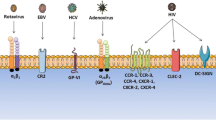
Abstract
Thrombocytopenia is a frequent complication of viral infections providing evidence that interaction of platelets with viruses is an important pathophysiological phenomenon. Multiple mechanisms are involved depending on the nature of the viruses involved. These include immunological platelet destruction, inappropriate platelet activation and consumption, and impaired megakaryopoiesis. Viruses bind platelets through specific receptors and identified ligands, which lead to mutual alterations of both the platelet host and the viral aggressor. We have shown that HIV-1 viruses are internalized specifically in platelets and megakaryocytes, where they can be either sheltered, unaltered (with potential transfer of the viruses into target organs), or come in contact with platelet secretory products leading to virus destruction and facilitated platelet clearance. In this issue, we have reviewed the various pathways that platelets use in order to interact with viruses, HIV and others. This review also shows that more work is still needed to precisely identify platelet roles in viral infections, and to answer the challenge of viral safety in platelet transfusion.








Similar content being viewed by others
References
Danon D, Jerushalmy Z, De Vries A (1959) Incorporation of influenza virus in human blood platelets in vitro electron microscopical observation. Virology 9:719–722
Jerushalmy Z, Kohn A, De Vries A (1961) Interaction of myxoviruses with human blood platelets in vitro. Proc Soc Exp Biol Med 106:462–466
White JG, Clawson CC (1982) Effects of small latex particle uptake on the surface connected canalicular system of blood platelets: a freeze-fracture and cytochemical study. Diagn Histopathol 5:3–10
Youssefian T, Drouin A, Masse JM, Guichard J, Cramer EM (2002) Host defense role of platelets: engulfment of HIV and Staphylococcus aureus occurs in a specific subcellular compartment and is enhanced by platelet activation. Blood 99:4021–4029
Pelletier JP, Transue S, Snyder EL (2006) Pathogen inactivation techniques. Best Pract Res Clin Haematol 19:205–242
Lin L, Hanson CV, Alter HJ, Jauvin V, Bernard KA, Murthy KK, Metzel P, Corash L (2005) Inactivation of viruses in platelet concentrates by photochemical treatment with amotosalen and long-wavelength ultraviolet light. Transfusion 45:580–590
Zucker-Franklin D, Seremetis S, Zheng ZY (1990) Internalization of human immunodeficiency virus type I and other retroviruses by megakaryocytes and platelets. Blood 75:1920–1923
Boukour S, Masse JM, Benit L, Dubart-Kupperschmitt A, Cramer EM (2006) Lentivirus degradation and DC-SIGN expression by human platelets and megakaryocytes. J Thromb Haemost 4:426–435
Kung SK, An DS, Chen IS (2000) A murine leukemia virus (MuLV) long terminal repeat derived from rhesus macaques in the context of a lentivirus vector and MuLV gag sequence results in high-level gene expression in human T lymphocytes. J Virol 74:3668–3681
Coulson BS, Londrigan SL, Lee DJ (1997) Rotavirus contains integrin ligand sequences and a disintegrin-like domain that are implicated in virus entry into cells. Proc Natl Acad Sci USA 94:5389–5394
Zhang Y, Bergelson JM (2005) Adenovirus receptors. J Virol 79:12125–12131
Eggerman TL, Mondoro TH, Lozier JN, Vostal JG (2002) Adenoviral vectors do not induce, inhibit, or potentiate human platelet aggregation. Hum Gene Ther 13:125–128
Stone D, Liu Y, Shayakhmetov D, Li ZY, Ni S, Lieber A (2007) Adenovirus-platelet interaction in blood causes virus sequestration to the reticuloendothelial system of the liver. J Virol 81:4866–4871
Mackow ER, Gavrilovskaya IN (2001) Cellular receptors and hantavirus pathogenesis. Curr Top Microbiol Immunol 256:91–115
Maginnis MS, Forrest JC, Kopecky-Bromberg SA, Dickeson SK, Santoro SA, Zutter MM, Nemerow GR, Bergelson JM, Dermody TS (2006) Beta1 integrin mediates internalization of mammalian reovirus. J Virol 80:2760–2770
Nunez D, Charriaut-Marlangue C, Barel M, Benveniste J, Frade R (1987) Activation of human platelets through gp140, the C3d/EBV receptor (CR2). Eur J Immunol 17:515–520
Ahmad A, Menezes J (1997) Binding of the Epstein-Barr virus to human platelets causes the release of transforming growth factor-beta. J Immunol 159:3984–3988
Zahn A, Jennings N, Ouwehand WH, Allain JP (2006) Hepatitis C virus interacts with human platelet glycoprotein VI. J Gen Virol 87:2243–2251
Othman M, Labelle A, Mazzetti I, Elbatarny HS, Lillicrap D (2007) Adenovirus-induced thrombocytopenia: the role of von Willebrand factor and P-selectin in mediating accelerated platelet clearance. Blood 109:2832–2839
Chaipan C, Soilleux EJ, Simpson P, Hofmann H, Gramberg T, Marzi A, Geier M, Stewart EA, Eisemann J, Steinkasserer A et al (2006) DC-SIGN and CLEC-2 mediate human immunodeficiency virus type 1 capture by platelets. J Virol 80:8951–8960
Basch RS, Kouri YH, Karpatkin S (1990) Expression of CD4 by human megakaryocytes. Proc Natl Acad Sci USA 87:8085–8089
Riviere C, Subra F, Cohen-Solal K, Cordette-Lagarde V, Letestu R, Auclair C, Vainchenker W, Louache F (1999) Phenotypic and functional evidence for the expression of CXCR4 receptor during megakaryocytopoiesis. Blood 93:1511–1523
Feinberg H, Mitchell DA, Drickamer K, Weis WI (2001) Structural basis for selective recognition of oligosaccharides by DC-SIGN and DC-SIGNR. Science 294:2163–2166
Geijtenbeek TB, Kwon DS, Torensma R, van Vliet SJ, van Duijnhoven GC, Middel J, Cornelissen IL, Nottet HS, KewalRamani VN, Littman DR et al (2000) DC-SIGN, a dendritic cell-specific HIV-1-binding protein that enhances trans-infection of T cells. Cell 100:587–597
Moris A, Nobile C, Buseyne F, Porrot F, Abastado JP, Schwartz O (2004) DC-SIGN promotes exogenous MHC-I-restricted HIV-1 antigen presentation. Blood 103:2648–2654
Geijtenbeek TB, Torensma R, van Vliet SJ, van Duijnhoven GC, Adema GJ, van Kooyk Y, Figdor CG (2000) Identification of DC-SIGN, a novel dendritic cell-specific ICAM-3 receptor that supports primary immune responses. Cell 100:575–585
Cramer EM, Vainchenker W, Vinci G, Guichard J, Breton-Gorius J (1985) Gray platelet syndrome: immunoelectron microscopic localization of fibrinogen and von Willebrand factor in platelets and megakaryocytes. Blood 66:1309–1316
Jerushalmy Z, Kaminski E, Kohn A, Devries A (1963) Interaction of Newcastle disease virus with megakaryocytes in cell cultures of Guinea pig bone marrow. Proc Soc Exp Biol Med 114:687–690
Crapnell K, Zanjani ED, Chaudhuri A, Ascensao JL, St Jeor S, Maciejewski JP (2000) In vitro infection of megakaryocytes and their precursors by human cytomegalovirus. Blood 95:487–493
Isomura H, Yoshida M, Namba H, Fujiwara N, Ohuchi R, Uno F, Oda M, Seino Y, Yamada M (2000) Suppressive effects of human herpesvirus-6 on thrombopoietin-inducible megakaryocytic colony formation in vitro. J Gen Virol 81:663–673
Gonelli A, Mirandola P, Grill V, Secchiero P, Zauli G (2002) Human herpesvirus 7 infection impairs the survival/differentiation of megakaryocytic cells. Haematologica 87:1223–1225
Kouri YH, Borkowsky W, Nardi M, Karpatkin S, Basch RS (1993) Human megakaryocytes have a CD4 molecule capable of binding human immunodeficiency virus-1. Blood 81:2664–2670
Chelucci C, Federico M, Guerriero R, Mattia G, Casella I, Pelosi E, Testa U, Mariani G, Hassan HJ, Peschle C (1998) Productive human immunodeficiency virus-1 infection of purified megakaryocytic progenitors/precursors and maturing megakaryocytes. Blood 91:1225–1234
Li X, Jeffers LJ, Garon C, Fischer ER, Scheffel J, Moore B, Reddy KR, Demedina M, Schiff ER (1999) Persistence of hepatitis C virus in a human megakaryoblastic leukaemia cell line. J Viral Hepat 6:107–114
Louache F, Bettaieb A, Henri A, Oksenhendler E, Farcet JP, Bierling P, Seligmann M, Vainchenker W (1991) Infection of megakaryocytes by human immunodeficiency virus in seropositive patients with immune thrombocytopenic purpura. Blood 78:1697–1705
La Russa VF, Innis BL (1995) Mechanisms of dengue virus-induced bone marrow suppression. Baillieres Clin Haematol 8:249–270
Ljungman P, Wang FZ, Clark DA, Emery VC, Remberger M, Ringden O, Linde A (2000) High levels of human herpesvirus 6 DNA in peripheral blood leucocytes are correlated to platelet engraftment and disease in allogeneic stem cell transplant patients. Br J Haematol 111:774–781
Weksler BB (2007) Review article: the pathophysiology of thrombocytopenia in hepatitis C virus infection and chronic liver disease. Aliment Pharmacol Ther 26(Suppl 1):13–19
McHutchison JG, Dusheiko G, Shiffman ML, Rodriguez-Torres M, Sigal S, Bourliere M, Berg T, Gordon SC, Campbell FM, Theodore D et al (2007) Eltrombopag for thrombocytopenia in patients with cirrhosis associated with hepatitis C. N Engl J Med 357:2227–2236
Krishnamurti C, Peat RA, Cutting MA, Rothwell SW (2002) Platelet adhesion to dengue-2 virus-infected endothelial cells. Am J Trop Med Hyg 66:435–441
Kalvegren H, Majeed M, Bengtsson T (2003) Chlamydia pneumoniae binds to platelets and triggers P-selectin expression and aggregation: a causal role in cardiovascular disease? Arterioscler Thromb Vasc Biol 23:1677–1683
Rahbar A, Soderberg-Naucler C (2005) Human cytomegalovirus infection of endothelial cells triggers platelet adhesion and aggregation. J Virol 79:2211–2220
Iannacone M, Sitia G, Isogawa M, Marchese P, Castro MG, Lowenstein PR, Chisari FV, Ruggeri ZM, Guidotti LG (2005) Platelets mediate cytotoxic T lymphocyte-induced liver damage. Nat Med 11:1167–1169
Tang YQ, Yeaman MR, Selsted ME (2002) Antimicrobial peptides from human platelets. Infect Immun 70:6524–6533
Maurice A, Marchand-Arvier M, Edert D, Le Faou A, Gondrexon G, Vigneron C (2002) The virucidal effect of platelet concentrates: preliminary study and first conclusions. Platelets 13:219–222
Kameyoshi Y, Dorschner A, Mallet AI, Christophers E, Schroder JM (1992) Cytokine RANTES released by thrombin-stimulated platelets is a potent attractant for human eosinophils. J Exp Med 176:587–592
Klinger MH, Wilhelm D, Bubel S, Sticherling M, Schroder JM, Kuhnel W (1995) Immunocytochemical localization of the chemokines RANTES and MIP-1 alpha within human platelets and their release during storage. Int Arch Allergy Immunol 107:541–546
Cocchi F, De Vico AL, Garzino-Demo A, Arya SK, Gallo RC, Lusso P (1995) Identification of RANTES, MIP-1 alpha, and MIP-1 beta as the major HIV-suppressive factors produced by CD8+ T cells. Science 270:1811–1815
Iannacone M, Sitia G, Isogawa M, Whitmire JK, Marchese P, Chisari FV, Ruggeri ZM, Guidotti LG (2008) Platelets prevent IFN-alpha/beta-induced lethal hemorrhage promoting CTL-dependent clearance of lymphocytic choriomeningitis virus. Proc Natl Acad Sci USA 105:629–634
Dodd RY, Notari EPt, Stramer SL (2002) Current prevalence and incidence of infectious disease markers and estimated window-period risk in the American Red Cross blood donor population. Transfusion 42:975–979
van Rhenen D, Gulliksson H, Cazenave JP, Pamphilon D, Ljungman P, Kluter H, Vermeij H, Kappers-Klunne M, de Greef G, Laforet M et al (2003) Transfusion of pooled buffy coat platelet components prepared with photochemical pathogen inactivation treatment: the euroSPRITE trial. Blood 101:2426–2433
McCullough J, Vesole DH, Benjamin RJ, Slichter SJ, Pineda A, Snyder E, Stadtmauer EA, Lopez-Plaza I, Coutre S, Strauss RG et al (2004) Therapeutic efficacy and safety of platelets treated with a photochemical process for pathogen inactivation: the SPRINT trial. Blood 104:1534–1541
Ruane PH, Edrich R, Gampp D, Keil SD, Leonard RL, Goodrich RP (2004) Photochemical inactivation of selected viruses and bacteria in platelet concentrates using riboflavin and light. Transfusion 44:877–885
AuBuchon JP, Herschel L, Roger J, Taylor H, Whitley P, Li J, Edrich R, Goodrich RP (2005) Efficacy of apheresis platelets treated with riboflavin and ultraviolet light for pathogen reduction. Transfusion 45:1335–1341
Mohr H (2001) Methylene blue and thionine in pathogen inactivation of plasma and platelet concentrates. Transfus Apher Sci 25:183–184
Acknowledgments
The authors acknowledge Dr. Karim Benhioud (Institut Gustave Roussy, Villejuif) for providing the mouse model of adenovirus infection and Miss Laurence Momeux for photographic help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Flaujac, C., Boukour, S. & Cramer-Bordé, E. Platelets and viruses: an ambivalent relationship. Cell. Mol. Life Sci. 67, 545–556 (2010). https://doi.org/10.1007/s00018-009-0209-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-009-0209-x