Abstract
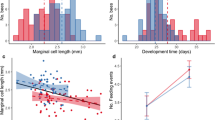
Eusocial insects, such as ants and honeybees, are characterized by the sophisticated division of labor among workers. In addition to age polyethism, the division of labor is maintained by task shifting of workers in fluctuating environments. While task experience is considered a key factor in realizing the task shifting, our previous study suggests that the experience promotes the task shifting of not nurses but foragers in a queenless ant, Diacamma cf. indicum from Japan. Therefore, we hypothesize that different mechanisms, e.g., reproductive potential, underlie task shifting between the nurse and the forager. In this study, we examined the relationship between the ovarian development of workers and their task shifting, using D. cf. indicum. We also examined the relationships between the expression levels of Vitellogenin (Vg), VgR, Vg-like A, Vg-like B, Vg-like C, and foraging gene and the task shifting of the nurses. Our results showed that more than half of the nurses maintained their reproductive potential—as evidenced by their well-developed ovaries—even when they had shifted to the foraging task. In contrast, the foragers—whether they had shifted to the nursing task or not—remained with regressed ovaries. Finally, we found that the nurses who shifted to the foraging task exhibited low expression levels of Vg, VgR, and Vg-like A. In conjunction with previous studies, our results suggest that this physiological contradiction may underlie the different mechanisms that lead to the task shifting between the nurse and the forager in this species.



Similar content being viewed by others
References
Amdam GV, Norberg K, Fondrk MK, Page RE (2004) Reproductive ground plan may mediate colony-level selection effects on individual foraging behavior in honey bees. Proc Natl Acad Sci 101:11350–11355. https://doi.org/10.1073/pnas.0403073101
Ament SA, Corona M, Pollock HS, Robinson GE (2008) Insulin signaling is involved in the regulation of worker division of labor in honey bee colonies. Proc Natl Acad Sci 105:4226–4231. https://doi.org/10.1073/pnas.0800630105
Bartoń K (2022) MuMIn: Multi-Model Inference. R package version 1.47.1
Beshers SN, Fewell JH (2001) Models of division of labor in social insects. Annu Rev Entomol 46:413–440. https://doi.org/10.1146/annurev.ento.46.1.413
Bonabeau E, Theraulaz G, Deneubourg JL (1996) Quantitative study of the fixed threshold model for the regulation of division of labour in insect societies. Proc R Soc B Biol Sci 263:1565–1569. https://doi.org/10.1098/rspb.1996.0229
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Mächler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400. https://doi.org/10.32614/rj-2017-066
Calabi P, Traniello JFA (1989) Behavioral flexibility in age castes of the ant Pheidole dentata. J Insect Behav 2:663–677. https://doi.org/10.1007/BF01065785
Camazine S, Deneubourg J-L, Franks NR, Sneyd J, Theraula G, Bonabeau E (2001) Self-Organization in Biological systems. Princeton University Press
Caminer MA, Libbrecht R, Majoe M, Ho DV, Baumann P, Foitzik S (2023) Task-specific odorant receptor expression in worker antennae indicates that sensory filters regulate division of labor in ants. Commun Biol 6:1–12. https://doi.org/10.1038/s42003-023-05273-4
Corona M, Libbrecht R, Wurm Y, Riba-Grognuz O, Studer RA, Keller L (2013) Vitellogenin Underwent Subfunctionalization to acquire caste and behavioral specific expression in the Harvester Ant Pogonomyrmex barbatus. PLoS Genet 9. https://doi.org/10.1371/journal.pgen.1003730
Crall JD, Gravish N, Mountcastle AM, Kocher SD, Oppenheimer RL, Pierce NE, Combes SA (2018) Spatial fidelity of workers predicts collective response to disturbance in a social insect. Nat Commun 9:1–13. https://doi.org/10.1038/s41467-018-03561-w
Dolezal AG, Brent CS, Hölldobler B, Amdam GV (2012) Worker division of labor and endocrine physiology are associated in the harvester ant, Pogonomyrmex californicus. J Exp Biol 215:454–460. https://doi.org/10.1242/jeb.060822
Duarte A, Weissing FJ, Pen I, Keller L (2011) An evolutionary perspective on Self-Organized Division of Labor in Social insects. Annu Rev Ecol Evol Syst 42:91–110. https://doi.org/10.1146/annurev-ecolsys0.1146/annurev-ecolsys-102710-145017-102710-145017
Dussutour A, Simpson SJ (2008) Description of a simple synthetic diet for studying nutritional responses in ants. Insectes Sociaux 55:329–333. https://doi.org/10.1007/s00040-008-1008-3
Enzmann BL, Nonacs P (2021) Age-related division of labor occurs in ants at the earliest stages of colony initiation. Behav Ecol Sociobiol 75. https://doi.org/10.1007/s00265-021-02974-w
Feldmeyer B, Elsner D, Foitzik S (2014) Gene expression patterns associated with caste and reproductive status in ants: worker-specific genes are more derived than queen-specific ones. Mol Ecol 23:151–161. https://doi.org/10.1111/mec.12490
Fox J, Weisberg S (2019) An R companion to Applied Regression. Sage, Thousand Oaks CA
Fujioka H, Okada Y, Abe MS (2021) Bipartite network analysis of ant-task associations reveals task groups and absence of colonial daily activity. R Soc Open Sci 8. https://doi.org/10.1098/rsos.201637
Fukumoto Y, Abe T, Taki A (1989) A novel form of colony organization in the ‘queenless’ ant Diacamma rugosum. Physiol Ecol Jpn 26:55–61
Guidugli KR, Nascimento AM, Amdam GV, Barchuk AR, Omholt S, Simões ZLP, Hartfelder K (2005) Vitellogenin regulates hormonal dynamics in the worker caste of a eusocial insect. FEBS Lett 579:4961–4965. https://doi.org/10.1016/j.febslet.2005.07.085
Hawkings C, Tamborindeguy C (2018) Expression analysis of vitellogenins in the workers of the red imported fire ant (Solenopsis invicta). PeerJ 2018. https://doi.org/10.7717/peerj.4875
Hölldobler B, Wilson EO (1990) The ants. Springer Berlin Heidelberg, Berlin, Heidelberg
Iwasa Y, Yamaguchi S (2020) Task allocation in a cooperative society: specialized castes or age-dependent switching among ant workers. Sci Rep 10:1–9. https://doi.org/10.1038/s41598-020-59920-5
Johnson BR (2002) Reallocation of labor in honeybee colonies during heat stress: the relative roles of task switching and the activation of reserve labor. Behav Ecol Sociobiol 51:188–196. https://doi.org/10.1007/s00265-001-0419-1
Kamhi JF, Traniello JFA (2013) Biogenic amines and Collective Organization in a superorganism: Neuromodulation of Social Behavior in ants. Brain Behav Evol 82:220–236. https://doi.org/10.1159/000356091
Kohlmeier P, Feldmeyer B, Foitzik S (2018) Vitellogenin-like A–associated shifts in social cue responsiveness regulate behavioral task specialization in an ant. PLoS Biol 16:e2005747. https://doi.org/10.1371/journal.pbio.2005747
Kohlmeier P, Alleman AR, Libbrecht R, Foitzik S, Feldmeyer B (2019) Gene expression is more strongly associated with behavioural specialization than with age or fertility in ant workers. Mol Ecol 28:658–670. https://doi.org/10.1111/mec.14971
Marco Antonio DS, Guidugli-Lazzarini KR, do Nascimento AM, Simões ZLP, Hartfelder K (2008) RNAi-mediated silencing of vitellogenin gene function turns honeybee (Apis mellifera) workers into extremely precocious foragers. Naturwissenschaften 95:953–961. https://doi.org/10.1007/s00114-008-0413-9
Matz MV (2020) MCMC.qpcr: Bayesian Analysis of qRT-PCR Data. R Package Version 124
Miyazaki S, Shimoji H, Suzuki R, Chinushi I, Takayanagi H, Yaguchi H, Miura T, Maekawa K (2021) Expressions of conventional vitellogenin and vitellogenin-like a in worker brains are associated with a nursing task in a ponerine ant. Insect Mol Biol 30:113–121. https://doi.org/10.1111/imb.12682
Morandin C, Hietala A, Helanterä H (2019) Vitellogenin and vitellogenin-like gene expression patterns in relation to caste and task in the ant Formica fusca. Insectes Sociaux 66:519–531. https://doi.org/10.1007/s00040-019-00725-9
Nakata K (1995) Age polyethism, idiosyncrasy and behavioural flexibility in the queenless ponerine ant, Diacamma Sp. J Ethol 13:113–123. https://doi.org/10.1007/BF02352570
Nakata K (1996) Does behavioral flexibility compensate or constrain colony productivity? Relationship among age structure, labor allocation, and production of workers in ant colonies. J Insect Behav 9:557–569. https://doi.org/10.1007/BF02213880
Nelson CM, Ihle KE, Fondrk MK, Page RE, Amdam GV (2007) The gene vitellogenin has multiple coordinating effects on social organization. PLoS Biol 5:0673–0677. https://doi.org/10.1371/journal.pbio.0050062
Okada Y, Miyazaki S, Miyakawa H, Ishikawa A, Tsuji K, Miura T (2010) Ovarian development and insulin-signaling pathways during reproductive differentiation in the queenless ponerine ant Diacamma Sp. J Insect Physiol 56:288–295. https://doi.org/10.1016/j.jinsphys.2009.10.013
Oster GF, Wilson EO (1978) Caste and ecology in the social insects. Princeton University Press, Princeton, NJ
Peeters C, Crewe R (1984) Insemination controls the reproductive division of labour in a ponerine ant. Naturwissenschaften 71:50–51. https://doi.org/10.1007/BF00365989
Peeters C, Tsuji K (1993) Reproductive conflict among ant workers in Diacamma sp. from Japan: dominance and oviposition in the absence of the gamergate. Insectes Sociaux 40:119–136. https://doi.org/10.1007/BF01240701
Powell S, Tschinkel WR (1999) Ritualized conflict in Odontomachus brunneus and the generation of interaction-based task allocation: a new organizational mechanism in ants. Anim Behav 58:965–972. https://doi.org/10.1006/anbe.1999.1238
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Ravary F, Lecoutey E, Kaminski G, Châline N, Jaisson P (2007) Individual experience alone can generate lasting Division of Labor in ants. Curr Biol 17:1308–1312. https://doi.org/10.1016/j.cub.2007.06.047
Richards MH (2019) Vitellogenin and vitellogenin-like genes: not just for egg production. Insectes Sociaux 66:505–506. https://doi.org/10.1007/s00040-019-00731-x
Robinson GE (1992) Regulation of division of labor in insect societies. Annu Rev Entomol 37:637–665. https://doi.org/10.1146/annurev.en.37.010192.003225
Robinson GE, Vargo EL (1997) Juvenile hormone in adult eusocial hymenoptera: Gonadotropin and behavioral pacemaker. Arch Insect Biochem Physiol 35:559–583. https://doi.org/10.1002/(SICI)1520-6327(1997)35:4<559::AID-ARCH13>3.0.CO;2-9
Robinson EJH, Feinerman O, Franks NR (2009) Flexible task allocation and the organization of work in ants. Proc R Soc B Biol Sci 276:4373–4380. https://doi.org/10.1098/rspb.2009.1244
Sasaki K, Okada Y, Shimoji H, Aonuma H, Miura T, Tsuji K (2021) Social Evolution with Decoupling of multiple roles of Biogenic Amines into different phenotypes in Hymenoptera. Front Ecol Evol 9:1–12. https://doi.org/10.3389/fevo.2021.659160
Shimoji H, Dobata S (2022) The build-up of dominance hierarchies in eusocial insects. Philos Trans R Soc Lond B Biol Sci 377:20200437. https://doi.org/10.1098/rstb.2020.0437
Shimoji H, Abe MS, Tsuji K, Masuda N (2014) Global network structure of dominance hierarchy of ant workers. J R Soc Interface 11. https://doi.org/10.1098/rsif.2014.0599
Shimoji H, Aonuma H, Miura T, Tsuji K, Sasaki K, Okada Y (2017) Queen contact and among-worker interactions dually suppress worker brain dopamine as a potential regulator of reproduction in an ant. Behav Ecol Sociobiol 71. https://doi.org/10.1007/s00265-016-2263-3
Shimoji H, Kikuchi T, Ohnishi H, Kikuta N, Tsuji K (2018) Social enforcement depending on the stage of colony growth in an ant. Proc R Soc B Biol Sci 285:4–9. https://doi.org/10.1098/rspb.2017.2548
Shimoji H, Kasutani N, Ogawa S, Hojo MK (2020) Worker propensity affects flexible task reversion in an ant. Behav Ecol Sociobiol 74. https://doi.org/10.1007/s00265-020-02876-3
Smith CR, Toth AL, Suarez AV, Robinson GE (2008) Genetic and genomic analyses of the division of labour in insect societies. Nat Rev Genet 9:735–748. https://doi.org/10.1038/nrg2429
Storey JD (2002) A direct approach to false discovery rates. J R Stat Soc Ser B Stat Methodol 64:479–498. https://doi.org/10.1111/1467-9868.00346
Storey JD, Taylor JE, Siegmund D (2004) Strong control, conservative point estimation and simultaneous conservative consistency of false discovery rates: a unified approach. J R Stat Soc Ser B Stat Methodol 66:187–205. https://doi.org/10.1111/j.1467-9868.2004.00439.x
Storey JD, Bass AJ, Alan Dabney A, Robinson D (2023) Qvalue: Q-value estimation for false discovery rate control. R Package Version 2.34.0
Tanaka Y, Hojo MK, Shimoji H (2022) Individual experience influences reconstruction of division of labour under colony disturbance in a queenless ant species. Front Zool 19. https://doi.org/10.1186/s12983-022-00466-9
Theraulaz G, Bonabeau E, Deneubourg JL (1998) Response threshold reinforcement and division of labour in insect societies. Proc R Soc B Biol Sci 265:327–332. https://doi.org/10.1098/rspb.1998.0299
Tripet F, Nonacs P (2004) Foraging for work and age-based polyethism: the roles of age and previous experience on task choice in ants. Ethology 110:863–877. https://doi.org/10.1111/j.1439-0310.2004.01023.x
Tsuji K, Egashira K, Hölldobler B (1999) Regulation of worker reproduction by direct physical contact in the ant Diacamma sp. from Japan. Anim Behav 58:337–343. https://doi.org/10.1006/anbe.1999.1161
Ulrich Y, Kawakatsu M, Tokita CK, Saragosti J, Chandra V, Tarnita CE, Kronauer DJC (2021) Response thresholds alone cannot explain empirical patterns of division of labor in social insects. PLoS Biol 19:e3001269. https://doi.org/10.1371/journal.pbio.3001269
Wakano JY, Nakata K, Yamamura N (1998) Dynamic model of optimal age polyethism in social insects under stable and fluctuating environments. J Theor Biol 193:153–165. https://doi.org/10.1006/jtbi.1998.0697
Wilson EO (1971) The insect societies. Belknap, Cambridge
Acknowledgements
We would like to thank Yusuke Ishizuka, Akari Nishina, Ayaka Tanooka, Rio Yamashita and Pan Zilin for collecting the ant colonies. We are also grateful to Prof. Toru Miura for supporting our histological observation. Finally, We would like to thank two anonymous reviewrs and Editor for helpfull commnets. This work was supported by Japan Society for the Promotion of Science KAKENHI grants (Nos. 22H02364 and 22H05068).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing or financial interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tanaka, Y., Oguchi, K., Miyazaki, S. et al. Reproductive potentials of task-shifting workers in a queenless ant. Insect. Soc. 71, 109–117 (2024). https://doi.org/10.1007/s00040-024-00949-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-024-00949-4