Abstract
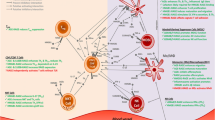
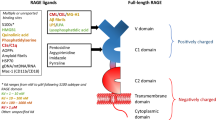
The actors in the pathogenesis of diabetes and its complications are many and multifaceted. The effects of elevated levels of glucose are myriad; among these is the generation of advanced glycation end products (AGEs), the products of nonenzymatic glycoxidation of proteins and lipids. The finding that AGEs stimulate signal transduction cascades through the multiligand receptor RAGE unveiled novel insights into diabetes and its complications. Inextricably woven into AGE–RAGE interactions in diabetes is the engagement of the innate and adaptive immune responses. Although glucose may be the triggering stimulus to draw RAGE into diabetes pathology, consequent cellular stress results in release of proinflammatory RAGE ligands S100/calgranulins and HMGB1. We predict that once RAGE is engaged in the diabetic tissue, a vicious cycle of ligand–RAGE perturbation ensues, leading to chronic tissue injury and suppression of repair mechanisms. Targeting RAGE may be a beneficial strategy in diabetes, its complications, and untoward inflammatory responses.

Similar content being viewed by others
References
Schmidt AM, Vianna M, Gerlach M et al (1992) Isolation and characterization of binding proteins for advanced glycosylation endproducts from lung tissue which are present on the endothelial cell surface. J Biol Chem 267:14987–14997
Ramasamy R, Vannucci SJ, Yan SS et al (2005) Advanced glycation end products and RAGE: a common thread in aging, diabetes, neurodegeneration, and inflammation. Glycobiol 15:16R–28R
Hofmann MA, Drury S, Fu C et al (1999) RAGE mediates a novel proinflammatory axis: a central cell surface receptor for S100/calgranulin polypeptides. Cell 97:889–901
Hori O, Brett J, Slattery T et al (1995) The receptor for advanced glycation endproducts (RAGE) is a cellular binding site for amphoterin: mediation of neurite outgrowth and co-expression of RAGE and amphoterin in the developing nervous system. J Biol Chem 270:25752–25761
Taguchi A, Blood DC, del Toro G et al (2000) Blockade of amphoterin/RAGE signalling suppresses tumor growth and metastases. Nature 405:354–360
Yan SD, Chen X, Fu J et al (1996) RAGE and amyloid-β peptide neurotoxicity in Alzheimer’s disease. Nature 382:685–691
Yan SD, Zhu H, Zhu A et al (2000) Receptor-dependent cell stress and amyloid accumulation in systemic amyloidosis. Nat Med 6:643–651
Chavakis T, Bierhaus A, Al-Fakhri N (2003) The pattern recognition receptor (RAGE) is a counterreceptor for leukocyte integrins: a novel pathway for inflammatory cell recruitment. J Exp Med 198:1507–1515
Brett J, Schmidt AM, Zou YS et al (1993) Tissue distribution of the receptor for advanced glycation endproducts (RAGE): expression in smooth muscle, cardiac myocytes, and neural tissue in addition to the vasculature. Am J Pathol 143:1699–1712
Kislinger T, Fu C, Huber B et al (1999) Ne (carboxymethyl)lysine modifications of proteins are ligands for RAGE that activate cell signalling pathways and modulate gene expression. J Biol Chem 274:31740–31749
Brownlee M (1992) Nonenzymatic glycosylation of macromolecules. Prospects of pharmacologic modulation. Diabetes 41(Suppl 2):57–60
Harja E, Bu DX, Hudson BI et al (2008) Vascular and inflammatory stresses mediate atherosclerosis via RAGE and its ligands in Apo E−/− mice. J Clin Invest 118:183–194
Bucciarelli LG, Ananthakrishnan R, Hwang YC et al (2008) RAGE and modulation of ischemic injury in the diabetic myocardium. Diabetes 57:1941–1951
Wautier MP, Chappey O, Corda S et al (2001) Activation of NADPH oxidase by AGEs links oxidant stress to altered gene expression via RAGE. Am J Physiol Endocrinol Metab 280:E685–E694
Kalousova M, Zima T, Tesar V et al (2005) Advanced glycoxidation end products in chronic diseases—clinical chemistry and genetic background. Mut Res 579:37–46
Witko-Sarsat M, Nguyen-Khoa T, Jungers P et al (1999) Advanced oxidation protein products as novel molecular basis of oxidative stress in uremia. Nephrol Dial Transplant 14(Suppl 1):76–78
Woods AA, Linton SM, Davies MJ (2003) Detection of HOCl-mediated protein oxidation products in the extracellular matrix of human atherosclerotic plaques. Biochem J 370:729–735
Kaneda H, Taguchi J, Ogasawara K et al (2002) Increased level of advanced oxidation protein products in patients with coronary artery disease. Atherosclerosis 162:221–225
Thornalley PJ (2007) Dietary AGEs and ALEs and risk to human health by their interaction with the receptor for advanced glycation endproducts (RAGE)—an introduction. Mol Nutr Food Res 51:1107–1110
Rahbar S (2007) Novel inhibitors of glycation and AGE formation. Cell Biochem Biophys 48:147–157
Donato R (2007) RAGE: a single receptor for several ligands and different cellular responses: the case of certain S100 proteins. Curr Mol Med 7:711–724
Ghavami S, Rashedi I, Dattilo BM et al (2008) S100A8/A9 at low concentration promotes tumor cell growth via RAGE ligation and MAP kinase-dependent pathway. J Leukoc Biol 83:1484–1492
Turovskaya O, Foell D, Sinha P et al (2008) RAGE, carboxylated glycans and S100A8/A9 play essential roles in colitis-associated carcinogenesis. Carcinogenesis 29:2035–2043
Fuentes MK, Nigavekar SS, Arumugam T et al (2007) RAGE activation by S100P in colon cancer stimulates growth, migration and cell signaling pathways. Dis Col Rectum 50:1230–1240
Scaffidi P, Misteli T, Bianchi ME (2002) Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature 418:191–195
Ellerman JE, Brown CK, de Vera M et al (2007) Masquerader: high mobility group box-1 and cancer. Clin Cancer Res 13:2836–2848
Porto A, Palumbo R, Pieroni M et al (2006) Smooth muscle cells in human atherosclerotic plaques secrete and proliferate in response to high mobility group box 1 protein. FASEB J 20:2565–2566
Inoue K, Kawahara K, Biswas KK et al (2007) HMGB1 expression by activated vascular smooth muscle cells in advanced human atherosclerosis plaques. Cardiovasc Pathol 16:136–143
Kalinina N, Agrotis A, Antropova Y et al (2004) Increased expression of the DNA-binding cytokine HMGB1 in human atherosclerotic lesions: role of activated macrophages and cytokines. Arterioscler Thromb Vasc Biol 24:2320–2325
Muhammad S, Barakat W, Stoyanov S et al (2008) The HMGB1 receptor RAGE mediates ischemic brain damage. J Neurosci 28:12023–12031
Andrassy M, Volz HC, Igwe JC et al (2008) High mobility group box-1 in ischemia–reperfusion injury of the heart. Circ 117:3216–32226
Arancio O, Zhang HP, Chen X et al (2004) RAGE potentiates Abeta-induced perturbation of neuronal function in transgenic mice. EMBO J 23:4096–4105
Li YM, Mitsuhashi T, Wojciechowicz D et al (1996) Molecular identity and cellular distribution of advanced glycation endproduct receptors: relationship of p60 to OST-48 and p90 to 80 K-H membrane proteins. Proc Natl Acad Sci USA 93:11047–11052
Ohgami N, Nagai R, Ikemoto M et al (2002) CD36 serves a receptor for advanced glycation endproducts (AGEs). J Diabetes Complicat 16:56–59
Horiuchi S, Sakamoto Y, Sakai M (2003) Scavenging receptors for oxidized and glycated proteins. Amino Acids 25:283–292
Lu C, He JC, Cai W et al (2004) Advanced glycation endproduct (AGE) receptor 1 is a negative regulator of the inflammatory response to AGE in mesangial cells. Proc Natl Acad Sci USA 101:11767–11772
McFarlane S, Glenn JV, Lichanska AM et al (2005) Characterization of the advanced glycation endproduct receptor complex in the retinal pigment epithelium. Br J Ophthalmol 89:107–112
Tian J, Avalos AM, Mao SY et al (2007) Toll-like receptor 9-dependent activation by DNA-containing immune complexes is mediated by HMGB1 and RAGE. Nat Immunol 8:487–496
Yu M, Wang H, Ding A et al (2006) HMGB1 signals through toll-like receptor (TLR) 4 and TLR2. Shock 26:174–179
Wendt TM, Tanji N, Guo J et al (2003) RAGE drives the development of glomerulosclerosis and implicates podocyte activation in the pathogenesis of diabetic nephropathy. Am J Pathol 162:1123–1137
Miyata T, Hori O, Zhang J et al (1996) The receptor for advanced glycation end products (RAGE) is a central mediator of the interaction of AGE-β2 microglobulin with human mononuclear phagocytes via an oxidant-sensitive pathway. Implications for the pathogenesis of dialysis-related amyloidosis. J Clin Invest 98:1088–1094
Andrassy M, Igwe J, Autschbach F et al (2006) Posttranslationally modified proteins as mediators of sustained intestinal inflammation. Am J Path 169:1223–1237
Ostendorp T, Leclerc E, Galichet A et al (2007) Structural and functional insights into RAGE activation by multimeric S100B. EMBO J 26:3868–3878
Xie J, Burz DS, He W et al (2007) Hexameric calgranulin C (S100A12) binds to the receptor for advanced glycated end products (RAGE) using symmetric hydrophobic target-binding patches. J Biol Chem 282:4218–4231
Leclerc E, Fritz G, Weibel M et al (2007) S100B and S100A6 differentially modulate cell survival by interacting with distinct RAGE immunoglobulin domains. J Biol Chem 282:31317–31331
Xie J, Reverdatto S, Frolov A et al (2008) Structural basis for pattern recognition by the receptor for advanced glycation end products (RAGE). J Biol Chem 283:27255–27269
Burke AP, Kolodgie FD, Zieske A et al (2004) Morphologic findings of coronary atherosclerotic plaques in diabetics: a postmortem study. Arterioscler Thromb Vasc Biol 24:1266–1271
Yonekura H, Sakurai S, Petrova RG et al (2003) Novel splice variants of the receptor for advanced glycation end-products expressed in human vascular endothelial cells and pericytes, and their putative roles in diabetes-induced vascular injury. Biochem J 370:1097–1109
Hudson BI, Carter AM, Harja E et al (2008) Identification, classification, and expression of RAGE gene splice variants. FASEB J 22:1572–1580
Raucci A, Cugusi S, Antonelli A et al (2008) A soluble form of the receptor for advanced glycation endproducts (RAGE) is produced by proteolytic cleavage of the membrane-bound form by the sheddase a disintegrin and metalloprotease 10 (ADAM10). FASEB J 22:3716–3727
Falcone C, Emanuele E, D’Angelo A et al (2005) Plasma levels of soluble receptor for advanced glycation endproducts and coronary artery disease in nondiabetic men. Arterioscler Thromb Vasc Biol 25:1032–1037
Katakami N, Matsuhisa M, Kaneto H et al (2007) Serum endogenous secretory RAGE levels are inversely associated with carotid IMT in type 2 diabetic patients. Atherosclerosis 190:22–23
Koyama H, Shoji T, Fukumoto S et al (2007) Low circulating endogenous secretory receptor for AGEs predicts cardiovascular mortality in patients with end-stage renal disease. Arterioscler Thromb Vasc Biol 27:147–153
Nakamura K, Yamagishi S, Adachi H et al (2007) Serum levels of sRAGE, the soluble form of receptor for advanced glycation endproducts, are associated with inflammatory markers in patients with type 2 diabetes. Mol Med 13:185–189
Nakamura K, Yamagishi S, Adachi H et al (2008) Serum levels of receptor for advanced glycation endproducts (sRAGE) are positively associated with circulating AGEs and soluble form of VCAM-1 in patients with type 2 diabetes. Microvasc Res 76:52–56
Koyama Y, Takeishi Y, Suzuki S et al (2008) Soluble receptor for advanced glycation endproducts is a prognostic factor for heart failure. J Card Fail 14:133–139
Falcone C, Campo I, Emanuele E et al (2005) −374T/A polymorphism of the RAGE gene promoter in relation to severity of coronary atherosclerosis. Clin Chim Acta 354:111–116
Falcone C, Geroldi D, Buzzi MP et al (2008) The -374T/A RAGE polymorphism protects against future cardiac events in nondiabetic patients with coronary artery disease. Arch Med Res 39:320–325
Hofmann MA, Yang Q, Harja E et al (2005) The RAGE Gly82Ser polymorphism is not associated with cardiovascular disease in the Framingham offspring study. Atherosclerosis 182:301–305
Park L, Raman KG, Lee KJ et al (1998) Suppression of accelerated diabetic atherosclerosis by the soluble receptor for advanced glycation endproducts. Nat Med 4:1025–1031
Wendt T, Harja E, Bucciarelli L et al (2006) RAGE modulates vascular inflammation and atherosclerosis in a murine model of type 2 diabetes. Atherosclerosis 185:70–77
Vikramadithyan RK, Hu Y, Noh HL et al (2005) Human aldose reductase expression accelerates diabetic atherosclerosis in transgenic mice. J Clin Invest 115:2434–2443
Bucciarelli LG, Wendt T, Qu W et al (2002) RAGE blockade stabilizes established atherosclerosis in diabetic apolipoprotein E null mice. Circ 106:2827–2835
Soro-Paavonen A, Watson AM, Li J et al (2008) RAGE deficiency attenuates the development of atherosclerosis in diabetes. Diabetes 57:2461–2469
Higashi T, Sano H, Saishoji T et al (1997) The receptor for advanced glycation end products mediates the chemotaxis of rabbit smooth muscle cells. Diabetes 46:463–472
Wang R, Kudo M, Yokoyama M et al (2001) Roles of advanced glycation end products (AGE) and receptor for AGE on vascular smooth muscle cell growth. J Nippon Med Scho 68:472–481
Lander HM, Tauras JM, Ogiste JS et al (1997) Activation of the receptor for advanced glycation end products triggers a p21(ras)-dependent mitogen-activated protein kinase pathway regulated by oxidant stress. J Biol Chem 272:17810–17814
Reddy MA, Li SL, Sahar S et al (2006) Key role of Src kinase in S100B-induced activation of the receptor for advanced glycation endproducts in vascular smooth muscle cells. J Biol Chem 281:13685–13693
Shaw SS, Schmidt AM, Banes AK et al (2003) S100B-RAGE-mediated augmentation of angiotensin II-induced activation of Jak2 in vascular smooth muscle cells is dependent on PLD2. Diabetes 52:2381–2388
Zhou Z, Wang K, Penn MS et al (2003) Receptor for AGE (RAGE) mediates neointimal formation in response to arterial injury. Circ 107:2238–2243
Sakaguchi T, Yan SF, Yan SD (2003) Arterial restenosis: central role of RAGE-dependent neointimal expansion. J Clin Invest 111:959–972
Wang K, Zhou Z, Zhang M et al (2006) Peroxisome proliferator-activated receptor gamma down-regulates receptor for advanced glycation end products and inhibits smooth muscle cell proliferation in a diabetic and nondiabetic rat carotid artery injury model. J Pharmacol Exp Ther 317:37–43
Yoshida T, Yamagishi S, Makamura K et al (2006) Telmisartan inhibits AGE-induced C-reactive protein production through downregulation of the receptor for AGE via peroxisome proliferators-activated receptor-gamma activation. Diabetologia 49:3094–3099
Chang JS, Wendt T, Qu W et al (2008) Oxygen deprivation triggers upregulation of early growth response-1 by the receptor for advanced glycation endproducts. Circ Res 102:905–913
Tanji N, Markowitz GS, Fu C et al (2000) The expression of advanced glycation endproducts and their cellular receptor RAGE in diabetic nephropathy and non-diabetic renal disease. J Am Soc Nephrol 11:1656–1666
Abel M, Ritthaler U, Zhang Y et al (1995) Expression of receptors for advanced glycosylated end products in renal disease. Nephrol Dial Transplant 19:1662–1667
Hou FF, Ren H, Owen WF Jr et al (2004) Enhanced expression of receptor for advanced glycation endproducts in chronic kidney disease. J Am Soc Nephrol 15:1889–1896
Suzuki D, Toyoda M, Yamamoto N et al (2006) Relationship between the expression of advanced glycation endproducts (AGE) and the receptor for AGE (RAGE) mRNA in diabetic nephropathy. Intern Med 45:435–441
Kalousova M, Hodkova M, Kazderova M et al (2006) Soluble receptor for advanced glycation endproducts in patients with decreased renal function. Am J Kidney Dis 47:406–411
Kalousova M, Jachymova M, Mestek O et al (2007) Receptor for advanced glycation endproducts—soluble form and gene polymorphisms in chronic haemodialysis patients. Nephrol Dial Transplant 22:2020–2026
Lindholm E, Bakhtadze E, Sjogren M et al (2006) The −374 T/A polymorphism in the gene encoding RAGE is associated with diabetic nephropathy and retinopathy in type 1 diabetic patients. Diabetologia 49:2745–2755
Prevost G, Fajardy I, Besmond C et al (2005) Polymorphisms of the receptor of advanced glycation endproducts (RAGE) and the development of nephropathy in type 1 diabetic patients. Diabetes Metabol 31:35–39
Rudofsky G Jr, Isermann B, Schilling T et al (2004) A 63 bp deletion in the promoter of RAGE correlates with a decreased risk for nephropathy in patients with type 2 diabetes. Exp Clin Endocrinol Diabetes 112:135–141
Matsunaga-Irie S, Maruyama T, Yamamoto Y et al (2004) Relation between development of nephropathy and the p22phox C242T and receptor for advanced glycation end product G1704T gene polymorphisms in type 2 diabetic patients. Diabetes Care 27:303–307
Pettersson-Fernholm K, Forsblom C, Hudson BI et al (2003) The functional −374T/A RAGE polymorphism is associated with proteinuria and cardiovascular disease in type 1 diabetic patients. Diabetes 52:891–894
Poirier O, Nicaud V, Vionnet N et al (2001) Polymorphism screening of four genes encoding advanced glycation endproduct putative receptors. Association study with nephropathy in type 1 diabetic patients. Diabetes 50:1214–1218
Kankova K, Stejskalova A, Hertlova M et al (2005) Haplotype analysis if the RAGE gene: identification of a haplotype marker for diabetic nephropathy in type 2 diabetes mellitus. Nephrol Dial Transplant 20:1093–1102
Forbes JM, Thorpe SR, Thallas-Bonke V et al (2005) Modulation of soluble receptor for advanced glycation end products by angiotensin-converting enzyme-1 inhibition in diabetic nephropathy. J Am Soc Nephrol 16:2363–2372
Nakamura K, Yamaghisi S, Nakamura Y et al (2005) Telmisartan inhibits expression of a receptor for advanced glycation end products (RAGE) in angiotensin II-exposed endothelial cells and decreases serum levels of soluble RAGE in patients with essential hypertension. Microvasc Res 70:137–141
Myint KM, Yamamoto Y, Doi T et al (2006) RAGE control of diabetic nephropathy in a mouse model: effects of RAGE gene disruption and administration of low-molecular weight heparin. Diabetes 55:2510–2522
Jensen LJ, Denner L, Schrijvers BF et al (2006) Renal effects of a neutralizing RAGE antibody in long-term streptozotocin-diabetic mice. J Endocrinol 188:493–501
Flyvbjerg A, Denner L, Schrijvers BF et al (2004) Long-term effects of a neutralizing RAGE antibody in obese type 2 diabetic mice. Diabetes 53:166–172
Bohlender JM, Franke S, Stein G et al (2005) Advanced glycation end products and the kidney. Am J Physiol Renal Physiol 289:F645–F659
Guo J, Ananthakrishnan R, Qu W et al (2008) RAGE mediates podocyte injury in adriamycin-induced glomerulosclerosis. J Am Soc Nephrol 19:961–972
Gu L, Hagiwara S, Fan Q et al (2006) Role of receptor for advanced glycation end-products and signalling events in advanced glycation end-product-induced monocyte chemoattractant protein-1 expression in differentiated mouse podocytes. Nephrol Dial Transplant 21:299–313
Oldfield MD, Bach LA, Forbes JM et al (2001) Advanced glycation end products cause epithelial-myofibroblast transdifferentiation via RAGE. J Clin Invest 108:1853–1863
Bierhaus A, Haslbeck KM, Humpert PM et al (2004) Loss of pain perception in diabetes is dependent on a receptor of the immunoglobulin superfamily. J Clin Invest 114:1741–1751
Wada R, Yagahiashi S (2005) Role of advanced glycation end products and their receptors in development of diabetic neuropathy. Ann NYAcad Sci 1043:598–604
Del Carro U, Fiorina P, Amadio S et al (2007) Evaluation of polyneuropathy markers in type 1 diabetic kidney transplant patients and effects of islet transplantation: neurophysiological and skin biopsy longitudinal analysis. Diabetes Care 30:3063–3069
Humpert PM, Papadopoulos G, Schaefer K et al (2007) sRAGE and esRAGE are not associated with peripheral or autonomic neuropathy in type 2 diabetes. Horm Metab Res 39:899–902
Toth C, Rong LL, Yang C et al (2008) Receptor for advanced glycation endproducts (RAGE) and experimental diabetic neuropathy. Diabetes 57:1002–1017
Rong LL, Trojaborg W, Qu W et al (2004) Antagonism of RAGE suppresses peripheral nerve regeneration. FASEB J 18:1812–1817
Rong LL, Yan SF, Wendt T et al (2004) RAGE modulates peripheral nerve regeneration via recruitment of both inflammatory and axonal outgrowth pathways. FASEB J 18:1818–1825
Wang L, Li S, Jungalwala FB (2008) Receptor for advanced glycation end products (RAGE) mediates neuronal differentiation and neurite outgrowth. J Neurosci Res 86:1254–1266
Collison KS, Parhar RS, Saleh SS et al (2002) RAGE-mediated neutrophil dysfunction is evoked by advanced glycation endproducts (AGEs). J Leukoc Biol 71:433–444
Schmidt AM, Yan SD, Brett J et al (1993) Regulation of human mononuclear phagocyte migration by cell surface-binding proteins for advanced glycation end products. J Clin Invest 91:2155–2168
Chen Y, Yan SS, Colgan J et al (2004) Blockade of the late stages of autoimmune diabetes by inhibition of the receptor for advanced glycation endproducts. J Immunol 173:1399–1405
Cataldegirmen G, Zeng S, Feirt N et al (2005) RAGE limits regeneration after massive liver injury by coordinated suppression of TNF-alpha and NF-kappaB. J Exp Med 201:473–484
Foell D, Frosch M, Sorg C et al (2004) Phagocyte-specific calcium-binding S100 proteins as clinical laboratory markers of inflammation. Clin Chim Acta 344:37–51
Kosaki A, Hasegawa T, Kimura T et al (2004) Increased plasma S100A12 (EN-RAGE) levels in patients with type 2 diabetes. J Clin Endocrinol Metab 89:5423–5428
Hatada T, Wada H, Nobori T et al (2005) Plasma concentrations and importance of high mobility group box protein in the prognosis of organ failure in patients with disseminated intravascular coagulation. Thromb Haemost 94:975–979
Hofmann MA, Drury S, Hudson BI et al (2002) RAGE and arthritis. The G82S polymorphism amplifies the inflammatory response. Genes Immun 3:123–135
Moser B, Szabolcs MJ, Ankersmit HJ et al (2007) Blockade of RAGE suppresses alloimmune reactions in vitro and delays allograft rejection in heart transplantation. Am J Transplant 7:293–302
Yan SSD, Wu ZY, Zhang HP et al (2003) Suppression of experimental autoimmune encephalomyelitis by selective blockade of encephalitogenic T-cell infiltration of the central nervous system. Nat Med 9:287–293
Chen Y, Akirav EM, Chen W et al (2008) RAGE ligation affects T cell activation and controls T cell differentiation. J Immunol 181:4272–4278
Moser B, Desai DD, Downie MP et al (2007) Receptor for advanced glycation end products expression on T cells contributes to antigen-specific cellular expansion in vivo. J Immunol 179:8051–8058
Diamanti-Kandarakis E, Piperi C, Patsouris E et al (2007) Immunohistochemical localization of advanced glycation endproducts (AGEs) and their receptor (RAGE) in polycystic and normal ovaries. Histochem Cell Biol 127:581–589
Diamanti-Kandarakis E, Katsikis I, Piperi C et al (2008) Increased serum advanced glycation end products is a distinct finding in lean women with polycystic ovary syndrome (PCOS). Clin Endocrinol (Oxf) 69:634–641
Liliensiek B, Weigand MA, Bierhaus A et al (2004) Receptor for advanced glycation end products (RAGE) regulates sepsis but not the adaptive immune response. J Clin Invest 113:1641–1650
Lutterloh EC, Opal SM, Pittman DD et al (2007) Inhibition of the RAGE products increases survival in experimental models of severe sepsis and systemic infection. Crit Care 11:R122
Acknowledgments
The authors gratefully acknowledge funding from the United States Public Health Service and the Juvenile Diabetes Research Foundation. AMS is a recipient of a JDRF Scholar Award. The authors are grateful to Ms. Latoya Woods for her excellent assistance in the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yan, S.F., Ramasamy, R. & Schmidt, A.M. Receptor for AGE (RAGE) and its ligands—cast into leading roles in diabetes and the inflammatory response. J Mol Med 87, 235–247 (2009). https://doi.org/10.1007/s00109-009-0439-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-009-0439-2