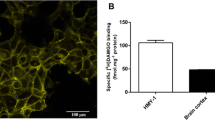
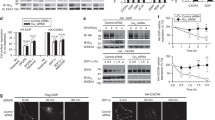
Abstract
Soon after internalization delta opioid receptors (DOPrs) are committed to the degradation path by G protein-coupled receptor (GPCR)-associated binding protein. Here we provide evidence that this classical post-endocytic itinerary may be rectified by downstream sorting decisions which allow DOPrs to regain to the membrane after having reached late endosomes (LE). The LE sorting mechanism involved ESCRT accessory protein Alix and the TIP47/Rab9 retrieval complex which supported translocation of the receptor to the TGN, from where it subsequently regained the cell membrane. Preventing DOPrs from completing this itinerary precipitated acute analgesic tolerance to the agonist DPDPE, supporting the relevance of this recycling path in maintaining the analgesic response by this receptor. Taken together, these findings reveal a post-endocytic itinerary where GPCRs that have been sorted for degradation can still recycle to the membrane.








Similar content being viewed by others
References
Gaveriaux-Ruff C, Kieffer BL (2011) Delta opioid receptor analgesia: recent contributions from pharmacology and molecular approaches. Behav Pharmacol 22(5–6):405–414. https://doi.org/10.1097/FBP.0b013e32834a1f2c
Gendron L, Cahill CM, von Zastrow M, Schiller PW, Pineyro G (2016) Molecular pharmacology of delta-opioid receptors. Pharmacol Rev 68(3):631–700. https://doi.org/10.1124/pr.114.008979
Audet N, Charfi I, Mnie-Filali O, Amraei M, Chabot-Dore AJ, Millecamps M, Stone LS, Pineyro G (2012) Differential association of receptor-Gbetagamma complexes with beta-arrestin2 determines recycling bias and potential for tolerance of delta opioid receptor agonists. J Neurosci 32(14):4827–4840. https://doi.org/10.1523/JNEUROSCI.3734-11.2012
Gupta A, Fujita W, Gomes I, Bobeck E, Devi LA (2015) Endothelin-converting enzyme 2 differentially regulates opioid receptor activity. Br J Pharmacol 172(2):704–719. https://doi.org/10.1111/bph.12833
Pradhan AA, Walwyn W, Nozaki C, Filliol D, Erbs E, Matifas A, Evans C, Kieffer BL (2010) Ligand-directed trafficking of the delta-opioid receptor in vivo: two paths toward analgesic tolerance. J Neurosci 30(49):16459–16468. https://doi.org/10.1523/JNEUROSCI.3748-10.2010
Henry AG, White IJ, Marsh M, von Zastrow M, Hislop JN (2011) The role of ubiquitination in lysosomal trafficking of delta-opioid receptors. Traffic 12(2):170–184. https://doi.org/10.1111/j.1600-0854.2010.01145.x
Whistler JL, Enquist J, Marley A, Fong J, Gladher F, Tsuruda P, Murray SR, Von Zastrow M (2002) Modulation of postendocytic sorting of G protein-coupled receptors. Science 297(5581):615–620. https://doi.org/10.1126/science.1073308
Pradhan AA, Becker JA, Scherrer G, Tryoen-Toth P, Filliol D, Matifas A, Massotte D, Gaveriaux-Ruff C, Kieffer BL (2009) In vivo delta opioid receptor internalization controls behavioral effects of agonists. PLoS One 4(5):e5425. https://doi.org/10.1371/journal.pone.0005425
Trapaidze N, Gomes I, Bansinath M, Devi LA (2000) Recycling and resensitization of delta opioid receptors. DNA Cell Biol 19(4):195–204. https://doi.org/10.1089/104454900314465
Archer-Lahlou E, Audet N, Amraei MG, Huard K, Paquin-Gobeil M, Pineyro G (2009) Src promotes delta opioid receptor (DOR) desensitization by interfering with receptor recycling. J Cell Mol Med 13(1):147–163. https://doi.org/10.1111/j.1582-4934.2008.00308.x
Henne WM, Buchkovich NJ, Emr SD (2011) The ESCRT pathway. Dev Cell 21(1):77–91. https://doi.org/10.1016/j.devcel.2011.05.015
Jacob C, Cottrell GS, Gehringer D, Schmidlin F, Grady EF, Bunnett NW (2005) c-Cbl mediates ubiquitination, degradation, and down-regulation of human protease-activated receptor 2. J Biol Chem 280(16):16076–16087. https://doi.org/10.1074/jbc.M500109200
Lauffer BE, Melero C, Temkin P, Lei C, Hong W, Kortemme T, von Zastrow M (2010) SNX27 mediates PDZ-directed sorting from endosomes to the plasma membrane. J Cell Biol 190(4):565–574. https://doi.org/10.1083/jcb.201004060
Hislop JN, Marley A, Von Zastrow M (2004) Role of mammalian vacuolar protein-sorting proteins in endocytic trafficking of a non-ubiquitinated G protein-coupled receptor to lysosomes. J Biol Chem 279(21):22522–22531. https://doi.org/10.1074/jbc.M311062200
Tanowitz M, Von Zastrow M (2002) Ubiquitination-independent trafficking of G protein-coupled receptors to lysosomes. J Biol Chem 277(52):50219–50222. https://doi.org/10.1074/jbc.C200536200
Cho DI, Zheng M, Min C, Kwon KJ, Shin CY, Choi HK, Kim KM (2013) ARF6 and GASP-1 are post-endocytic sorting proteins selectively involved in the intracellular trafficking of dopamine D(2) receptors mediated by GRK and PKC in transfected cells. Br J Pharmacol 168(6):1355–1374. https://doi.org/10.1111/bph.12025
Tappe-Theodor A, Agarwal N, Katona I, Rubino T, Martini L, Swiercz J, Mackie K, Monyer H, Parolaro D, Whistler J, Kuner T, Kuner R (2007) A molecular basis of analgesic tolerance to cannabinoids. J Neurosci 27(15):4165–4177. https://doi.org/10.1523/JNEUROSCI.5648-06.2007
Rosciglione S, Theriault C, Boily MO, Paquette M, Lavoie C (2014) Galphas regulates the post-endocytic sorting of G protein-coupled receptors. Nat Commun 5:4556. https://doi.org/10.1038/ncomms5556
Sonnichsen B, De Renzis S, Nielsen E, Rietdorf J, Zerial M (2000) Distinct membrane domains on endosomes in the recycling pathway visualized by multicolor imaging of Rab4, Rab5, and Rab11. J Cell Biol 149(4):901–914
van der Sluijs P, Hull M, Webster P, Male P, Goud B, Mellman I (1992) The small GTP-binding protein rab4 controls an early sorting event on the endocytic pathway. Cell 70(5):729–740
Raiborg C, Bache KG, Gillooly DJ, Madshus IH, Stang E, Stenmark H (2002) Hrs sorts ubiquitinated proteins into clathrin-coated microdomains of early endosomes. Nat Cell Biol 4(5):394–398. https://doi.org/10.1038/ncb791
Rink J, Ghigo E, Kalaidzidis Y, Zerial M (2005) Rab conversion as a mechanism of progression from early to late endosomes. Cell 122(5):735–749. https://doi.org/10.1016/j.cell.2005.06.043
Rojas R, Kametaka S, Haft CR, Bonifacino JS (2007) Interchangeable but essential functions of SNX1 and SNX2 in the association of retromer with endosomes and the trafficking of mannose 6-phosphate receptors. Mol Cell Biol 27(3):1112–1124. https://doi.org/10.1128/MCB.00156-06
Temkin P, Lauffer B, Jager S, Cimermancic P, Krogan NJ, von Zastrow M (2011) SNX27 mediates retromer tubule entry and endosome-to-plasma membrane trafficking of signalling receptors. Nat Cell Biol 13(6):715–721. https://doi.org/10.1038/ncb2252
Feng Y, Press B, Wandinger-Ness A (1995) Rab 7: an important regulator of late endocytic membrane traffic. J Cell Biol 131(6 Pt 1):1435–1452
Press B, Feng Y, Hoflack B, Wandinger-Ness A (1998) Mutant Rab7 causes the accumulation of cathepsin D and cation-independent mannose 6-phosphate receptor in an early endocytic compartment. J Cell Biol 140(5):1075–1089
Diaz E, Pfeffer SR (1998) TIP47: a cargo selection device for mannose 6-phosphate receptor trafficking. Cell 93(3):433–443
Lombardi D, Soldati T, Riederer MA, Goda Y, Zerial M, Pfeffer SR (1993) Rab9 functions in transport between late endosomes and the trans Golgi network. EMBO J 12(2):677–682
Bissig C, Gruenberg J (2014) ALIX and the multivesicular endosome: ALIX in Wonderland. Trends Cell Biol 24(1):19–25. https://doi.org/10.1016/j.tcb.2013.10.009
Matsuo H, Chevallier J, Mayran N, Le Blanc I, Ferguson C, Faure J, Blanc NS, Matile S, Dubochet J, Sadoul R, Parton RG, Vilbois F, Gruenberg J (2004) Role of LBPA and Alix in multivesicular liposome formation and endosome organization. Science 303(5657):531–534. https://doi.org/10.1126/science.1092425
De Matteis MA, Luini A (2008) Exiting the Golgi complex. Nat Rev Mol Cell Biol 9(4):273–284. https://doi.org/10.1038/nrm2378
Ladinsky MS, Wu CC, McIntosh S, McIntosh JR, Howell KE (2002) Structure of the Golgi and distribution of reporter molecules at 20 degrees C reveals the complexity of the exit compartments. Mol Biol Cell 13(8):2810–2825. https://doi.org/10.1091/mbc.01-12-0593
Tanowitz M, von Zastrow M (2003) A novel endocytic recycling signal that distinguishes the membrane trafficking of naturally occurring opioid receptors. J Biol Chem 278(46):45978–45986. https://doi.org/10.1074/jbc.M304504200
Cao H, Weller S, Orth JD, Chen J, Huang B, Chen JL, Stamnes M, McNiven MA (2005) Actin and Arf1-dependent recruitment of a cortactin–dynamin complex to the Golgi regulates post-Golgi transport. Nat Cell Biol 7(5):483–492. https://doi.org/10.1038/ncb1246
Wakana Y, van Galen J, Meissner F, Scarpa M, Polishchuk RS, Mann M, Malhotra V (2012) A new class of carriers that transport selective cargo from the trans Golgi network to the cell surface. EMBO J 31(20):3976–3990. https://doi.org/10.1038/emboj.2012.235
Salvarezza SB, Deborde S, Schreiner R, Campagne F, Kessels MM, Qualmann B, Caceres A, Kreitzer G, Rodriguez-Boulan E (2009) LIM kinase 1 and cofilin regulate actin filament population required for dynamin-dependent apical carrier fission from the trans-Golgi network. Mol Biol Cell 20(1):438–451. https://doi.org/10.1091/mbc.E08-08-0891
Camera P, da Silva JS, Griffiths G, Giuffrida MG, Ferrara L, Schubert V, Imarisio S, Silengo L, Dotti CG, Di Cunto F (2003) Citron-N is a neuronal Rho-associated protein involved in Golgi organization through actin cytoskeleton regulation. Nat Cell Biol 5(12):1071–1078. https://doi.org/10.1038/ncb1064
Itoh K, Yoshioka K, Akedo H, Uehata M, Ishizaki T, Narumiya S (1999) An essential part for Rho-associated kinase in the transcellular invasion of tumor cells. Nat Med 5(2):221–225. https://doi.org/10.1038/5587
Poteryaev D, Datta S, Ackema K, Zerial M, Spang A (2010) Identification of the switch in early-to-late endosome transition. Cell 141(3):497–508. https://doi.org/10.1016/j.cell.2010.03.011
Vitelli R, Santillo M, Lattero D, Chiariello M, Bifulco M, Bruni CB, Bucci C (1997) Role of the small GTPase Rab7 in the late endocytic pathway. J Biol Chem 272(7):4391–4397
Girard E, Chmiest D, Fournier N, Johannes L, Paul JL, Vedie B, Lamaze C (2014) Rab7 is functionally required for selective cargo sorting at the early endosome. Traffic 15(3):309–326. https://doi.org/10.1111/tra.12143
Kucera A, Bakke O, Progida C (2016) The multiple roles of Rab9 in the endolysosomal system. Commun Integr Biol 9(4):e1204498. https://doi.org/10.1080/19420889.2016.1204498
Ganley IG, Carroll K, Bittova L, Pfeffer S (2004) Rab9 GTPase regulates late endosome size and requires effector interaction for its stability. Mol Biol Cell 15(12):5420–5430. https://doi.org/10.1091/mbc.E04-08-0747
Aivazian D, Serrano RL, Pfeffer S (2006) TIP47 is a key effector for Rab9 localization. J Cell Biol 173(6):917–926. https://doi.org/10.1083/jcb.200510010
Dores MR, Chen B, Lin H, Soh UJ, Paing MM, Montagne WA, Meerloo T, Trejo J (2012) ALIX binds a YPX(3)L motif of the GPCR PAR1 and mediates ubiquitin-independent ESCRT-III/MVB sorting. J Cell Biol 197(3):407–419. https://doi.org/10.1083/jcb.201110031
Tomas A, Vaughan SO, Burgoyne T, Sorkin A, Hartley JA, Hochhauser D, Futter CE (2015) WASH and Tsg101/ALIX-dependent diversion of stress-internalized EGFR from the canonical endocytic pathway. Nat Commun 6:7324. https://doi.org/10.1038/ncomms8324
Le Blanc I, Luyet PP, Pons V, Ferguson C, Emans N, Petiot A, Mayran N, Demaurex N, Faure J, Sadoul R, Parton RG, Gruenberg J (2005) Endosome-to-cytosol transport of viral nucleocapsids. Nat Cell Biol 7(7):653–664. https://doi.org/10.1038/ncb1269
Pfeffer SR (2009) Multiple routes of protein transport from endosomes to the trans Golgi network. FEBS Lett 583(23):3811–3816. https://doi.org/10.1016/j.febslet.2009.10.075
Chia PZ, Gasnereau I, Lieu ZZ, Gleeson PA (2011) Rab9-dependent retrograde transport and endosomal sorting of the endopeptidase furin. J Cell Sci 124(Pt 14):2401–2413. https://doi.org/10.1242/jcs.083782
Mittal N, Roberts K, Pal K, Bentolila LA, Fultz E, Minasyan A, Cahill C, Pradhan A, Conner D, DeFea K, Evans C, Walwyn W (2013) Select G-protein-coupled receptors modulate agonist-induced signaling via a ROCK, LIMK, and beta-arrestin 1 pathway. Cell Rep 5(4):1010–1021. https://doi.org/10.1016/j.celrep.2013.10.015
Cao TT, Mays RW, von Zastrow M (1998) Regulated endocytosis of G-protein-coupled receptors by a biochemically and functionally distinct subpopulation of clathrin-coated pits. J Biol Chem 273(38):24592–24602
Boulay PL, Schlienger S, Lewis-Saravalli S, Vitale N, Ferbeyre G, Claing A (2011) ARF1 controls proliferation of breast cancer cells by regulating the retinoblastoma protein. Oncogene 30(36):3846–3861. https://doi.org/10.1038/onc.2011.100
Nagi K, Charfi I, Pineyro G (2015) Kir3 channels undergo arrestin-dependant internalization following delta opioid receptor activation. Cell Mol Life Sci 72(18):3543–3557. https://doi.org/10.1007/s00018-015-1899-x
Audet N, Paquin-Gobeil M, Landry-Paquet O, Schiller PW, Pineyro G (2005) Internalization and Src activity regulate the time course of ERK activation by delta opioid receptor ligands. J Biol Chem 280(9):7808–7816. https://doi.org/10.1074/jbc.M411695200
Boussif O, Lezoualc’h F, Zanta MA, Mergny MD, Scherman D, Demeneix B, Behr JP (1995) A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: polyethylenimine. Proc Natl Acad Sci USA 92(16):7297–7301
Vargas GA, Von Zastrow M (2004) Identification of a novel endocytic recycling signal in the D1 dopamine receptor. J Biol Chem 279(36):37461–37469. https://doi.org/10.1074/jbc.M401034200
Scherrer G, Tryoen-Toth P, Filliol D, Matifas A, Laustriat D, Cao YQ, Basbaum AI, Dierich A, Vonesh JL, Gaveriaux-Ruff C, Kieffer BL (2006) Knockin mice expressing fluorescent delta-opioid receptors uncover G protein-coupled receptor dynamics in vivo. Proc Natl Acad Sci USA 103(25):9691–9696. https://doi.org/10.1073/pnas.0603359103
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL (1994) Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 53(1):55–63
Team RC (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org/
Acknowledgements
This research was supported by the Natural Sciences and Engineering Research Council of Canada [Grants RGPIN-2015-05213 (to L.G.) and 311997 (to G.P.)] and the Canadian Institutes of Health Research [Grants MOP 123399 and MOP 136871 (to L.G.); MOP 79432 and MOP 324876 (to G.P.)]. L.G. is the recipient of a Chercheur-boursier Senior and I. C. of a doctoral award from the Fonds de la Recherche du Québec-Santé. The authors thank L. Posa for technical assistance in production of supplementary figure 5c and Dr. P. Dallaire for expert advice on statistical analyses.
Author information
Authors and Affiliations
Contributions
IC and GP conceived the project and wrote the manuscript. IC performed all of the experiments except those of Fig. 6 (done by KA). LG and KA conceived and analyzed animal studies.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial and no financial interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
18_2017_2732_MOESM1_ESM.tif
Supplementary Fig. 1. DOPrs do not rely upon Rab4-dependent transport to regain the membrane. Cortical neurons were transfected with Flag-DOPrs (red) and pcDNA3 or Rab4N121I (green) as indicated, treated with vehicle (0.05% DMSO) or DPDPE (10 µM, 60 min) and processed as in Fig. 1. Upper panels show intracellular labeling immediately after end of indicated treatment, and lower panels show images for intracellular and surface labeling in DPDPE-treated cells that were allowed to recover (60 min) in the absence of agonist. Scatter plots and histograms correspond to intracellular labeling density (mean ± SEM; n = 18) in neurons labeled immediately after DPDPE treatment and neurons that were allowed to recover from DPDPE exposure. Statistical comparisons by two-way ANOVA followed by Tukey’s posthoc test revealed reduction in intracellular labeling following recovery both in cells transfected with pcDNA3 and Rab4N121I (***p˂0.0001). Scale bar, 50 µm (a). HEK293 cells were co-transfected with Flag-DOPrs and pcDNA3 or Rab4N121I, exposed to DPDPE (1 µM, 30 min) and recycling assessed upon removal of the agonist. Results were expressed as % of maximal recycling observed in vector-transfected controls and correspond to mean ± SEM (n = 4). Curves were fit to one phase exponential kinetics and plateaux for pcDNA3 and Rab4N121I were compared using ‘extra sum of squares F test’ which revealed no significant difference among conditions: plateau pcDNA3 = 92.09 ± 14.91, Plateau Rab4N121I = 94.29 ± 10.62, p = 0.8994. (b). (TIFF 2149 kb)
18_2017_2732_MOESM2_ESM.tif
Supplementary Fig. 2. DOPr recycling in HEK cells is blocked by inactive Rab7 but not VPS35, which nonetheless blocks β2ADR receptor recycling in neurons and HEK cells. Cortical neuron cultures were transfected with Flag-β2ARs and either scrambled (Scr-) or VPS35 siRNA. On the day of the experiment they were labeled, treated with isoproterenol (1 µM; 60 min) or vehicle (0.05% DMSO) and processed as in previous figures. Upper panels show intracellular labeling immediately after end of indicated treatment, and lower panels show images for intracellular and surface labeling in isoproterenol-treated cells that were allowed to recover (60 min) in the absence of agonist. Scatter plots and histograms (mean ± SEM; n = 18) represent intracellular labeling density in neurons processed immediately after treatment, or neurons that were allowed to recover. Statistical comparisons by two-way ANOVA followed by Tukey’s posthoc test revealed significant reduction of intracellular labeling following recovery in neurons transfected with scrambled (***p˂0.0001), but not VPS35 siRNA (a). HEK293 cells stably expressing Flag-β2ARs were transfected with either scrambled or VPS35 siRNA, were treated with isoproterenol (1 µM; 30 min) and recycling assessed upon removal of the agonist. Results were expressed as % of maximal recycling observed in scrambled-transfected controls after 60 min recovery from agonist exposure (mean ± SEM; n = 6). Statistical comparison using two-tailed Student’s t test; p = 0.0039 (b). HEK293 cells were co-transfected with Flag-DOPrs and pcDNA3 or Rab7N125I, exposed to DPDPE (1 µM, 30 min) and recycling assessed upon removal of the agonist. Results were expressed as % of maximal recycling observed in vector-transfected controls (mean ± SEM; n = 5). Curves were fit to one phase exponential kinetics and plateaux for pcDNA3 and Rab7N125I were compared using ‘extra sum of squares F test’ to reveal an effect of inactive Rab7: plateau pcDNA3 = 99.8 ± 22.22, plateau Rab7N125I = 49.7 ± 17.97, p < 0.0001. (c). Recycling was assessed in HEK293 cells expressing Flag-DOPr and transfected with scrambled or VPS35 siRNA and DPDPE (1 µM, 30 min). Results correspond to mean ± SEM (n = 6). Curves were fit to one phase exponential kinetics and plateaux for scrambled or VPS35 siRNA compared using ‘extra sum of squares F test’ to reveal no effect of VPS35 silencing: plateau scrambled = 101.5 ± 19.2, plateau VPS35 siRNA = 73.72 ± 9.2, p = 0.1705 (d). (TIFF 2820 kb)
18_2017_2732_MOESM3_ESM.tif
Supplementary Fig. 3. DOPrs colocalize with TGN46 in neurons. Neurons transfected with Flag-DOPrs were treated with DPDPE (10 μM; 60 min) or vehicle (0.05% DMSO) and either immediately processed (a) or allowed to recover in presence or absence of ROCK inhibitor Y27632 (10 µM) (b) before revealing DOPr (red) and endogenous TGN46 (green). Insets: show DOPr colocalization with TGN46 for corresponding conditions. (TIFF 2200 kb)
18_2017_2732_MOESM4_ESM.tif
Supplementary Fig. 4. MOPr recycling in neurons is not affected by temperature and DOPr recycling in neurons is insensitive to PKD inhibitor CID755673. Cortical neuron cultures transfected with Flag-MOPrs were then labeled at the membrane with first antibody and exposed to DAMGO (10 µM; 60 min). At the end of treatment cells were immediately processed for visualization of intracellular labeling or allowed to recover from treatment (60 min) at 37oC or 20oC before revealing intracellular or surface labeling. Scatter plots and histograms below correspond to intracellular labeling (mean ± SEM; n = 18) as indicated. One-way ANOVA, followed by Tukey multiple comparisons, was used for statistical analysis and results are shown in the figure (a). Cortical neurons transfected with Flag-DOPrs were pretreated or not with CID755673 (20 µM) for 24hs. On the day of the experiment the medium was changed for one with freshly prepared blocker and neurons were exposed to DPDPE (10 µM; 60 min) or vehicle (0.05% DMSO). At the end of treatment, they were immediately processed or allowed to recover from treatment (60 min) before revealing intracellular or surface labeling. Scatter plots and histograms correspond to intracellular labeling density (mean ± SEM; n = 18) as indicated. Statistical comparisons by two-way ANOVA followed by Tukey multiple comparisons revealed significant reduction of intracellular labeling following recovery in cells exposed or not to CID755673, ** p < 0.01, *** p < 0.0001. Scale bar, 50 µm. (TIFF 1927 kb)
18_2017_2732_MOESM5_ESM.tif
Supplementary Fig. 5. DOPr recycling in HEK cells is sensitive to temperature block and to PKD inhibitor CID755673. HEK293 cells expressing Flag-DOPr (a) or Flag-MOPr (b) were treated with corresponding agonist as indicated (1 µM; 30 min), and recycling evaluated at 37oC or 20oC. Curves were fit to one phase exponential kinetics and time constant obtained at different temperatures compared using ‘extra sum of squares F test’. Comparisons revealed an effect of temperature for Flag-DOPrs (time constant 37oC = 0.047 ± 0.01, time constant 20oC = 0.001 ± 0.01, p < 0.0001; n = 5) but not Flag-MOPr (time constant 37oC = 0.038 ± 0.02, time constant 20oC = 0.028 ± 0.01, p = 0.6680; n = 3). HEK293 cells expressing Flag-DOPr were treated with DPDPE (1 µM; 30 min) and recycling was evaluated in control cells and cells exposed to Y-27632 (10 µM) (c) or Cytochalasin D (10 µM) (d) during recovery. Another set of cells was pretreated or not with CID755673 (20 µM) for 24 hs before recycling experiment was carried out (e). Curves were fit to one phase exponential kinetics and corresponding plateaux were compared using ‘extra sum of squares F test’. There was no effect of Y-27632 (plateau CTL = 91.5 ± 6.5, plateau Y-27632 = 86.9 ± 8.4, p = 0.7188; n = 6) or Cytochalasin D (plateau CTL = 107.1 ± 8.6, plateau Cytochalasin D = 102.4 ± 12.9, p = 0.0920; n = 6), but the analysis revealed an effect of PKD inhibitor for CID755673 (plateau CTL = 100 ± 5.5, plateau CID755673 = 69.8 ± 5.6, p = 0.0104; n = 3). (TIFF 1659 kb)
18_2017_2732_MOESM6_ESM.tif
Supplementary Fig. 6. Definition of region of interest for quantifiucation of intracellular and surface labeling. Total cytoplasmic labeling density (CLD) was obtained by measuring fluorescence intensity within the region confined between the external (white) and nuclear perimeters (yellow) and dividing this value by the corresponding area (a). Total surface labeling density (SLD) was defined by measuring fluorescence within internal (yellow) and external (white) perimeters of surface-labeled neurons and dividing this value by the corresponding area (b). Nuclear labeling density (fluorescence within nuclear perimeter/nuclear area) was considered background. (TIFF 1252 kb)
Rights and permissions
About this article
Cite this article
Charfi, I., Abdallah, K., Gendron, L. et al. Delta opioid receptors recycle to the membrane after sorting to the degradation path. Cell. Mol. Life Sci. 75, 2257–2271 (2018). https://doi.org/10.1007/s00018-017-2732-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-017-2732-5