Abstract
Objective
We selected a hit compound, 2-(4-{2-[(phenylthio)acetyl]-carbonohydrazonoyl}-phenoxy)acetamide (PA), by a molecular docking simulation between 636,565 compounds and caspase-1 protein. We examined the effect of PA on allergic rhinitis (AR) animal model.
Methods
We assessed the therapeutic effects and the regulatory mechanisms of ovalbumin (OVA)-sensitized mouse model of AR.
Results
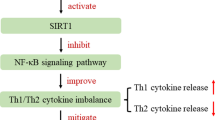
A molecular docking simulation and a kinetic assay indicated that PA regulates the caspase-1 activation through the interaction with the caspase-1 active site. In the AR animal model, PA significantly reduced the rub scoring increased by OVA. The up-regulated IgE, histamine, interleukin (IL)-1β, and thymic stromal lymphopoietin (TSLP) levels in the serum of OVA-sensitized mice were significantly decreased by the treatment with PA. Protein levels of IL-1β, IL-5, IL-6, IL-13, tumor necrosis factor-α, TSLP, cyclooxygenase-2, macrophage inflammatory protein-2, and intercellular adhesion molecule-1 were also significantly inhibited by the treatment with PA in the nasal mucosa tissues of the OVA-sensitized mice. In the PA-treated mice, the number of eosinophils and mast cells infiltrated by OVA-sensitization were also reduced. In addition, PA reduced the mast cell-derived caspase-1 activity and expression in the nasal mucosa tissues of the OVA-sensitized mice.
Conclusions
PA showed the possibility to regulate AR in OVA-induced AR models, suggesting that it has therapeutic potential for the management of AR as a lead compound.






Similar content being viewed by others
References
Howarth PH, Salagean M, Dokic D. Allergic rhinitis: not purely a histamine-related disease. Allergy. 2000;64:7–16.
Kramer MF, Jordan TR, Klemens C, Hilgert E, Hempel JM, Pfrogner E, Rasp G. Factors contributing to nasal allergic late phase eosinophilia. Am J Otolaryngol. 2006;27:190–9.
Zhu Z, Homer RJ, Wang Z, Chen Q, Geba GP, Wang J, Zhang Y, Elias JA. Pulmonary expression of interleukin-13 causes inflammation, mucus hypersecretion, subepithelial fibrosis, physiologic abnormalities, and eotaxin production. J Clin Invest. 1999;103:779–88.
Franchi L, Eigenbrod T, Munoz-Planillo R, Nunez G. The inflammasome: a caspase-1-activation platform that regulates immune responses and disease pathogenesis. Nat Immunol. 2009;10:241–7.
Kuida K, Lippke JA, Ku G, Harding MW, Livingston DJ, Su MS, Flavell RA. Altered cytokine export and apoptosis in mice deficient in interleukin-1 beta converting enzyme. Science. 1995;267:2000–3.
Shamaa OR, Mitra S, Gavrilin MA, Wewers WD. Monocyte caspase-1 is released in a stable, active high molecular weight complex distinct from the unstable cell lysate-activated caspase-1. PLoS One. 2015;10:e0142203.
Friesner RA, Banks JL, Murphy RB, Halgren TA, Klicic JJ, Mainz DT, Repasky MP, Knoll EH, Shelley M, Perry JK, Shaw DE, Francis P, Shenkin PS. Glide: a new approach for rapid, accurate docking and scoring. 1. Method and assessment of docking accuracy. J Med Chem. 2004;47:1739–49.
Oh HA, Ryu JG, Cha WS, Kim HM, Jeong HJ. Therapeutic effects of traditional Korean medicine, Jeechool-Whan in allergic rhinitis model. TANG. 2012;2:e9.
Grzelewska-Rzymowska I, Pietrzkowicz M. Role of tumor necrosis factor alpha in allergic inflammation and airway hyperresponsiveness. Pol Merkur Lekarski. 2004;16:173–8.
Profita M, Sala A, Bonnano A, Riccobono L, Siena L, Melis MR, Di Giorgi R, Mirabella F, Gjomarkaj M, Bonsignore G, Vignola AM. Increased prostaglandin E2 concentrations and cyclooxygenase-2 expression in asthmatic subjects with sputum eosinophilia. J Allergy Clin Immunol. 2003;112:709–16.
Oh HA, Kim HM, Jeong HJ. Distinct effects of imperatorin on allergic rhinitis: imperatorin inhibits caspase-1 activity in vivo and in vitro. J Pharmacol Exp Ther. 2011;339:72–81.
Spencer LA, Szela CT, Perez SA, Kirchhoffer CL, Neves JS, Radke AL, Weller PF. Human eosinophils constitutively express multiple Th1, Th2, and immunoregulatory cytokines that are secreted rapidly and differentially. J Leukoc Biol. 2009;85:117–23.
Lee SH, Stehlik C, Reed JC. Cop, a caspase recruitment domain-containing protein and inhibitor of caspase-1 activation processing. J Biol Chem. 2001;276:34495–500.
Mandhane SN, Shah JH, Thennati R. Allergic rhinitis: an update on disease, present treatments and future prospects. Int Immunopharmacol. 2011;11:1646–62.
Rosenberg HF, Dyer KD, Foster PS. Eosinophils: changing perspectives in health and disease. Nat Rev Immunol. 2013;13:9–22.
Finkelman FD, Urban JF Jr. The other side of the coin: the protective role of the TH2 cytokines. J Allergy Clin Immunol. 2001;107:772–80.
Artis D, Humphreys NE, Bancroft AJ, Rothwell NJ, Potten CS, Grencis RK. Tumor necrosis factor alpha is a critical component of interleukin 13-mediated protective T helper cell type 2 responses during helminth infection. J Exp Med. 1999;190:953–62.
Ziegler SF. The role of thymic stromal lymphopoietin (TSLP) in allergic disorders. Curr Opin Immunol. 2010;22:795–9.
Liu X, Li M, Wu Y, Zhou Y, Zeng L, Huang T. Anti-IL-33 antibody treatment inhibits airway inflammation in a murine model of allergic asthma. Biochem Biophys Res Commun. 2009;386:181–5.
Seder RA, Paul WE, Davis MM. Groth de St, Fazekas B. The presence of interleukin 4 during in vitro priming determines the lymphokine-producing potential of CD4 + T cells from T cell receptor transgenic mice. J Exp Med. 1992;176:1091–8.
Pawankar R, Okuda M, Yssel H, Okumura K, Ra C. Nasal mast cells in perennial allergic rhinitics exhibit increased expression of the Fc epsilonRI, CD40L, IL-4, and IL-13, and can induce IgE synthesis in B cells. J Clin Invest. 1997;99:1492–9.
Pang L, Pitt D, Petkova D, Knox AJ. The COX-1/COX-2 balance in asthma. Clin Exp Allergy. 1998;28:1050–8.
Smith WL, Dewitt DL, Garavito RM. Cyclooxygenase: structure, cellular and molecular biology. Annu Rev Biochem. 2000;69:145–81.
Melvin TA, Ramanathan M Jr. Role of innate immunity in the pathogenesis of allergic rhinitis. Curr Opin Otolaryngol. 2012;20:194–8.
Isobe Y, Kato T, Arita M. Emerging roles of eosinophils and eosinophil-derived lipid mediators in the resolution of inflammation. Front Immunol. 2012;3:270.
Sollberger G, Strittmatter GE, Garstkiewicz M, Sand J, Beer HD. Caspase-1: the inflammasome and beyond. Innate Immun. 2014;20:115–25.
Pawankar R, Lee KH, Nonaka M, Takizawa R. Role of mast cells and basophils in chronic rhinosinusitis. Clin Allergy Immunol. 2007;20:93–101.
Gilfillan AM, Beaven MA. Regulation of mast cell responses in health and disease. Crit Rev Immunol. 2012;31:475–529.
Rothenberg ME, Hogan SP. The eosinophil. Annu Rev Immunol. 2006;24:147–74.
Oh HA, Kim HM, Jeong HJ. Alleviation of allergic rhinitis symptoms with Pyeongwee-San extract (KMP6). Immunopharmacol Immunotoxicol. 2012;34:35–142.
Grzegorczyk J, Kowalski ML, Pilat A, Iwaszkiewicz J. Increased apoptosis of peripheral blood mononuclear cells in patients with perennial allergic asthma/rhinitis: relation to serum markers of apoptosis. Mediat Inflamm. 2002;11:225–33.
Cunha TM, Talbot J, Pinto LG, Vieira SM, Souza GR, Guerrero AT, Sonego F, Verri WA Jr, Zamboni DS, Ferreira SH, Cunha FQ. Caspase-1 is involved in the genesis of inflammatory hypernociception by contributing to peripheral IL-1β maturation. Mol Pain. 2010;6:63.
Moon PD, Kim HM. Thymic stromal lymphopoietin is expressed and produced by caspase-1/NF-kB pathway in mast cells. Cytokine. 2011;54:239–43.
Acknowledgments
This research was supported by the Academic Research fund of Hoseo University in 2015 (2015-0228).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: John Di Battista.
Rights and permissions
About this article
Cite this article
Kim, HY., Nam, SY., Jang, JB. et al. 2-(4-{2-[(phenylthio)acetyl]carbonohydrazonoyl}phenoxy)acetamide as a new lead compound for management of allergic rhinitis. Inflamm. Res. 65, 963–973 (2016). https://doi.org/10.1007/s00011-016-0979-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-016-0979-1