Casao MC, Barakate A, Schreiber M, Stephen G, Zhang R, Brown JWS, Waugh R, Simpson CG (2019) BaRTv1.0: an improved barley reference transcript dataset to determine accurate changes in the barley transcriptome using RNA-seq. BMC Genomics 20:968
Ren XF, Sun DF, Dong WB, Sun GL, Li CD (2014) Molecular detection of QTL controlling plant height components in a doubled haploid barley population. Genet Mol Res 13:3089–3099
Rogers SO, Bendich AJ (1985) Extraction of DNA from milligram amounts of fresh, herbarium and mummified plant tissues. Plant Mol Biol 5:69–76
Russell J, Mascher M, Dawson IK, Kyriakidis S, Calixto C, Freund F, Bayer M, Milne I, Marshall-Griffiths T, Heinen S, Hofstad A, Sharma R, Himmelbach A, Knauft M, van Zonneveld M, Brown JWS, Schmid K, Kilian B, Muehlbauer GJ, Stein N, Waugh R (2016) Exome sequencing of geographically diverse barley landraces and wild relatives gives insights into environmental adaptation. Nat Genet 48:1024
Rutger JN, Carnahan HL (1981) A fourth genetic element to facilitate hybrid cereal production—a recessive tall in rice. Crop Sci 21:373–376
Sameri M, Nakamura S, Nair SK, Takeda K, Komatsuda T (2009) A quantitative trait locus for reduced culm internode length in barley segregates as a Mendelian gene. Theor Appl Genet 118:643–652
Sasaki A, Ashikari M, Ueguchi-Tanaka M, Itoh H, Nishimura A, Swapan D, Ishiyama K, Saito T, Kobayashi M, Khush GS, Kitano H, Matsuoka M (2002) Green revolution: A mutant gibberellin-synthesis gene in rice - New insight into the rice variant that helped to avert famine over thirty years ago. Nature 416:701–702
Schnurbusch T (2019) Wheat and barley biology: Towards new frontiers. J Integr Plant Biol 61:198–203
Shen ZT, Yang CD, He ZH (1987) Studies on eliminating panicle enclosure in WA type MS line of rice (Oryza sativa subsp. Indica). Chin J Rice Sci 1(1):95–99
Spielmeyer W, Ellis MH, Chandler PM (2002) Semidwarf (sd-1), “green revolution” rice, contains a defective gibberellin 20-oxidase gene. Proc Natl Acad Sci USA 99:9043–9048
Takagi H, Abe A, Yoshida K, Kosugi S, Natsume S, Mitsuoka C, Uemura A, Utsushi H, Tamiru M, Takuno S, Innan H, Cano LM, Kamoun S, Terauchi R (2013) QTL-seq: rapid mapping of quantitative trait loci in rice by whole genome resequencing of DNA from two bulked populations. Plant J 74:174–183
Tao Y, Yi X, Lin Y, Wang Z, Wu F, Jiang X, Liu S, Deng M, Ma J, Chen G, Wei Y, Zheng Y, Liu Y (2019) Quantitative trait locus mapping for panicle exsertion length in common wheat using two related recombinant inbred line populations. Euphytica 215:104
Thomas SG, Phillips AL, Hedden P (1999) Molecular cloning and functional expression of gibberellin 2-oxidases, multifunctional enzymes involved in gibberellin deactivation. Proc Natl Acad Sci USA 96:4698–4703
Tsugama D, Liu S, Takano T (2012) A putative myristoylated 2C-type protein phosphatase, PP2C74, interacts with SnRK1 in Arabidopsis. FEBS Lett 586:693–698
Wang DR, Wolfrum EJ, Virk P, Ismail A, Greenberg AJ, McCouch SR (2016) Robust phenotyping strategies for evaluation of stem non-structural carbohydrates (NSC) in rice. J Exp Bot 67:6125–6138
Wang J, Qin H, Zhou S, Wei P, Zhang H, Zhou Y, Miao Y, Huang R (2020) The ubiquitin-binding protein OsDSK2a mediates seedling growth and salt responses by regulating Gibberellin metabolism in rice. Plant Cell 32:414–428
Weiss D, Ori N (2007) Mechanisms of cross talk between gibberellin and other hormones (vol 144, pg 1240, 2007). Plant Physiol 144:2024–2024
Xie Y, Zhang Y, Han J, Luo J, Li G, Huang J, Wu H, Tian Q, Zhu Q, Chen Y, Kawano Y, Liu Y-G, Chen L (2018) The Intronic cis element SE1 recruits trans-acting repressor complexes to repress the expression of ELONGATED UPPERMOST INTERNODE1 in rice. Mol Plant 11:720–735
Xu Y, Li K, Zhu K, Tian Y, Yu Q, Zhang W, Wang Z (2020) Effect of exogenous plant hormones on agronomic and physiological performance of a leaf early-senescent rice mutantosled. Plant Growth Regul 92:517–533
Xue S, Xu F, Tang M, Zhou Y, Li G, An X, Lin F, Xu H, Jia H, Zhang L, Kong Z, Ma Z (2011) Precise mapping Fhb5, a major QTL conditioning resistance to Fusarium infection in bread wheat (Triticum aestivum L.). Theor Appl Genet 123:1055–1063
Yaish MW, El-kereamy A, Zhu T, Beatty PH, Good AG, Bi Y-M, Rothstein SJ (2010) The APETALA-2-like transcription factor OsAP2-39 controls key interactions between abscisic Acid and gibberellin in rice. PLoS Genet 6:e1001098
Yang R, Zhang S, Huang R, Yang S, Zhang Q (2002) Breeding technology of eui-hybrids of rice. Sci Agric Sin 35:233–237
Yin C, Gan L, Ng D, Zhou X, Xia K (2007) Decreased panicle-derived indole-3-acetic acid reduces gibberellin A(1) level in the uppermost internode, causing panicle enclosure in male sterile rice Zhenshan 97A. J Exp Bot 58:2441–2449
Yu M, Mao S-L, Chen G-Y, Pu Z-E, Wei Y-M, Zheng Y-L (2014) QTLs for uppermost internode and spike length in two wheat RIL populations and their affect upon plant height at an individual QTL level. Euphytica 200:95–108
Zeng X, Guo Y, Xu Q, Mascher M, Guo G, Li S, Mao L, Liu Q, Xia Z, Zhou J, Yuan H, Tai S, Wang Y, Wei Z, Song L, Zha S, Li S, Tang Y, Bai L, Zhuang Z, He W, Zhao S, Fang X, Gao Q, Yin Y, Wang J, Yang H, Zhang J, Henry RJ, Stein N, Tashi N (2018) Origin and evolution of qingke barley in Tibet. Nat Commun 9:5433
Zhao C, Fan X, Wang W, Zhang W, Han J, Chen M, Ji J, Cui F, Li J (2015) Genetic composition and its transmissibility analysis of wheat candidate backbone parent kenong 9204. Acta Agron Sin 41:574–584
Zhu YY, Nomura T, Xu YH, Zhang YY, Peng Y, Mao BZ, Hanada A, Zhou HC, Wang RX, Li PJ, Zhu XD, Mander LN, Kamiya Y, Yamaguchi S, He ZH (2006) ELONGATED UPPERMOST INTERNODE encodes a cytochrome P450 monooxygenase that epoxidizes gibberellins in a novel deactivation reaction in rice. Plant Cell 18:442–456
Zhu T, Liang CZ, Meng ZG, Li YY, Wu YY, Guo SD and Zhang R (2017) PrimerServer: a high-throughput primer design and specificity-checking platform. bioRxiv 181941
Acknowledgments
This work is supported by the Science and Technology Support Project of Sichuan Province, China (2016NZ0103), the Science and Technology Service Network Initiative of the Chinese Academy of Sciences (KFJ-STS-QYZD-2021-22-001), the Major Tibet Science and Technology Projects (XZ2021NA01), and the Innovation Team of Triticeae Crops of Sichuan Province.
Funding
Science and Technology Support Project (2016NZ0103) by Department of Science and Technology of Sichuan Province; Science and Technology Service Network Initiative (KFJ-STS-QYZD-2021-22-001) by Chinese Academy of Sciences; Major Tibet Science and Technology Projects (XZ2021NA01) by Science and Technology Department of Tibet; Innovation Team of Triticeae Crops of Sichuan Province by Sichuan Provincial Department of Agriculture and Rural Affairs;
Author information
Authors and Affiliations
Contributions
XP, YT, MZ, GD, LL, YS, XQ, JW, and ZY participated in the field trials, sampling and phenotyping; YT, MZ, GD, and XP developed populations and YT (Yawei Tang) provided barley germplasm; XP, MZ, JZ performed genotyping; XP, HL, MZ, and TL analyzed data; XP, HL, HZ, JL, and MY discussed results; PX and HL wrote the manuscript; HL, PX, and XQ revised manuscript; HL, GD, and YT (Yawei Tang) acquired funding. HL conceived and designed the study, and all authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethics approval
The authors declare that this research has no human and animal participants and that the experiments comply with the current laws of China.
Availability of data and material
All sequence data generated and used in this study has been deposited in The Genome Sequence Archive (GSA) (CRA004088), Beijing Institute of Genomics, Chinese Academy of Sciences, and is publicly available.
Additional information
Communicated by Takao Komatsuda.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pu, X., Tang, Y., Zhang, M. et al. Identification and candidate gene mining of HvSS1, a novel qualitative locus on chromosome 6H, regulating the uppermost internode elongation in barley (Hordeum vulgare L.). Theor Appl Genet 134, 2481–2494 (2021). https://doi.org/10.1007/s00122-021-03837-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-021-03837-8