Abstract
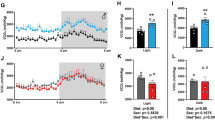
Background: An increase in lipid storage as a consequence of feeding animals with high-glycemic index (GI) diets has been observed by many authors. Ghrelin is one of the most important orexigenic hormones, and curiously, its fasting plasma levels are decreased in human obesity. Aim: As ghrelin secretion is affected by insulin concentration, we hypothesized that carbohydrates with different glycemic responses might influence fasting plasma ghrelin levels. Material and methods: Twenty rats were divided into two groups and fed adlibituma low-GI or a high-GI diet for 21 days. Results: In rats fed a high- vs low-GI diet we observed: increased food intake (18.9±0.6 vs 16.4±2.0 g/day; p<0.01), increased weight gain (28.8±6.6 vs 16.4±6% of initial weight; p<0.01), higher relative weight of epididymal fat pads (1.7±0.4 vs 1.4±0.3%; p=0.05), but lower total fasting ghrelin levels (41.1 ±10.7 vs 59.5±9.8 pg/ml; p=0.05). Conclusions: Ghrelin appeared to be downregulated in rats fed a high-GI diet; this observation could be related to the higher food intake and fat mass observed in these rats and to the effects of insulin response on ghrelin levels.
Similar content being viewed by others
References
Livesey G, Taylor R, Hulshof T, Howlett J. Glycemic response and health—a systematic review and meta-analysis: relations between dietary glycemic properties and health outcomes. Am J Clin Nutr 2008, 87: 258S–68S.
Howlett J, Ashwell M. Glycemic response and health: summary of a workshop. Am J Clin Nutr 2008, 87: 212S-6S.
Alfenas RC, Mattes RD. Influence of glycemic index/load on glycemic response, appetite, and food intake in healthy humans. Diabetes Care 2005, 28: 2123–9.
Flint A, Møller BK, Raben A, et al. Glycemic and insulinemic responses as determinants of appetite in humans. Am J Clin Nutr 2006, 84: 1365–73.
Flint A, Gregersen NT, Gluud LL, et al. Associations between postprandial insulin and blood glucose responses, appetite sensations and energy intake in normal weight and overweight individuals: ameta-analysis of test meal studies. Br J Nutr 2007, 98: 17–25.
Thomas DE, Elliott EJ, Baur L. Low glycaemic index or low glycaemic load diets for overweight and obesity. Cochrane Database Syst Rev 2007, 18: CD005105.
Kabir M, Rizkalla SW, Quignard-Boulangé A, et al. A high glycemic index starch diet affects lipid storage-related enzymes in normal and to a lesser extent in diabetic rats. J Nutr 1998, 128: 1878–83.
Maegawa H, Kobayashi M, Ishibashi O, Takata Y, Shigeta Y. Effect of diet change on insulin action: difference between muscles and adipocytes. Am J Physiol 1986, 251: E616–23.
Pawlak DB, Kushner JA, Ludwig DS. Effects of dietary glycaemic index on adiposity, glucose homoeostasis, and plasma lipids in animals. Lancet 2004, 364: 778–85.
Scribner KB, Pawlak DB, Aubin CM, Majzoub JA, Ludwig DS. Long-term effects of dietary glycemic index on adiposity, energy metabolism, and physical activity in mice. Am J Physiol Endocrinol Metab 2008, 295: E1126–31.
McGarry JD, Mannaerts GP, Foster DW. A possible role for malonyl- CoA in the regulation of hepatic fatty acid oxidation and ketogenesis. J Clin Invest 1977, 60: 265–70.
Ludwig DS. The glycemic index: physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA 2002, 287: 2414–23.
Du H, van der A DL, van Bakel MM, et al. Glycemic index and glycemic load in relation to food and nutrient intake and metabolic risk factors in a Dutch population. Am J Clin Nutr 2008, 87: 655–61.
Barclay AW, Petocz P, McMillan-Price J, et al. Glycemic index, glycemic load, and chronic disease risk, a meta-analysis of observational studies. Am J Clin Nutr 2008, 87: 627–37.
De Vriese C, Delporte C. Influence of ghrelin on food intake and energy homeostasis. Curr Opin Clin Nutr Metab Care 2007, 10: 615–9.
Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999, 402: 656–60.
Cummings DE, Purnell JQ, Frayo RS, Schmidova K, Wisse BE, Weigle DS. A preprandial rise in plasma ghrelin levels suggests a role in meal initiation in humans. Diabetes 2001, 50: 1714–9.
Callahan HS, Cummings D, Pepe M, Breen P, Matthys C, Weigle D. Postprandial suppression of plasma ghrelin level is proportional to ingested caloric load but does not predict intermeal interval in humans. J Clin Endocrinol Metab 2004, 89: 1319–24.
Tschöp M, Wawarta R, Riepl R, Bidlingmaier M, Landgraf R, Folwaczny C. Post-prandial decrease of circulating human ghrelin levels. J Endocrinol Invest 2001, 24: RC19–21.
Tschöp M, Weyer C, Tataranni PA, Devanarayan V, Ravussin E, Heiman ML. Circulating ghrelin levels are decreased in human obesity. Diabetes 2001, 50: 707–9.
English PJ, Ghatei M, Malik I, Bloom S, Wilding J. Food fails to suppress ghrelin levels in obese humans. J Clin Endocrinol Metab 2002, 87: 2984–7.
McLaughlin T, Abbasi F, Lamendola C, Frayo RS, Cummings DE. Plasma ghrelin concentrations are decreased in insulin-resistant obese adults relative to equally obese insulin-sensitive controls. J Clin Endocrinol Metab 2004, 89: 1630–5.
Katsuki A, Urakawa H, Gabazza EC, et al. Circulating levels of active ghrelin is associated with abdominal adiposity, hyperinsulinemia and insulin resistance in patients with type 2 diabetes mellitus. Eur J Endocrinol 2004, 151: 573–7.
Dezaki K, Sone H, Yada T. Ghrelin is a physiological regulator of insulin release in pancreatic islets and glucose homeostasis. Pharmacol Ther 2008, 118: 239–49.
Pawlak DB, Bryson JM, Denyer GS, Brand-Miller JC. High glycemic index starch promotes hypersecretion of insulin and higher body fat in rats without affecting insulin sensitivity. J Nutr 2001, 131: 99–104.
Scribner KB, Pawlak DB, Aubin CM, Majzoub JA, Ludwig DS. Long-term effects of dietary glycemic index on adiposity, energy metabolism, and physical activity in mice. Am J Physiol Endocrinol Metab 2008, 295: E1126–31.
Pereira MA, Swain J, Goldfine AB, Rifai N, Ludwig DS. Effects of a low-glycemic load diet on resting energy expenditure and heart diseas risk factors during weight loss. JAMA 2004, 292: 2482–90.
Salbe AD, Tschöp MH, DelParigi A, Venti CA, Tataranni PA. Negative relationship between fasting plasma ghrelin concentrations and ad libitum food intake. J Clin Endocrinol Metab 2004, 89: 2951–6.
Nakagawa E, Nagaya N, Okumura H, et al. Hyperglycaemia suppresses the secretion of ghrelin, a novel growth-hormone-releasing peptide: responses to the intravenous and oral administration of glucose. Clin Sci (Lond) 2002, 103: 325–8.
Möhlig M, Spranger J, Otto B, Ristow M, Tschöp M, Pfeiffer AF. Euglycemic hyperinsulinemia, but not lipid infusion, decreases circulating ghrelin levels in humans. J Endocrinol Invest 2002, 25: RC36–8.
Greenman Y, Golani N, Gilad S, Yaron M, Limor R, Stern N. Ghrelin secretion is modulated in a nutrient- and gender-specific manner. Clin Endocrinol (Oxf) 2004, 60: 382–8.
Broglio F, Arvat E, Benso A, et al. Ghrelin, a natural GH secretagogue produced by the stomach, induces hyperglycemia and reduces insulin secretion in humans. J Clin Endocrinol Metab 2001, 86: 5083–6.
Cui C, Ohnuma H, Daimon M, et al. Ghrelin infused into the portal vein inhibits glucose-stimulated insulin secretion in Wistar rats. Peptides 2008, 29: 1241–6.
Blom WA, Stafleu A, de Graaf C, Kok FJ, Schaafsma G, Hendriks HF. Ghrelin response to carbohydrate-enriched breakfast is related to insulin. Am J Clin Nutr 2005, 81: 367–75.
Tschöp M, Weyer C, Tataranni PA, Devanarayan V, Ravussin E, Heiman ML. Circulating ghrelin levels are decreased in human obesity. Diabetes 2001, 50: 707–9.
Fagerberg B, Hultén LM, Hulthe J. Plasma ghrelin, body fat, insulin resistance, and smoking in clinically healthy men: the atherosclerosis and insulin resistance study. Metabolism. 2003, 52: 1460–3.
Qi X, Reed JT, Wang G, Han S, Englander EW, Greeley Jr GH. Ghrelin secretion is not reduced by increased fat mass during diet-induced obesity. Am J Physiol Regul Integr Comp Physiol 2008, 295: R429–35.
Salbe AD, Tschop M, DelParigi A, Venti C, Tataranni P. Negative relationship between fasting plasma ghrelin concentrations and ad libitum food intake. J Clin Endocrinol Metab 2004, 89: 2951–6.
Katsuki A, Urakawa H, Gabazza EC, et al. Circulating levels of active ghrelin is associated with abdominal adiposity, hyperinsulinemia and insulin resistance in patients with type 2 diabetes mellitus. Eur Jour of Endocrinol 2004, 151: 573–7.
Langenberg C, Bergstrom J, Laughlin GA, Barrett-Connor E. Ghrelin and the metabolic syndrome in older adults. J Clin Endocrinol Metab 2005, 90: 6448–53.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sculati, M., Rossi, F., Cena, H. et al. Effect of dietary glycemic index on food intake, adiposity, and fasting plasma ghrelin levels in animals. J Endocrinol Invest 33, 250–253 (2010). https://doi.org/10.1007/BF03345788
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03345788