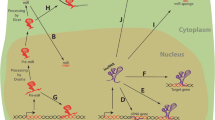
Abstract
Endogenous antisense RNA has been detected for a range of eukaryotic genes and now appears to be a common phenomenon in mammalian cells. Its abundance compared to levels of its complementary sense mRNA indicates that antisense RNA may be involved in posttrancriptional regulation of a gene. In general a downregulating effect on gene expression has been demonstrated or suggested. Due to the heterogeneity in origin and character of different antisense transcripts alternative functions such as stabilizing the corresponding sense transcript and being part of gene recombination must be considered. Regulation by endogenous antisense RNA has been shown for a plethora of genes, including cardiac genes, such as myosin heavy chainMHC, atrial light chain, and troponin I. There is now growing evidence that antisense transcription is involved in human disease, and it is reasonable to consider antisense as a target for intervention procedures. Here we review the progress in our understanding of as well as the controversies arising from investigating the regulatory mechanisms of antisense RNA, with special focus on cardiac genes. Finally, links between antisense transcription and heart disease and the possible use of antisense as a target of cardiac intervention procedures are discussed.



Similar content being viewed by others
Abbreviations
- ALC :
-
Atrial light chain
- bFGF :
-
Basic fibroblast growth factor
- cTnI :
-
Cardiac-specific troponin I
- ds :
-
Double-stranded
- MLC :
-
Myosin light chain
- MHC :
-
Myosin heavy chain
- RACE :
-
Rapid amplification of cDNA ends
- RNAi :
-
RNA interference
- VLC :
-
Ventricular light chain
References
Yelin R, Dahary D, Sorek R, Levanon EY, Goldstein O, Shoshan A, Diber A, Biton S, Tamir Y, Khosravi R, Nemzer S, Pinner E, Walach S, Bernstein J, Savitsky K, Rotman G (2003) Widespread occurrence of antisense transcription in the human genome. Nat Biotechnol 21:379–386
Wagner EG, Simons RW (1994) Antisense RNA control in bacteria, phages, and plasmids. Annu Rev Microbiol 48:713–742
Dolfini S, Consonni G, Mereghetti M, Tonelli C (1993) Antiparallel expression of the sense and antisense transcripts of maize alpha-tubulin genes. Mol Gen Genet 241–:161–169
Werner A, Preston-Fayers K, Dehmelt L, Nalbant P (2002) Regulation of the NPT gene by a naturally occurring antisense transcript. Cell Biochem Biophys 36:241–252
Kimelman D, Kirschner MW (1989) An antisense mRNA directs the covalent modification of the transcript encoding fibroblast growth factor in Xenopus oocytes. Cell 59:687–696
Heywood SM, Kennedy DS, Bester AJ (1975) Studies concerning the mechanism by which translational-control RNA regulates protein synthesis in embryonic muscle. Eur J Biochem 58:587–593
Murphy PR, Knee RS (1994) Identification and characterization of an antisense RNA transcript (gfg) from the human basic fibroblast growth factor gene. Mol Endocrinol 8:852–859
Armstrong B, Krystal G (1992) Isolation and characterization of complementary DNA for N-myc, a gene encoded by the DNA strand opposite to N-myc. Cell Growth Differ 3:385–390
Zezza DJ, Heywood SM (1986) Analysis of tcRNA102 associated with myosin heavy chain-mRNPs in control and dystrophic chick pectoralis muscle. J Biol Chem 261:7461–7645
McCarthy TL, Siegel E, Mroczkowski B, Heywood SM (1983) Characterization of translational-control ribonucleic acid isolated from embryonic chick muscle. Biochemistry 22:935–941
Simons RW (1994) The control of prokaryotic and eukaryotic gene expression by naturally occurring antisense RNA. In: Crooke ST, LeBlew B (eds) Antisense research and applications. CRC, Boca Raton, pp 97–124
Vanhee-Brossollet C, Vaquero C (1998) Do natural antisense transcripts make sense in eukaryotes? Gene 211:1–9
Kim JH, McLinden RJ, Mosca JD, Vahey MT, Greene WC, Redfield RR (1996) Inhibition of HIV replication by sense and antisense rev response elements in HIV-based retroviral vectors. J Acquir Immune Defic Syndr Hum Retrovirol 12:343–351
Sharma HW, Narayanan R (1995) The therapeutic potential of antisense oligonucleotides. Bioessays 17:1055–1063
Krystal GW, Armstrong BC, Battey JF (1990) N-myc mRNA forms an RNA-RNA duplex with endogenous antisense transcripts. Mol Cell Biol 10:4180–4191
Podlowski S, Bramlage P, Baumann G, Morano I, Luther HP (2002) Cardiac troponin I sense-antisense RNA duplexes in the myocardium. J Cell Biochem 85:198–207
Fedoroff N, Wellauer PK, Wall R (1977) Intermolecular duplexes in heterogeneous nuclear RNA from HeLa cells. Cell 10:597–610
Kumar M, Carmichael GG (1998) Antisense RNA: function, fate of duplex RNA in cells of higher eukaryotes. Microbiol Mol Biol Rev 62:1415–1434
Wu H, MacLeod AR, Lima WF, Crooke ST (1998) Identification and partial purification of human double strand RNase activity. A novel terminating mechanism for oligoribonucleotide antisense drugs. J Biol Chem 273:2532–2542
Bass BL, Weintraub H (1988) An unwinding activity that covalently modifies its double-stranded RNA substrate. Cell 55:1089–1098
Kumar M, Carmichael GG (1997) Nuclear antisense RNA induces extensive adenosine modifications and nuclear retention of target transcripts. Proc Natl Acad Sci USA 94:3542–3547
Proud CG (1995) PKR: a new name and new roles Trends Biochem Sci 20:241–246
Kerr IM, Brown RE (1978) pppA2’p5’A2’p5’A: an inhibitor of protein synthesis synthesized with an enzyme fraction from interferon-treated cells Proc Natl Acad Sci USA 75:256–260
Wu H, Xu H, Miraglia LJ, Crooke ST (2000) Human RNase III is a 160-kDa protein involved in preribosomal RNA processing. J Biol Chem 275:36957–36965
Crooke ST, Bennett CF (1996) Progress in antisense oligonucleotide therapeutics. Annu Rev Pharmacol Toxicol 36:107–129
Hildebrandt M, Nellen W (1992) Differential antisense transcription from the Dictyostelium EB4 gene locus: implications on antisense-mediated regulation of mRNA stability. Cell 69:197–204
Merino E, Balbas P, Puente J, Bolivar F (1994) Antisense overlapping openreading frames in genes from bacteria to humans. Nucleic Acids Res 22:1903–1908
Fire A (1999) RNA-triggered gene silencing. Trends Genet 15:358–363
Korneev SA, Park JH, O’Shea M (1999) Neuronal expression of neural nitric oxide synthase (nNOS) protein is suppressed by an antisense RNA transcribed from an NOS pseudogene. J Neurosci 19:7711–7720
Lewin B (1990) Genes IV. Oxford University Press, New York
Tommasi S, Pfeifer GP (1999) In vivo structure of two divergent promoters at the human PCNA locus. Synthesis of antisense RNA and S phase-dependent binding of E2F complexes in intron 1. J Biol Chem 274:27829–27838
Volloch V, Schweitzer B, Rits S (1996) Antisense globin RNA in mouse erythroid tissues: structure, origin, and possible function. Proc Natl Acad Sci USA 93:2476–2481
Merzendorfer H, Harvey WR, Wieczorek H (1997) Sense and antisense RNA for the membrane associated 40 kDa subunit M40 of the insect V-ATPase. FEBS Lett 411:239–244
Røsok O, Sioud M (2004) Systematic identification of sense-antisense transcripts in mammalian cells. Nat Biotechnol 22:104–108
Leinwand LA, Saez L, McNally E, Nadal-Ginard B (1983) Isolation and characterization of human myosin heavy chain genes. Proc Natl Acad Sci USA 80:3716–3720
Lompre AM, Nadal-Ginard B, Mahdavi V (1984) Expression of the cardiac ventricular alpha- and beta-myosin heavy chain genes is developmentally and hormonally regulated J Biol Chem 259:6437–6446
Lompre AM, Schwartz K, d’Albis A, Lacombe G, Van Thiem N, Swynghedauw B (1979) Myosin isoenzyme redistribution in chronic heart overload. Nature 282:105–107
Boheler KR, Chassagne C, Martin X, Wisnewsky C, Schwartz K (1992) Cardiac expressions of alpha- and beta-myosin heavy chains and sarcomeric alpha-actins are regulated through transcriptional mechanisms. Results from nuclear run-on assays in isolated rat cardiac nuclei. J Biol Chem 267:12979–12985
Luther HP, Morwinski R, Wallukat G, Haase H, Morano (1997) Expression of sense and naturally occurring antisense mRNA of myosin heavy chain in rat heart tissue and cultivated cardiomyocytes. J Mol Cell Cardiol 29:27–35
Luther HP, Haase H, Hohaus A, Beckmann G, Reich J, Morano I (1998) Characterization of naturally occurring myosin heavy chain antisense mRNA in rat heart. J Cell Biochem 70:110–120
Haddad F, Bodell PW, Qin AX, Giger JM, Baldwin KM (2003) Role of antisense RNA in coordinating cardiac myosin heavy chain gene switching. J Biol Chem 278:37132–37138
Mercadier JJ, Bouveret P, Gorza L, Schiaffino S, Clark WA, Zak R, Swynghedauw B, Schwartz K (1983) Myosin isoenzymes in normal and hypertrophied human ventricular myocardium. Circ Res 53:52–62
Ritter O, Luther HP, Haase H, Baltas LG, Baumann G, Schulte HD, Morano I (1999) Expression of atrial myosin light chains but not alpha-myosin heavy chains is correlated in vivo with increased ventricular function in patients with hypertrophic obstructive cardiomyopathy. J Mol Med 77:677–685
Luther HP, Podlowski S, Hetzer R, Baumann G (2001) Analysis of sense and naturally occurring antisense transcripts of myosin heavy chain in the human myocardium J Cell Biochem 80:596–605
Nakao K, Minobe W, Roden R, Bristow M, Leinwand L (1997) Myosin heavy chain gene expression in human heart failure. J Clin Invest 100:2362–2370
Miyata S, Minobe W, Bristow M, Leinwand L (2000) Myosin heavy chain isoform expression in the failing and nonfailing human heart. Circ Res 86:386–390
Lowey S, Risby D (1971) Light chains from fast and slow muscle myosins. Nature 234:81–85
Morano M, Zacharzowski U, Maier M, Lange PE, Alexi-Meskishvili V, Haase H, Morano I (1996) Regulation of human heart contractility by essential myosin light chain isoforms. J Clin Invest 98:467–473
Sutsch G, Brunner U, von Schulthess C, Hirzel H, Hess O, Turina M, Krayenbuehl H, Schaub M (1992) Hemodynamic performance and myosin light chain-1 expression of the hypertrophied left ventricle in aortic valve disease before and after valve replacement. Circ Res 70:1035–1043
Morano I (1999) Tuning the human heart molecular motors by myosin light chains. J Mol Med 77:544–555
Ritter O, Haase H, Schulte HD, Lange PE, Morano I (1999) Remodeling of the hypertrophied human myocardium by cardiac bHLH transcription factors. J Cell Biochem 74:551–561
Bartsch H, Voigtsberger S, Baumann G, Morano I, Luther HP (2004) Detection of a novel sense-antisense RNA-hybrid structure by RACE experiments on endogenous Troponin I antisense RNA. RNA 10:1215–1224
Stein P, Svobode P, Anger M, Schultz RM (2003) RNAi: mammalian oocytes do it without RNA-dependent RNA polymerase. RNA 9:187–192
Herbert A (2004) The four Rs of RNA-directed evolution. Nat Genetics 36:19–25
Morelli, S, Delia D, Capacioli S, Quattrone, Schiavone N, Bevilacqua A, Tomasini S, Nicolin A (1997) The antisense bcl-2-IgH transcript is an optimal target for synthetic oligonucleotides. Proc Natl Acad Sci USA 94:8150–8155
Acknowledgements
Our original work on endogenous antisense RNA was supported by the Deutsche Forschungsgemeinschaft Lu 694/1-1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luther, H.P. Role of endogenous antisense RNA in cardiac gene regulation. J Mol Med 83, 26–32 (2005). https://doi.org/10.1007/s00109-004-0613-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-004-0613-5