Abstract
Background
The urokinase plasminogen activator system (uPA, uPAR, PAI‑1) is upregulated in cancer and high plasma levels are associated with poor prognosis. Their interaction with hypoxia-related osteopontin (OPN) which is also overexpressed in malignant tumors suggests potential clinical relevance. However, the prognostic role of the uPA system in the radiotherapy (RT) of non-small-cell lung cancer (NSCLC), particularly in combination with OPN, has not been investigated so far.
Methods
uPA, uPAR, PAI‑1 and OPN plasma levels of 81 patients with locally advanced or metastasized NSCLC were prospectively analyzed by ELISA before RT and were correlated to clinical patient/tumor data and prognosis after RT.
Results
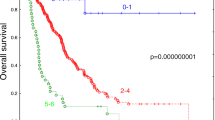
uPAR plasma levels were higher in M1; uPA and PAI‑1 levels were higher in M0 NSCLC patients. uPAR correlated with uPA (p < 0.001) which also correlated with PAI‑1 (p < 0.001). The prognostic impact of OPN plasma levels in the RT of NSCLC was previously reported by our group. PAI‑I plasma levels significantly impacted overall (OS) and progression-free survival (PFS). Low PAI‑1 levels were associated with a significantly reduced OS and PFS with a nearly 2‑fold increased risk of death (p = 0.029) and tumor progression (p = 0.029). In multivariate analysis, PAI‑1 levels remained an independent prognostic factor for OS and PFS with a 3‑fold increased risk of death (p = 0.001). If PAI‑1 plasma levels were combined with OPN or tumor volume, we found an additive prognostic impact on OS and PFS with a 2.5- to 3‑fold increased risk of death (p = 0.01).
Conclusion
Our results suggest that PAI-1 but not uPA and uPAR might add prognostic information in patients with advanced NSCLC undergoing RT. High pretreatment PAI-1 plasma levels were found predominantly in M0-stage patients and indicate a favorable prognosis as opposed to OPN where high plasma levels are associated with poor survival and metastasis. In combination, PAI-1 and OPN levels successfully predicted outcome and additively correlated with prognosis. These findings support the notion of an antidromic prognostic impact of OPN and PAI-1 plasma levels in the RT of advanced NSCLC.
Zusammenfassung
Hintergrund
Das Urokinase-Plasminogen-Aktivator-System (uPA, uPAR, PAI‑1) ist in malignen Tumoren hochreguliert und hohe Plasmaspiegel mit schlechter Prognose assoziiert. Deren Zusammenspiel mit hypoxieassoziierten Proteinen wie Osteopontin (OPN), welches ebenfalls in malignen Tumoren überexprimiert ist, legt eine potenzielle klinische Relevanz nahe. Die prognostische Bedeutung des uPA-Systems für die RT des NSCLC („non-small-cell lung cancer“), insbesondere in Kombination mit OPN, wurde bisher noch nicht untersucht.
Methoden
Bei 81 Patienten mit lokal fortgeschrittenem bzw. metastasiertem NSCLC wurden Plasmaspiegel von uPA, uPAR, PAI-1 und OPN vor RT prospektiv mittels ELISA bestimmt und mit klinischen Patienten- und Tumorparametern sowie der Prognose nach RT korreliert.
Ergebnisse
uPAR-Plasmalevel waren bei M1-Patienten höher; uPA- und PAI-1-Plasmaspiegel waren bei nichtmetastasierten Patienten (M0) erhöht. uPAR korrelierte mit uPA (p < 0,001), das ebenfalls mit PAI‑1 korrelierte (p < 0,001). Der prognostische Effekt von OPN-Plasmaspiegeln in der RT von NSCLC wurde bereits in unseren Vorarbeiten gezeigt. PAI-1-Plasmaspiegel korrelierten signifikant mit Gesamtüberleben (OS) und progressionsfreiem Überleben (PFS). Niedrige PAI-1-Spiegel waren mit reduziertem OS und PFS verbunden sowie mit einem fast 2‑fach erhöhtem Risiko zu versterben (p = 0,029) bzw. einen Tumorprogress im Verlauf zu erleiden (p = 0,029). In der multivariaten Analyse zeigten sich PAI-1-Plasmaspiegel als unabhängiger prognostischer Marker für OS und PFS, wobei bei erhöhten PAI-1-Spiegeln ein 3‑fach erhöhtes Risiko zu versterben vorlag (p = 0,001). Bei der Kombination von PAI‑1 mit OPN-Plasmaspiegeln bzw. Tumorvolumen zeigte sich ein additiver prognostischer Effekt für OS und PFS sowie ein 2,5- bis 3‑fach erhöhtes Risiko zu versterben (p = 0,01).
Schlussfolgerung
Die Ergebnisse zeigen, dass PAI-1-Plasmaspiegel im Gegensatz zu uPA- bzw. uPAR-Plasmaspiegeln zusätzliche prognostisch relevante Informationen für die RT beim fortgeschrittenen NSCLC liefern könnten. Hohe prätherapeutische PAI-1-Plasmaspiegel liegen v. a. bei nichtmetastasierten Patienten vor und zeigen eine günstige Prognose an – im Gegensatz zu hohen OPN-Plasmaspiegeln, die mit reduziertem OS nach RT und Metastasierung assoziiert sind. In der Kombination mit OPN prädizierten PAI-1-Plasmaspiegel erfolgreich das prognostische Outcome nach RT. Die Resultate sprechen für eine differenzielle prognostische Bedeutung von OPN- und PAI-1-Plasmaspiegeln bei der RT des fortgeschrittenen NSCLC.



Similar content being viewed by others
Abbreviations
- BRMS1:
-
Breast cancer metastasis suppressor gene 1
- CAIX:
-
Carbonic anhydrase IX
- CCRT:
-
Concurrent chemoradiotherapy
- CI:
-
Confidence interval
- CT:
-
Computed tomography
- EDTA:
-
Ethylenediaminetetraacetate
- ELISA:
-
Enzyme-linked immunosorbent assay
- Fx:
-
Fractions
- GTV:
-
Gross tumor volume
- M0:
-
Non-metastasized
- M1:
-
Metastasized
- mRNA:
-
Messenger RNA
- NSCLC:
-
Non-small-cell lung cancer
- OPN:
-
Osteopontin
- OS:
-
Overall survival
- P :
-
p-value
- PAI-1:
-
Plasminogen activator inhibitor-1
- PET:
-
Positron emission tomography
- PFS:
-
Progression-free survival
- RR:
-
Relative risk
- rs :
-
Spearman’s rank correlation coefficient
- RT:
-
Radiotherapy
- SABR:
-
Stereotactic ablative radiotherapy
- SD:
-
Standard deviation
- SPSS:
-
Statistical package for the social sciences
- UICC:
-
Union internationale contre le cancer
- uPA:
-
Urokinase-type plasminogen activator
- uPAR:
-
Urokinase-type plasminogen activator receptor
- VEGF:
-
Vascular endothelial growth factor
- WHO:
-
World Health Organization
References
Goldstraw P, Crowley J, Chansky K et al (2007) The IASLC Lung Cancer Staging Project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM Classification of malignant tumours. J Thorac Oncol 2:706–714
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics 2016. Ca Cancer J Clin 66:7–30
Parkin DM, Bray F, Ferlay J et al (2001) Estimating the world cancer burden: Globocan 2000. Int J Cancer 94:153–156
Worldwide cancer statistics (2015) Cancer research UK. http://www.cancerresearchuk.org/health-professional/cancer-statistics/worldwide-cancer#heading-Zero. Accessed 2 Aug 2015
Guckenberger M, Allgäuer M, Appold S et al (2013) Safety and efficacy of stereotactic body radiotherapy for stage 1 non-small-cell lung cancer in routine clinical practice: a patterns-of-care and outcome analysis. J Thorac Oncol 8:1050–1058
Pass HI, Carbone DP, Johnson DH et al (2012) Principles and practice of lung cancer: the official reference text of the International Association for the Study of Lung Cancer (IASLC). Lippincott Williams & Wilkins, Philadelphia
Semrau S, Bier A, Thierbach U et al (2007) 6‑year experience of concurrent radiochemotherapy with vinorelbine plus a platin compound in multimorbid or aged patients with inoperable non-small cell lung cancer. Strahlenther Onkol 1:30–35
Oberije C, De Ruysscher D, Houben R et al (2015) A validated prediction model for overall survival from stage III non-small cell lung cancer: toward survival prediction for individual patients. Int J Radiat Oncol Biol Phys 92:935–944
Eberhardt WE, Pöttgen C, Gauler TC et al (2015) Phase III study of surgery versus definitive concurrent chemoradiotherapy boost in patients with resectable stage IIIA(N2) and selected IIIB non-small-cell lung cancer after induction chemotherapy and concurrent chemoradiotherapy (ESPATUE). J Clin Oncol 33(35):4194–4201
Baisi A, Raveglia F, De Simone M et al (2013) Micrometastasis and skip metastasis as predictive factors in no small-cell lung cancer staging. Eur J Cardiothorac Surg 5:1075
Vaupel P, Mayer A (2007) Hypoxia in cancer: significance and impact on clinical outcome. Cancer Metastasis Rev 26:225–239
Bache M, Kappler M, Said HM et al (2008) Detection and specific targeting of hypoxic regions within solid tumors: current preclinical and clinical strategies. Curr Med Chem 15:322–338
Ostheimer C, Bache M, Güttler A et al (2014) A pilot study on potential plasma hypoxia markers in the radiotherapy of non-small cell lung cancer. Osteopontin, carbonic anhydrase IX and vascular endothelial growth factor. Strahlenther Onkol 190:276–282
Ostheimer C, Bache M, Güttler A et al (2014) Prognostic information of serial plasma osteopontin measurement in radiotherapy of non-small-cell lung cancer. Bmc Cancer 14:858
Duffy MJ, McGowan PM, Gallagher WM (2008) Cancer invasion and metastasis: changing views. J Pathol 214:283–293
McMahon B, Kwaan HC (2008) The plasminogen activator system and cancer. Pathophysiol Haemost Thromb 36:184–194
Dass K, Ahmad A, Azmi AS et al (2008) Evolving role of uPA/uPAR system in human cancers. Cancer Treat Rev 34:122–136
de Bock CE, Wang Y (2004) Clinical significance of urokinase-type plasminogen activator receptor (uPAR) expression in cancer. Med Res Rev 24:13–39
Mondino A, Blasi F (2004) uPA and uPAR in fibrinolysis, immunity and pathology. Trends Immunol 25:450–455
Duffy MJ (2004) The urokinase plasminogen activator system: role in malignancy. Curr Pharm Des 10:39–49
Brünner N, Foekens J, Harbeck N et al (2010) Characteristics of the levelof-evidence-1 disease forecast cancer biomarkers uPA and its inhibitor PAI-1. Expert Rev Mol Diagn 10:947–962
Schmitt M, Mengele K, Napieralski R et al (2010) Clinical utility of level-ofevidence-1 disease forecast cancer biomarkers uPA and its inhibitor PAI. Expert Rev Mol Diagn 10:1051–1067
Choong PF, Fernö M, Akerman M et al (1996) Urokinase-plasminogen-activator levels and prognosis in 69 soft-tissue sarcomas. Int J Cancer 69:268–272
Taubert H, Würl P, Greither T et al (2010) Codetection of members of the urokinase plasminogen activator system in tumor tissue and in serum correlates with a poor prognosis for softtissue sarcoma patients. Br J Cancer 102:731–737
He C, He P, Liu LP et al (2001) Analysis of expressions of components in the plasminogen activator system in high- and low-metastatic human lung cancer cells. J Cancer Res Clin Oncol 127:180–186
Hundsdorfer B, Zeilhofer HF, Bock KP (2005) Tumour-associated urokinase-type plasminogen activator (uPA) and its inhibitor PAI-1 in normal and neoplastic tissues of patients with squamous cell cancer of the oral cavity—clinical relevance and prognostic value. J Craniomaxillofac Surg 33:191–196
Sprague LD, Mengele K, Schilling D et al (2006) Effect of reoxygenation on the hypoxia-induced up-regulation of serine protease inhibitor PAI-1 in head and neck cancer cells. Oncology 71:282–291
Bayer C, Schilling D, Hoetzel J et al (2008) PAI-1 levels predict response to fractionated irradiation in 10 human squamous cell carcinoma lines of the head and neck. Radiother Oncol 86:361–368
Vaschetto R, Navalesi P, Clemente N et al (2015) Osteopontin induces soluble urokinase-type plasminogen activator receptor production and release. Minerva Anestesiol 81:157–165
Das R, Philip S, Mahabeleshwar GH et al (2005) Osteopontin: it’s role in regulation of cell motility and nuclear factor kappa B‑mediated urokinase type plasminogen activator expression. IUBMB Life 57:441–447
Sheng XJ, Zhou DM, Liu Q et al (2014) BRMS1 inhibits expression of NF-kappaB subunit p65, uPA and OPN in ovarian cancer cells. Eur J Gynaecol Oncol 35:236–242
Wai PY, Kuo PC (2004) The role of osteopontin in tumor metastasis. J Surg Res 121:228–241
Furger KA, Menon RK, Tuck AB et al (2001) The functional and clinical roles of osteopontin in cancer and metastasis. Curr Mol Med 1:621–632
Swartz JE, Pothen AJ, Stegeman I et al (2015) Clinical implications of hypoxia biomarker expression in head and neck squamous cell carcinoma: a systematic review. Cancer Med 4:1101–1116
Tuck AB, Hota C, Chambers AF et al (2001) Osteopontin(OPN)-induced increase in human mammary epithelial cell invasiveness is urokinase (uPA)-dependent. Breast Cancer Res Treat 70:197–204
Chen RX, Xia YH, Xue TC et al (2011) Osteopontin promotes hepatocellular carcinoma invasion by up-regulating MMP-2 and uPA expression. Mol Biol Rep 38:3671–3677
Das R, Mahabeleshwar GH, Kundu GC et al (2004) Osteopontin induces AP-1-mediated secretion of urokinase-type plasminogen activator through c‑Src-dependent epidermal growth factor receptor transactivation in breast cancer cells. J Biol Chem 279:11051–11064
Das R, Mahabeleshwar GH, Kundu GC et al (2003) Osteopontin stimulates cell motility and nuclear factor kappaB-mediated secretion of urokinase type plasminogen activator through phosphatidylinositol 3‑kinase/Akt signaling pathways in breast cancer cells. J Biol Chem 278:28593–28606
Angenete E, Langenskiöld M, Palmgren I (2009) uPA and PAI-1 in rectal cancer—relationship to radiotherapy and clinical outcome. J Surg Res 153:46–53
Salden M, Splinter TA, Peters HA et al (2000) The urokinase-type plasminogen activator system in resected non-small-cell lung cancer. Rotterdam Oncology Thoracic Study Group. Ann Oncol 11:327–332
Brünner N, Nielsen HJ, Hamers M et al (1999) The urokinase plasminogen activator receptor in blood from healthy individuals and patients with cancer. APMIS 107:160–167
Pappot H, Høyer-Hansen G, Rønne E et al (1997) Elevated plasma levels of urokinase plasminogen activator receptor in non-small cell lung cancer patients. Eur J Cancer 33:867–872
Yang SF, Hsieh YS, Lin CL et al (2005) Increased plasma levels of urokinase plasminogen activator and matrix metalloproteinase-9 in nonsmall cell lung cancer patients. Clin Chim Acta 354:91–99
Kotzsch M, Magdolen V, Greither T et al (2011) Combined mRNA expression levels of members of the urokinase plasminogen activator (uPA) system correlate with disease-associated survival of soft-tissue sarcoma patients. BMC Cancer 11:273
Harbeck N, Kates R, Gauger K et al (2004) Urokinase-type plasminogen activator (uPA) and its inhibitor PAI-1: novel tumor-derived factors with a high prognostic and predictive impact in breast cancer. Thromb Haemost 91:450–456
Castelló R, Landete JM, España F et al (2007) Expression of plasminogen activatorinhibitors type 1 and type 3 and urokinase plasminogen activator protein and mRNA in breast cancer. Thromb Res 120:753–762
Xue A, Scarlett CJ, Jackson CJ et al (2008) Prognostic significance of growth factors and the urokinase-type plasminogen activator system in pancreatic ductal adenocarcinoma. Pancreas 36:160–167
Biermann JC, Holzscheiter L, Kotzsch M et al (2008) Quantitative RT-PCR assays for the determination of urokinase-type plasminogen activator and plasminogen activator inhibitor type 1 mRNA in primary tumor tissue of breast cancer patients: comparison to antigen quantification by ELISA. Int J Mol Med 21:251–259
Schmidt U, Fuessel S, Koch R et al (2006) Quantitative multi-gene expression profiling of primary prostate cancer. Prostate 66:1521–1534
Hsu LH, Hsu PC, Liao TL et al (2016) Pleural fluid osteopontin, vascular endothelial growth factor, and urokinase-type plasminogen activator levels as predictors of pleurodesis outcome and prognosticators in patients with malignant pleural effusion: a prospective cohort study. BMC Cancer 16:463
Brown LF, Papadopoulos-Sergiou A, Berse B et al (1994) Osteopontin expression and distribution in human carcinomas. Am J Pathol 145:610–623
Hu Z, Lin D, Yuan J et al (2005) Overexpression of osteopontin is associated with more aggressive phenotypes in human non-small cell lung cancer. Clin Cancer Res 11:4646–4652
Zhao B, Sun T, Meng F et al (2011) Osteopontin as a potential biomarker of proliferation and invasiveness for lung cancer. J Cancer Res Clin Oncol 137:1061–1070
Pavey SJ, Hawson GA, Marsh NA et al (2001) Impact of the fi brinolytic enzyme system on prognosis and survival associated with non-small cell lung carcinoma. Blood Coagul Fibrinolysis 12:51–58
Nagamine Y, Medcalf RL, Muñoz-Cánoves P et al (2005) Transcriptional and posttranscriptional regulation of the plasminogen activator system. Thromb Haemost 93:661–675
Volm M, Mattern J, Koomagi R et al (1999) Relationship of urokinase and urokinase receptor in non-small cell lung cancer to proliferation, angiogenesis, metastasis and patient survival. Oncol Rep 6:611–615
Hoyer-Hansen G, Lund IK et al (2007) Urokinase receptor variants in tissue and body fluids. Adv Clin Chem 44:65–102
Nijziel MR, Van Oerle R, Hellenbrand D et al (2003) The prognostic value of the soluble urokinase-type plasminogen activator receptor (s-uPAR) in plasma of breast cancer patients with and without metastatic disease. J Thromb Haemost 1:982–986
Duffy MJ, Duggan C (2004) The urokinase plasminogen activator system: a rich source of tumour markers for the individualised management of patients with cancer. Clin Biochem 37:541–548
Halamkova J (2011) Clinical relevance of uPA, uPAR, PAI 1 and PAI 2 tissue expression and plasma PAI 1 level in colorectal carcinoma patients. Hepatogastroenterology 58:1918–1925
Kwaan HC, Mazar AP, McMahon BJ et al (2013) The apparent uPA/PAI-1 paradox in cancer: more than meets the eye. Semin Thromb Hemost 39:382–391
Almasi CE et al (2011) Urokinase receptor forms in serum from non-small cell lung cancer patients: relation to prognosis. Lung Cancer 74(3):510–515
Langenskiöld M, Holmdahl L, Angenete E et al (2009) Differential prognostic impact of uPA and PAI-1 in colon and rectal cancer. Tumour Biol 30:210–220
Käsmann L, Niyazi M, Blanck O et al (2017) Predictive and prognostic value of tumor volume and its changes during radical radiotherapy of stage III non-small cell lung cancer: a systematic review. Strahlenther Onkol. https://doi.org/10.1007/s00066-017-1221-y
Gunther S, Ostheimer C, Stangl S et al (2015) Correlation of Hsp70 serum levels with gross tumor volume and composition of lymphocyte subpopulations in patients with squamous cell and Adeno non-small cell lung cancer. Front Immunol 6:556
Ostheimer C, Gunther S, Bache M et al (2017) Dynamics of heat shock protein 70 serum levels as a predictor of clinical response in non-small-cell lung cancer and correlation with the hypoxia-related marker osteopontin. Front Immunol 8(18):1305
Funding
The authors declare that there is no external funding of the study. The study was entirely financially covered by the Department of Radiation Oncology of the Martin Luther University Halle-Wittenberg.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
C. Ostheimer, C. Evers, M. Bache, T. Reese and D. Vordermark declare that they have no competing interests.
Additional information
C. Ostheimer and C. Evers contributed equally to the manuscript and shared first authorship.
Rights and permissions
About this article
Cite this article
Ostheimer, C., Evers, C., Bache, M. et al. Prognostic implications of the co-detection of the urokinase plasminogen activator system and osteopontin in patients with non-small-cell lung cancer undergoing radiotherapy and correlation with gross tumor volume. Strahlenther Onkol 194, 539–551 (2018). https://doi.org/10.1007/s00066-017-1255-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-017-1255-1
Keywords
- Non-small-cell lung cancer (NSCLC)
- Radiotherapy
- Biomarker
- Plasminogen-activator inhibitor 1 (PAI-1)
- Urokinase plasminogen activator (uPA)
- Urokinase plasminogen activator receptor (uPAR)
- Prognosis