Abstract
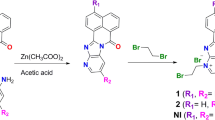
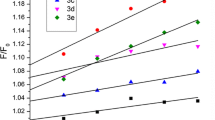
A series of novel water-soluble tri-substituted naphthalene diimide (NDI) derivatives bearing 2-oligoethoxy ethanamine side chain end-labeled with tertiary amino groups have been designed and synthesized. The cytotoxicity of the novel naphthalene diimides against cancer cell lines (K562, A549 and HeLa) showed that these compounds had excellent anti-cancer activity and the short length of the side chain (having one oxyethylene) was found to be beneficial to the observed anti-proliferative activity. Their DNA-binding properties were also studied using UV–Vis spectroscopy, fluorescence spectroscopy, and circular dichroism. The results showed that such tri-substituted NDI as DNA intercalators exhibited strong π–π stack binding capability and high binding affinities towards calf thymus (Ct) DNA and G-quadruplex (G4) DNA. The order of the binding constants is Ct-DNA > G4 DNA, which suggested that these compounds are more effective ligands to the duplex DNA.
Graphical Abstract
A series of novel water-soluble tri-substituted NDI derivatives bearing 2-oligoethoxy ethanamine side chains end-labeled with tertiary amino groups have been designed and synthesized. These compounds had excellent anti-cancer activity and the short length of the side chain (having one oxyethylene) would be beneficial to their anti-proliferative activity.









Similar content being viewed by others
References
Allenmark S (2003) Induced circular dichroism by chiral molecular interaction. Chirality 15:409–422
Berman HM, Young PR (1981) The interaction of intercalating drugs with nucleic acids. Annu Rev Biophys Bioeng 10:87–114
Campbell NH, Parkinson GN, Reszka AP, Neidle S (2008) Structural basis of DNA quadruplex recognition by an acridine drug. J Am Chem Soc 130:6722–6724
Collie GW, Promontorio R, Hampel SM, Micco M, Neidle S, Parkinson GN (2012) Structural basis for telomeric G-quadruplex targeting by naphthalene diimide ligands. J Am Chem Soc 134:2723–2731
Corpet DE, Parnaud G, Delverdier M, Peiffer G, Tache S (2000) Consistent and fast inhibition of colon carcinogenesis by polyethylene glycol in mice and rats given various carcinogens. Cancer Res 60:3160–3164
Cragg GM, Grothaus PG, Newman DJ (2009) Impact of natural products on developing new anti-cancer agents. Chem Rev 109:3012–3043
Cuenca F, Greciano O, Gunaratnam M, Haider S, Munnur D, Nanjunda R, Wilson WD, Neidle S (2008) Tri- and tetra-substituted naphthalene diimides as potent G-quadruplex ligands. Bioorg Med Chem Lett 18:1668–1673
Di Antonio M, Doria F, Richter SN, Bertipaglia C, Mella M, Sissi C, Palumbo M, Freccero M (2009) Quinone methides tethered to naphthalene diimides as selective G-quadruplex alkylating agents. J Am Chem Soc 131:13132–13141
Doria F, Nadai M, Sattin G, Pasotti L, Richter SN, Freccero M (2012) Water soluble extended naphthalene diimides as pH fluorescent sensors and G-quadruplex ligands. Org Biomol Chem 10:3830–3840
Faulhaber K, Granzhan A, Ihmels H, Otto D, Thomas L, Wells S (2011) Studies of the fluorescence light-up effect of amino-substituted benzo[b]quinolizinium derivatives in the presence of biomacromolecules. Photochem Photobiol Sci 10:1535–1545
Gunaratnam M, Swank S, Haider SM, Galesa K, Reszka AP, Beltran M, Cuenca F, Fletcher JA, Neidle S (2009) Targeting human gastrointestinal stromal tumor cells with a quadruplex-binding small molecule. J Med Chem 52:3774–3783
Gunaratnam M, de la Fuente M, Hampel SM, Todd AK, Reszka AP, Schatzlein A, Neidle S (2011) Targeting pancreatic cancer with a G-quadruplex ligand. Bioorg Med Chem 19:7151–7157
Hampel SM, Sidibe A, Gunaratnam M, Riou JF, Neidle S (2010) Tetrasubstituted naphthalene diimide ligands with selectivity for telomeric G-quadruplexes and cancer cells. Bioorg Med Chem Lett 20:6459–6463
Jain AK, Paul A, Maji B, Muniyappa K, Bhattacharya S (2012) Dimeric 1,3-phenylene-bis(piperazinyl benzimidazole)s: synthesis and structure-activity investigations on their binding with human telomeric G-quadruplex DNA and telomerase inhibition properties. J Med Chem 55:2981–2993
Janovec L, Kožurková M, Sabolová D, Ungvarský J, Paulíková H, Plšíková J, Vantová Z, Imrich J (2011) Cytotoxic 3,6-bis((imidazolidinone) imino) acridines: synthesis, DNA binding and molecular modeling. Bioorg Med Chem 19:1790–1801
Jiang X, Shang L, Wang ZX, Dong S (2005) Spectrometric and voltammetric investigation of interaction of neutral red with calf thymus DNA: pH effect. Biophys Chem 118:42–50
Long EC, Barton JK (1990) On demonstrating DNA intercalation. Acc Chem Res 23:271–273
Lv M, Xu H (2009) Overview of naphthalimide analogs as anticancer agents. Curr Med Chem 16:4797–4813
McKnight RE, Reisenauer E, Pintado MV, Polasani SR, Dixon DW (2011) Substituent effect on the preferred DNA binding mode and affinity of a homologous series of naphthalene diimides. Bioorg Med Chem Lett 21:4288–4291
Micco M, Collie GW, Dale AG, Ohnmacht SA, Pazitna I, Gunaratnam M, Reszka AP, Neidle S (2013) Structure-based design and evaluation of naphthalene diimide G-quadruplex ligands as telomere targeting agents in pancreatic cancer cells. J Med Chem 56:2959–2974
Mosmann TJ (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. Immunol Meth 65:55–63
Pandey S, Baker GA, Kane MA, Bonzagni NJ, Bright FV (2000) O2 quenching of ruthenium(II) tris(2,2′-bypyridyl)2+ within the water pool of perfluoropolyether-based reverse micelles formed in supercritical carbon dioxide. Langmuir 16:5593–5599
Peduto A, Pagano B, Petronzi C, Massa A, Esposito V, Virgilio A, Paduano F, Trapasso F, Fiorito F, Florio S, Giancola C, Galeone A, Filosa R (2011) Design, synthesis, biophysical and biological studies of trisubstituted naphthalimides as G-quadruplex ligands. Bioorg Med Chem 19:6419–6429
Purcell M, Neault JF, Tajmir-Riahi HA (2000) Interaction of taxol with human serum albumin. Biochem Biophys Acta 1478:61–68
Read MA, Neidle S (2000) Structural characterization of a guanine-quadruplex ligand complex. Biochemistry 39:13422–13432
Sahoo D, Bhattacharya P, Chakravorti S (2010) Reverse micelle induced flipping of binding site and efficiency of albumin protein with an ionic styryl dye. J Phys Chem B 114:10442–10450
Tumiatti V, Milelli A, Minarini A, Micco M, Campani AG, Roncuzzi L, Baiocchi D, Marinello J, Capranico G, Zini M, Stefanelli C, Melchiorre C (2009) Design, synthesis, and biological evaluation of substituted naphthalene imides and diimides as anticancer agent. J Med Chem 52:7873–7877
Uma Maheswari P, Palaniandavar M (2004) DNA binding and cleavage activity of (Ru(NH3)4(diimine))Cl2 complexes. Inorg Chim Acta 357:901–912
Uma V, Kanthimathi M, Weyhermuller T, Nair BU (2005) Oxidative DNA cleavage mediated by a new copper (II) terpyridine complex: crystal structure and DNA binding studies. J Inorg Biochem 99:2299–2307
Wang J, Wang D, Miller EK, Moses D, Bazan GC, Heeger AJ (2000a) Photoluminescence of water-soluble conjugated polymers: origin of enhanced quenching by charge transfer. Macromolecules 33:5153–5158
Wang Z, Liu D, Dong S (2000b) Voltammetric and spectroscopic studies on methyl green and cationic lipid bound to calf thymus DNA. Biophys Chem 87:179–184
Wang YX, Zhang XB, Zhao J, Xie SQ, Wang CJ (2012a) Nonhematotoxic naphthalene diimide modified by polyamine: synthesis and biological evaluation. J Med Chem 55:3502–3512
Wang SX, Li HD, Chen C, Zhang JC, Li SH, Qin XY, Li XL, Wang KR (2012b) Cytotoxicity and DNA binding property of phenanthrene imidazole with polyglycol side chains. Bioorg Med Chem Lett 22:6347–6351
Williamson JR (1993) Guanine quarters. Curr Opin Struct Biol 3:357–362
Xie LJ, Cui JN, Qian XH (2011) 5-Non-amino aromatic substituted naphthalimides as potential antitumor agents: synthesis via Suzuki reaction, antiproliferative activity, and DNA-binding behavior. Bioorg Med Chem 19:961–967
Zhang ZC, Yang YY, Zhang DN, Wang YY, Qian XH, Liu FY (2006) Acenaphtho[1,2-b]pyrrole derivatives as new family of intercalators: various DNA binding geometry and interesting antitumor capacity. Bioorg Med Chem 14:6962–6970
Zhang PZ, Wei C, Duan XX, Zhang XY, Wang KR, Chen H, Li XL (2012) Selective synthesis of bromo-substituted naphthalene dianhydride derivatives. Chem J Chin Univ 33:1466–1470
Acknowledgments
Financial supports from the 973 Program (2010CB534913) and the National Natural Science Foundation of China (NSFC) (20902016) are acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wei, H., Lv, M., Duan, X. et al. Cytotoxicity and DNA-binding property of water-soluble naphthalene diimide derivatives bearing 2-oligoethoxy ethanamine side chain end-labeled with tertiary amino groups. Med Chem Res 23, 2277–2286 (2014). https://doi.org/10.1007/s00044-013-0823-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-013-0823-x