Abstract
Background/objective
Naringenin is a member of the flavonoid family that can perform many biological processes to treat a wide range of inflammatory diseases and pathological conditions related to oxidative stress (OS). Naringenin immunomodulatory activities have been the subject of recent research as an effective alternative treatment for autoimmune disorders. The effects of naringenin on the levels of inflammatory biomarkers and OS factors in animal models of autoimmune disorders (ADs) were studied in this meta-analysis.
Methods
Up until January 2022, electronic databases such as Cochrane Library and EMBASE, PubMed, Web of Science, and Scopus were used to conduct a comprehensive literature search in English language. To evaluate the effect of naringenin on inflammatory mediators, such as TNF-α, IL-6, IL-β, IFN-γ, NF-κB, and nitric oxide, and OS biomarkers, such as CAT, SOD, GPx, GSH and MDA, in AD models, we measured the quality assessment and heterogeneity test using the PRISMA checklist protocol and I2 statistic, respectively. A random-effects model was employed based on the heterogeneity test, and then pooled data were standardized as mean difference (SMD) with a 95% confident interval (CI).
Results
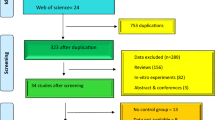
We excluded all clinical trials, cell experiment studies, animal studies with different parameters, non-autoimmune disease models, and an inadequate series of studies for quantitative synthesis. Finally, from 627 potentially reports, 12 eligible studies were included in the meta-analysis. Data were collected from several groups. Of these, 153 were in the naringenin group and 149 were in the control group. Our meta-analysis of the pooled data for the parameters of inflammation and OS indicated that naringenin significantly reduced the levels of NF-κB (SMD − 3.77, 95% CI [− 6.03 to − 1.51]; I2 = 80.1%, p = 0.002), IFN-γ (SMD − 6.18, 95% CI [− 8.73 to − 3.62]; I2 = 53.7%, p = 0.115), and NO (SMD − 3.97, 95% CI [− 5.50 to − 2.45]; I2 = 73.4%, p = 0.005), IL-1β (SMD − 4.23, 95% CI [− 5.09 to − 3.37]; I2 = 0.0%, p = 0.462), IL-6 (SMD − 5.84, 95% CI [− 7.83 to − 3.85]; I2 = 86.5%, p < 0.001), and TNF-α (SMD − 5.10, 95% CI [− 6.34 to − 3.86]; I2 = 74.7%, p < 0.001). These findings also demonstrated the efficacy of naringenin on increasing the levels of CAT (SMD 4.19, 95% CI [1.33 to 7.06]; I2 = 79.9%, p = 0.007), GSH (SMD 4.58, 95% CI [1.64 to 7.51]; I2 = 90.5%, p < 0.001), and GPx (SMD 9.65, 95% CI [2.56 to 16.74]; I2 = 86.6%, p = 0.001) and decreasing the levels of MDA (SMD − 3.65, 95% CI [− 4.80 to − 2.51]; I2 = 69.4%, p = 0.001) than control groups. However, treatment with naringenin showed no statistically difference in SOD activity (SMD 1.89, 95% CI [− 1.11 to 4.89]; I2 = 93.6%, p < 0.001).
Conclusion
Overall, our findings revealed the immunomodulatory potential of naringenin as an alternative treatment on inhibition of inflammation and OS in several autoimmune-related diseases. Nevertheless, regarding the limitation of clinical trials, strong preclinical models and clinical settings in the future are needed that address the effects of naringenin on ADs. Before large-scale clinical studies, precise human pharmacokinetic investigations are required to determine the dosage ranges and evaluate the initial safety profile of naringenin.





Similar content being viewed by others
References
Rosenblum MD, Remedios KA, Abbas AK. Mechanisms of human autoimmunity. J Clin Investig. 2015;125:2228–33.
Research IoMCoWsH. Women’s health research: progress, pitfalls, and promise. Washington, DC: National Academies Press; 2010.
Tavakolpour S. Towards personalized medicine for patients with autoimmune diseases: opportunities and challenges. Immunol Lett. 2017;190:130–8.
O’prey J, Brown J, Fleming J, Harrison PR. Effects of dietary flavonoids on major signal transduction pathways in human epithelial cells. Biochem Pharmacol. 2003;66:2075–88.
Kumar S, Pandey AK. Chemistry and biological activities of flavonoids: an overview. Sci World J. 2013; 2013.
Faramarzi F, Alimohammadi M, Rahimi A, Alizadeh-Navaei R, Shakib RJ, Rafiei A. Naringenin induces intrinsic and extrinsic apoptotic signaling pathways in cancer cells: A systematic review and meta-analysis of in vitro and in vivo data. Nutr Res. 2022. https://doi.org/10.1016/j.nutres.2022.05.003.
Gutierrez-Merino C, Lopez-Sanchez C, Lagoa R, Samhan-Arias AK, Bueno C, Garcia-Martinez V. Neuroprotective actions of flavonoids. Curr Med Chem. 2011;18:1195–212.
Lee H, Kim YO, Kim H, Kim SY, Noh HS, Kang SS, et al. Flavonoid wogonin from medicinal herb is neuroprotective by inhibiting inflammatory activation of microglia. FASEB J. 2003;17:1–21.
Park S, Sapkota K, Kim S, Kim H, Kim S. Kaempferol acts through mitogen-activated protein kinases and protein kinase B/AKT to elicit protection in a model of neuroinflammation in BV2 microglial cells. Br J Pharmacol. 2011;164:1008–25.
Sternberg Z, Chadha K, Lieberman A, Drake A, Hojnacki D, Weinstock-Guttman B, et al. Immunomodulatory responses of peripheral blood mononuclear cells from multiple sclerosis patients upon in vitro incubation with the flavonoid luteolin: additive effects of IFN-β. J Neuroinflammation. 2009;6:1–8.
Rodriguez-Canales M, Martinez-Galero E, Nava-Torres AD, Sanchez-Torres LE, Garduño-Siciliano L, Canales-Martinez MM, et al. Anti-Inflammatory and antioxidant activities of the methanolic extract of Cyrtocarpa procera bark reduces the severity of ulcerative colitis in a chemically induced colitis model. Mediators Inflamm. 2020;2020:1–11.
Manchope MF, Artero NA, Fattori V, Mizokami SS, Pitol DL, Issa JP, et al. Naringenin mitigates titanium dioxide (TiO 2)-induced chronic arthritis in mice: role of oxidative stress, cytokines, and NFκB. Inflamm Res. 2018;67:997–1012.
Al-Rejaie SS, Abuohashish HM, Al-Enazi MM, Al-Assaf AH, Parmar MY, Ahmed MM. Protective effect of naringenin on acetic acid-induced ulcerative colitis in rats. World J Gastroenterol: WJG. 2013;19:5633.
Fan R, Pan T, Zhu A-L, Zhang M-H. Anti-inflammatory and anti-arthritic properties of naringenin via attenuation of NF-κB and activation of the heme oxygenase (HO)-1/related factor 2 pathway. Pharmacol Rep. 2017;69:1021–9.
Wang J, Qi Y, Niu X, Tang H, Meydani SN, Wu D. Dietary naringenin supplementation attenuates experimental autoimmune encephalomyelitis by modulating autoimmune inflammatory responses in mice. J Nutr Biochem. 2018;54:130–9.
Abrego-Peredo A, Romero-Ramirez H, Espinosa E, Lopez-Herrera G, Garcia-Garcia F, Flores-Munoz M, et al. Naringenin mitigates autoimmune features in lupus-prone mice by modulation of T-cell subsets and cytokines profile. PLoS ONE. 2020;15: e0233138.
Wojnar W, Zych M, Kaczmarczyk-Sedlak I. Antioxidative effect of flavonoid naringenin in the lenses of type 1 diabetic rats. Biomed Pharmacother. 2018;108:974–84.
Li Y-R, Chen D-Y, Chu C-L, Li S, Chen Y-K, Wu C-L, et al. Naringenin inhibits dendritic cell maturation and has therapeutic effects in a murine model of collagen-induced arthritis. J Nutr Biochem. 2015;26:1467–78.
Naeini F, Namkhah Z, Ostadrahimi A, Tutunchi H, Hosseinzadeh-Attar MJ. A comprehensive systematic review of the effects of naringenin, a citrus-derived flavonoid, on risk factors for nonalcoholic fatty liver disease. Adv Nutr. 2021;12:413–28.
Rahimi A, Alimohammadi M, Faramarzi F, Alizadeh-Navaei R, Rafiei A. The effects of apigenin administration on the inhibition of inflammatory responses and oxidative stress in the lung injury models: a systematic review and meta-analysis of preclinical evidence. Inflammopharmacology. 2022. https://doi.org/10.1007/s10787-022-00994-0.
Liberati A, Altman D, Tetzlaff J. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. BMJ. 2009; 339.
Hooijmans CR, Rovers MM, de Vries R, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE’s risk of bias tool for animal studies. BMC Med Res Methodol. 2014;14:1–9.
Amaro MI, Rocha J, Vila-Real H, Eduardo-Figueira M, Mota-Filipe H, Sepodes B, et al. Anti-inflammatory activity of naringin and the biosynthesised naringenin by naringinase immobilized in microstructured materials in a model of DSS-induced colitis in mice. Food Res Int. 2009;42:1010–7.
Chaen Y, Yamamoto Y, Suzuki T. Naringenin promotes recovery from colonic damage through suppression of epithelial tumor necrosis factor–α production and induction of M2-type macrophages in colitic mice. Nutr Res. 2019;64:82–92.
Dou W, Zhang J, Sun A, Zhang E, Ding L, Mukherjee S, et al. Protective effect of naringenin against experimental colitis via suppression of Toll-like receptor 4/NF-κB signalling. Br J Nutr. 2013;110:599–608.
Hajizadeh A, Abtahi Froushani S, Tehrani A, Azizi S, Bani Hashemi S. Effects of Naringenin on experimental rheumatoid arthritis in Wistar rats. Archives of Razi Institute; 2020.
Wang J, Wei T, Gao J, He H, Chang X, Yan T. Effects of Naringenin on inflammation in complete freund’s adjuvant-induced arthritis by regulating Bax/Bcl-2 balance. Inflammation. 2015;38:245–51.
Niu X, Sang H, Wang J. Naringenin attenuates experimental autoimmune encephalomyelitis by protecting the intact of blood-brain barrier and controlling inflammatory cell migration. J Nutr Biochem. 2021;89: 108560.
Gupta SC, Tyagi AK, Deshmukh-Taskar P, Hinojosa M, Prasad S, Aggarwal BB. Downregulation of tumor necrosis factor and other proinflammatory biomarkers by polyphenols. Arch Biochem Biophys. 2014;559:91–9.
Jin L, Zeng W, Zhang F, Zhang C, Liang W. Naringenin ameliorates acute inflammation by regulating intracellular cytokine degradation. J Immunol. 2017;199:3466–77.
Du G, Jin L, Han X, Song Z, Zhang H, Liang W. Naringenin: a potential immunomodulator for inhibiting lung fibrosis and metastasis. Can Res. 2009;69:3205–12.
Glass CK, Saijo K, Winner B, Marchetto MC, Gage FH. Mechanisms underlying inflammation in neurodegeneration. Cell. 2010;140:918–34.
Pinho-Ribeiro FA, Zarpelon AC, Mizokami SS, Borghi SM, Bordignon J, Silva RL, et al. The citrus flavonone naringenin reduces lipopolysaccharide-induced inflammatory pain and leukocyte recruitment by inhibiting NF-κB activation. J Nutr Biochem. 2016;33:8–14.
Yoshida H, Watanabe W, Oomagari H, Tsuruta E, Shida M, Kurokawa M. Citrus flavonoid naringenin inhibits TLR2 expression in adipocytes. J Nutr Biochem. 2013;24:1276–84.
Pinho-Ribeiro FA, Zarpelon AC, Fattori V, Manchope MF, Mizokami SS, Casagrande R, et al. Naringenin reduces inflammatory pain in mice. Neuropharmacology. 2016;105:508–19.
Manchope MF, Calixto-Campos C, Coelho-Silva L, Zarpelon AC, Pinho-Ribeiro FA, Georgetti SR, et al. Naringenin inhibits superoxide anion-induced inflammatory pain: role of oxidative stress, cytokines, Nrf-2 and the NO− cGMP− PKG− KATPChannel signaling pathway. PLoS ONE. 2016;11: e0153015.
Martinez RM, Pinho-Ribeiro FA, Steffen VS, Caviglione CV, Vignoli JA, Barbosa DS, et al. Naringenin inhibits UVB irradiation-induced inflammation and oxidative stress in the skin of hairless mice. J Nat Prod. 2015;78:1647–55.
Hämäläinen M, Nieminen R, Vuorela P, Heinonen M, Moilanen E. Anti-inflammatory effects of flavonoids: genistein, kaempferol, quercetin, and daidzein inhibit STAT-1 and NF-κB activations, whereas flavone, isorhamnetin, naringenin, and pelargonidin inhibit only NF-κB activation along with their inhibitory effect on iNOS expression and NO production in activated macrophages. Mediat Inflamm 2007; 2007.
Smith AM, Rahman FZ, Hayee BH, Graham SJ, Marks DJ, Sewell GW, et al. Disordered macrophage cytokine secretion underlies impaired acute inflammation and bacterial clearance in Crohn’s disease. J Exp Med. 2009;206:1883–97.
Zeng W, Jin L, Zhang F, Zhang C, Liang W. Naringenin as a potential immunomodulator in therapeutics. Pharmacol Res. 2018;135:122–6.
Hartmann RM, Martins MIM, Tieppo J, Fillmann HS, Marroni NP. Effect of Boswellia serrata on antioxidant status in an experimental model of colitis rats induced by acetic acid. Dig Dis Sci. 2012;57:2038–44.
Closa D, Folch-Puy E. Oxygen free radicals and the systemic inflammatory response. IUBMB Life. 2004;56:185–91.
Isozaki Y, Yoshida N, Kuroda M, Takagi T, Handa O, Kokura S, et al. Effect of a novel water-soluble vitamin E derivative as a cure for TNBS-induced colitis in rats. Int J Mol Med. 2006;17:497–502.
Yoshikawa T, Ueda S, Naito Y, Takahashi S, Oyamada H, Morita Y, et al. Role of oxygen-derived free radicals in gastric mucosal injury induced by ischemia or ischemia-reperfusion in rats. Free Radical Res Commun. 1989;7:285–91.
Bitiren M, Karakilcik AZ, Zerin M, Ozardalı I, Selek S, Nazlıgül Y, et al. Protective effects of selenium and vitamin E combination on experimental colitis in blood plasma and colon of rats. Biol Trace Elem Res. 2010;136:87–95.
Ademoglu E, Erbil Y, Tam B, Barbaros U, Ilhan E, Olgac V, et al. Do vitamin E and selenium have beneficial effects on trinitrobenzenesulfonic acid-induced experimental colitis. Dig Dis Sci. 2004;49:102–8.
Jain A, Yadav A, Bozhkov A, Padalko V, Flora S. Therapeutic efficacy of silymarin and naringenin in reducing arsenic-induced hepatic damage in young rats. Ecotoxicol Environ Saf. 2011;74:607–14.
Han X, Gao S, Cheng Y, Sun Y, Liu W, Tang L, et al. Protective effect of naringenin-7-O-glucoside against oxidative stress induced by doxorubicin in H9c2 cardiomyocytes. Biosci Trends. 2012;6:19–25.
Wang J, Yang Z, Lin L, Zhao Z, Liu Z, Liu X. Protective effect of naringenin against lead-induced oxidative stress in rats. Biol Trace Elem Res. 2012;146:354–9.
Vasanthi P, Nalini G, Rajasekhar G. Role of tumor necrosis factor-alpha in rheumatoid arthritis: a review. APLAR J Rheumatol. 2007;10:270–4.
Pannu A, Goyal RK, Ojha S, Nandave M. Naringenin: a promising flavonoid for herbal treatment of rheumatoid arthritis and associated inflammatory disorders. In: Bioactive Food as Dietary Interventions for Arthritis and Related Inflammatory Diseases. Amsterdam: Elsevier; 2019. p. 343–54.
Arend WP. The pathophysiology and treatment of rheumatoid arthritis. Arthritis Rheumatism. 1997;40:595–7.
Rengasamy KR, Khan H, Gowrishankar S, Lagoa RJ, Mahomoodally FM, Khan Z, et al. The role of flavonoids in autoimmune diseases: therapeutic updates. Pharmacol Ther. 2019;194:107–31.
Bayat P, Farshchi M, Yousefian M, Mahmoudi M, Yazdian-Robati R. Flavonoids, the compounds with anti-inflammatory and immunomodulatory properties, as promising tools in multiple sclerosis (MS) therapy: a systematic review of preclinical evidence. Int Immunopharmacol. 2021;95: 107562.
Hisamatsu T, Kanai T, Mikami Y, Yoneno K, Matsuoka K, Hibi T. Immune aspects of the pathogenesis of inflammatory bowel disease. Pharmacol Ther. 2013;137:283–97.
Hundorfean G, Neurath MF, Mudter J. Functional relevance of T helper 17 (Th17) cells and the IL-17 cytokine family in inflammatory bowel disease. Inflamm Bowel Dis. 2012;18:180–6.
Azuma T, Shigeshiro M, Kodama M, Tanabe S, Suzuki T. Supplemental naringenin prevents intestinal barrier defects and inflammation in colitic mice. J Nutr. 2013;143:827–34.
Lee J-H, Lee B, Lee H-S, Bae E-A, Lee H, Ahn Y-T, et al. Lactobacillus suntoryeus inhibits pro-inflammatory cytokine expression and TLR-4-linked NF-κB activation in experimental colitis. Int J Colorectal Dis. 2009;24:231–7.
Fort MM, Mozaffarian A, Stöver AG, da Silva CJ, Johnson DA, Crane RT, et al. A synthetic TLR4 antagonist has anti-inflammatory effects in two murine models of inflammatory bowel disease. J Immunol. 2005;174:6416–23.
Cury Y, Picolo G, Gutierrez VP, Ferreira SH. Pain and analgesia: The dual effect of nitric oxide in the nociceptive system. Nitric Oxide. 2011;25:243–54.
Niedbala W, Cai B, Liew F. Role of nitric oxide in the regulation of T cell functions. Ann Rheum Dis. 2006;65:iii37–40.
Funding
No sources of support was received.
Author information
Authors and Affiliations
Contributions
MA: investigation, conceptualization, writing—original draft preparation, methodology. AR: writing—review and editing, data curation. FF: writing-review, methodology. RA-N: formal analysis. AR: writing—review and editing, project administration, resources.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that not conflict of interest exists with the publication of this article.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alimohammadi, M., Mohammad, R.N., Rahimi, A. et al. The effect of immunomodulatory properties of naringenin on the inhibition of inflammation and oxidative stress in autoimmune disease models: a systematic review and meta-analysis of preclinical evidence. Inflamm. Res. 71, 1127–1142 (2022). https://doi.org/10.1007/s00011-022-01599-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-022-01599-7