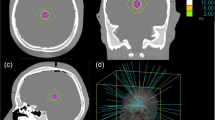
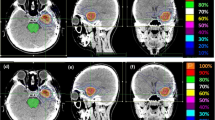
Abstract
Background and purpose
Stereotactic radiosurgery with an adapted linear accelerator (linac-SRS) is an established therapy option for brain metastases, benign brain tumors, and arteriovenous malformations. We intended to investigate whether the dosimetric quality of treatment plans achieved with a CyberKnife (CK) is at least equivalent to that for linac-SRS with circular or micromultileaf collimators (microMLC).
Patients and methods
A random sample of 16 patients with 23 target volumes, previously treated with linac-SRS, was replanned with CK. Planning constraints were identical dose prescription and clinical applicability. In all cases uniform optimization scripts and inverse planning objectives were used. Plans were compared with respect to coverage, minimal dose within target volume, conformity index, and volume of brain tissue irradiated with ≥ 10 Gy.
Results
Generating the CK plan was unproblematic with simple optimization scripts in all cases. With the CK plans, coverage, minimal target volume dosage, and conformity index were significantly better, while no significant improvement could be shown regarding the 10 Gy volume. Multiobjective comparison for the irradiated target volumes was superior in the CK plan in 20 out of 23 cases and equivalent in 3 out of 23 cases. Multiobjective comparison for the treated patients was superior in the CK plan in all 16 cases.
Conclusion
The results clearly demonstrate the superiority of the irradiation plan for CK compared to classical linac-SRS with circular collimators and microMLC. In particular, the average minimal target volume dose per patient, increased by 1.9 Gy, and at the same time a 14 % better conformation index seems to be an improvement with clinical relevance.
Zusammenfassung
Hintergrund und Zielsetzung
Stereotaktische Radiochirurgie mit einem adaptierten Linearbeschleuniger (Linac-SRS) ist eine erfolgreiche und etablierte Therapieoption für Hirnmetastasen, benigne Hirntumoren und arteriovenöse Malformationen. Ziel war es, zu untersuchen, ob die mit einem CyberKnife (CK) erreichbare dosimetrische Planqualität mindestens gleichwertig ist wie bei der Linac-SRS mit Rundkollimatoren und mit Mikro-Multileafkollimatoren (microMLC).
Patienten und Methoden
Eine repräsentative Stichprobe von 16 Patienten mit 23 Zielvolumen, die mit Linac-SRS behandelt wurden, wurde am CK nachgeplant. Randbedingungen waren gleiche Dosisverschreibung und klinische Applizierbarkeit. In allen Fällen wurden einheitliche Optimierungsskripte und Optimierungsziele verwendet. Der Planvergleich erfolgte im Rahmen der multikriteriellen Entscheidungstheorie. Entscheidungskriterien waren Coverage, minimale Dosis im Zielvolumen, Konformitätsindex und Volumen des mit mehr als 10 Gy bestrahlten Hirngewebes.
Ergebnisse
Die Erzeugung der CK-Pläne war in allen Fällen ohne Probleme mit einfachen Optimierungsskripten möglich. Bei den CK-Plänen waren Coverage, minimale Zielvolumendosis und Konformitätsindex signifikant und das 10-Gy-Volumen nicht signifikant besser als bei den Linac-SRS-Plänen. Der multikriterielle Vergleich für die bestrahlten Zielvolumen zeigte eine Überlegenheit der CK-Pläne in 20 von 23 Fällen und in 3 von 23 Fällen eine Gleichwertigkeit. Der multikriterielle Vergleich für die behandelten Patienten ergab eine Überlegenheit der CK-Pläne in allen 16 Fällen.
Schlussfolgerung
Im Ergebnis zeigt der Vergleich die deutliche Überlegenheit der Bestrahlungspläne für das CK gegenüber der klassischen Linac-SRS mit Rundkollimatoren und auch mit microMLC. Speziell die im Mittel pro Patient um 1,9 Gy höhere minimale Zielvolumendosis bei gleichzeitig um 14 % besserem Konformitätsindex erscheint als Verbesserung von klinischer Relevanz.


Similar content being viewed by others
References
Adler JR Jr, Chang SD, Murphy MJ, Doty J, Geis P, Hancock SL (1997) The Cyberknife: a frameless robotic system for radiosurgery. Stereotact Funct Neurosurg 69:124–128
Antypas C, Pantelis E (2008) Performance evaluation of a CyberKnife G4 image-guided robotic stereotactic radiosurgery system. Phys Med Biol 53:4697–4718
Blamek S, Grządziel A, Miszczyk L (2013) Robotic radiosurgery versus micro-multileaf collimator: a dosimetric comparison for large or critically located arteriovenous malformations. Radiat Oncol 8:205
Bortfeld T, Oelfke U, Nill S (2000) What is the optimum leaf width of a multileaf collimator? Med Phys 27:2494–2502
Boström JP, Meyer A, Pintea B, Gerlach R, Surber G, Lammering G, Hamm K (2014) Risk-adapted single or fractionated stereotactic high-precision radiotherapy in a pooled series of nonfunctioning pituitary adenomas: High local control and low toxicity. Strahlenther Onkol. 2014 Aug 5. [Epub ahead of print]
Cetin IA, Ates R, Dhaens J, Storme G (2012) Retrospective analysis of linac-based radiosurgery for arteriovenous malformations and testing of the Flickinger formula in predicting radiation injury. Strahlenther Onkol 188:1133–1138
Correa SF, Marta GN, Teixeira MJ (2014) Neurosymptomatic carvenous sinus meningioma: a 15-years experience with fractionated stereotactic radiotherapy and radiosurgery. Radiat Oncol 9:27
Dieterich S, Cavedon C, Chuang CF, Cohen AB, Garrett JA, Lee CL, Lowenstein JR, d'Souza MF, Taylor DD Jr, Wu X, Yu C (2011) Report of AAPM TG 135: quality assurance for robotic radiosurgery. Med Phys 38:2914–2936
Dutta D, Balaji Subramanian S, Murli V, Sudahar H, Gopalakrishna Kurup PG, Potharaju M (2012) Dosimetric comparison of Linac-based (BrainLAB®) and robotic radiosurgery (CyberKnife ®) stereotactic system plans for acoustic schwannoma. J Neurooncol 106:637–642
Echner GG, Kilby W, Lee M, Earnst E, Sayeh S, Schlaefer A, Rhein B, Dooley JR, Lang C, Blanck O, Lessard E, Maurer CR Jr, Schlegel W (2009) The design, physical properties and clinical utility of an iris collimator for robotic radiosurgery. Phys Med Biol 54:5359–5380
El Majdoub F, Elawady M, Bührle C, El-Khatib M, Hoevels M, Treuer H, Müller RP, Sturm V, Maarouf M (2012) μMLC-LINAC radiosurgery for intracranial meningiomas of complex shape. Acta Neurochir (Wien) 154:599–604
Feuvret L, Noël G, Mazeron JJ, Bey P (2006) Conformity index: a review. Int J Radiat Oncol Biol Phys 64:333–342
Fu D, Kuduvalli G (2008) A fast, accurate, and automatic 2D-3D image registration for image-guided cranial radiosurgery. Med Phys 35:2180–2194
Fuetsch M, El Majdoub F, Hoevels M, Müller RP, Sturm V, Maarouf M (2012) Stereotactic LINAC radiosurgery for the treatment of brainstem cavernomas. Strahlenther Onkol 188:311–316
Gevaert T, Levivier M, Lacornerie T, Verellen D, Engels B, Reynaert N, Tournel K, Duchateau M, Reynders T, Depuydt T, Collen C, Lartigau E, De Ridder M (2013) Dosimetric comparison of different treatment modalities for stereotactic radiosurgery of arteriovenous malformations and acoustic neuromas. Radiother Oncol 106:192–197
Henzel M, Hamm K, Sitter H, Gross MW, Surber G, Kleinert G, Engenhart-Cabillic R (2009) Comparison of stereotactic radiosurgery and fractionated stereotactic radiotherapy of acoustic neurinomas according to 3-D tumor volume shrinkage and quality of life. Strahlenther Onkol 185:567–573
Kocher M, Wittig A, Piroth MD, Treuer H, Seegenschmiedt H, Ruge M, Grosu AL, Guckenberger M (2014) Stereotactic radiosurgery for treatment of brain metastases. A report of the DEGRO Working Group on Stereotactic Radiotherapy. Strahlenther Onkol 190:521–532
Kuo JS, Yu C, Petrovich Z, Apuzzo ML (2003) The CyberKnife stereotactic radiosurgery system: description, installation, and an initial evaluation of use and functionality. Neurosurgery 53:1235–1239
Lomax NJ, Scheib SG (2003) Quantifying the degree of conformity in radiosurgery treatment planning. Int J Radiat Oncol Biol Phys 55:1409–1419
Ma L, Petti P, Wang B, Descovich M, Chuang C, Barani IJ, Kunwar S, Shrieve DC, Sahgal A, Larson DA (2011) Apparatus dependence of normal brain tissue dose in stereotactic radiosurgery for multiple brain metastases. J Neurosurg 114:1580–1584
Paddick I (2000) A simple scoring ratio to index the conformity of radiosurgical treatment plans. Technical note. J Neurosurg 93(Suppl 3):219–222
Pastyr O, Hartmann GH, Schlegel W, Schabbert S, Treuer H, Lorenz WJ, Sturm V (1989) Stereotactically guided convergent beam irradiation with a linear accelerator: localization-technique. Acta Neurochir (Wien) 99:61–64
Phillips MH, Holdsworth C (2011) When is better best? A multiobjective perspective. Med Phys 38:1635–1640
Romanelli P, Schaal DW, Adler JR (2006) Image-guided radiosurgical ablation of intra- and extra-cranial lesions. Technol Cancer Res Treat 5:421–428
Ruge MI, Kocher M, Maarouf M, Hamisch C, Treuer H, Voges J, Sturm V (2011) Comparison of stereotactic brachytherapy (125 iodine seeds) with stereotactic radiosurgery (LINAC) for the treatment of singular cerebral metastases. Strahlenther Onkol 187:7–14
Runge MJ, Maarouf M, Hunsche S, Kocher M, Ruge MI, El Majdoub F, Treuer H, Mueller RP, Voges J, Sturm V (2012) LINAC-radiosurgery for nonsecreting pituitary adenomas. Long-term results. Strahlenther Onkol 188:319–325
Schlaefer A, Schweikard A (2008) Stepwise multi-criteria optimization for robotic radiosurgery. Med Phys 35:2094–2103
Schlegel W, Pastyr O, Kubesch R, Stein J, Diemer T, Höver KH, Rhein B (1997) A computer controlled micromultileaf-collimator for stereotactic conformal radiotherapy. In: Leavitt DD, Starkschall G (eds) Proceedings of the 12th International Conference on the use of computers in radiation therapy. Medical Physics Publishing, Madison, pp 79–82
Schoonbeek A, Monshouwer R, Hanssens P, Raaijmakers E, Nowak P, Marijnissen JP, Lagerwaard FJ, Cuijpers JP, Vonk EJ, van der Maazen RW (2010) Intracranial radiosurgery in the Netherlands. A planning comparison of available systems with regard to physical aspects and workload. Technol Cancer Res Treat 9:279–290
Seung SK, Larson DA, Galvin JM, Mehta MP, Potters L, Schultz CJ, Yajnik SV, Hartford AC, Rosenthal SA (2013) American College of Radiology (ACR) and American Society for Radiation Oncology (ASTRO) Practice Guideline for the Performance of Stereotactic Radiosurgery (SRS). Am J Clin Oncol 36:310–315
Sharma SC, Ott JT, Williams JB, Dickow D (2007) Commissioning and acceptance testing of a CyberKnife linear accelerator. J Appl Clin Med Phys 8:119–125
Shaw E, Kline R, Gillin M, Souhami L, Hirschfeld A, Dinapoli R, Martin L (1993) Radiation Therapy Oncology Group: radiosurgery quality assurance guidelines. Int J Radiat Oncol Biol Phys 27:1231–1239
Treuer H, Kocher M, Hoevels M, Hunsche S, Luyken K, Maarouf M, Voges J, Müller RP, Sturm V (2006) Impact of target point deviations on control and complication probabilities in stereotactic radiosurgery of AVMs and metastases. Radiother Oncol 81:25–32
van’t Riet A, Mak AC, Moerland MA, Elders LH, van der Zee W (1997) A conformation number to quantify the degree of conformality in brachytherapy and external beam irradiation: application to the prostate. Int J Radiat Oncol Biol Phys 37:731–736
Voges J, Treuer H, Sturm V, Büchner C, Lehrke R, Kocher M, Staar S, Kuchta J, Müller RP (1996) Risk analysis of linear accelerator radiosurgery. Int J Radiat Oncol Biol Phys 36:1055–1063
Yu C, Jozsef G, Apuzzo ML, Petrovich Z (2003) Dosimetric comparison of CyberKnife with other radiosurgical modalities for an ellipsoidal target. Neurosurgery 53:1155–62 (discussion 1162–1163)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
H. Treuer, M. Hoevels, K. Luyken, V. Visser-Vandewalle, J. Wirths, M. Kocher, and M. Ruge state that there are no conflicts of interest.
The accompanying manuscript does not include studies on humans or animals.
Rights and permissions
About this article
Cite this article
Treuer, H., Hoevels, M., Luyken, K. et al. Intracranial stereotactic radiosurgery with an adapted linear accelerator vs. robotic radiosurgery. Strahlenther Onkol 191, 470–476 (2015). https://doi.org/10.1007/s00066-014-0786-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-014-0786-y
Keywords
- Brain neoplasms
- Neoplasm metastasis
- Arteriovenous malformations
- Radiotherapy planning, computer-assisted
- Radiosurgery