Summary
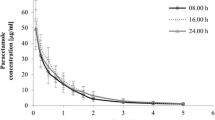
The pharmacokinetics of thymoxamine hydrochloride were studied in rabbits by the assessment of its ocular and systemic absorption after instillation of thymoxamine hydrochloride 0.5% eye drops. Plasma levels were compared with those observed after i.v. bolus administration of thymoxamine hydrochloride at 2.5 mg/kg. Deacetylthymoxamine is the main metabolite of thymoxamine, generated by esterase hydrolysis. It was evaluated, as an indication of the parent drug, in aqueous humor and plasma by an HPLC method with fluorescence detection (detection limit = 5 ng/ml). Thymoxamine was found to permeate the cornea and to be hydrolysed very quickly, showing very good absorption with a maximum aqueous humor concentration of deacetylthymoxamine of 2329 ng/ml 15 min after eye drop instillation. The study of the systemic absorption of thymoxamine allowed the exclusion of the possibility of systemic side effects following ocular treatment. In fact, considering the detection limit of the method, the plasma levels of deacetylthymoxamine are certainly more than 100-times lower than those observed with intravenous treatment.
Similar content being viewed by others
References
Birmingham A.T., Szolcsany I.J. (1965): Competitive blockade of adrenergic-receptors and histamine receptors by thymoxamine. J. Pharm. Pharmacol., 17, 449–458.
Foster R.W. (1966): The nature of the adrenergic receptors of the trachea of the guinea pig. J. Pharm. Pharmacol., 18, 1–12.
Drew G.M. (1976): Effects of alpha-adrenoceptor agonists and antagonists on pre- and post-synaptically located alpha-adrenoceptors. Eur. J. Pharmacol., 36, 313–320.
Bonomi L., Tomazzoli L. (1977): Thymoxamine and intraocular pressure. Graefes Arch. Clin. Exp. Ophthalmol., 204, 95–100.
Wand M., Grant W.M. (1976): Thymoxamine hydrochloride: effects on the facility of outflow and intraocular pressure. Invest. Ophthalmol., 15, 400–403.
Mapstone R. (1970): Safe mydriasis. Br. J. Ophthalmol., 54, 690–692.
Rutkowski P.C., Fernandez J.L., Galin M.A., Halasa A.H. (1973): Alpha adrenergic blockade in the treatment of angle-closure glaucoma. Trans Am. Acad. Ophthalmol. Otolaryngol., 77, 137–142.
Feniou C., Neau B., Prat G., Cheze C., Fauran F., Roquebert J. (1980): Metabolism of thymoxamine: identification of metabolites in rat. J. Pharm. Pharmacol., 32, 104–107.
Nielsen-Kudsk F. Jakobsen P., Magnussen I.B. (1980): Plasma induced biotransformation of thymoxamine and its kinetics. Acta Pharmacol. Toxicol., 47, 11–16.
Wollmer K.O., Liedtke B., Poisson A., Hodenberg A., Steinbrecher W. (1985): Metabolism of thymoxamine. I. Studies with14C-thymoxamine in rats. Eur. J. Drug Metab. Pharmacokinet., 10, 61–69.
Duchene P., Bernouillet C., Bromet-petit J., et al. (1988): Metabolism of14C-thymoxamine in rat and man. Xenobiotica, 8, 919–928.
Marquer C., Trouvin J.H., Lacolle J.Y., Dupont Ch., Jacquot C. (1991): Pharmacokinetics of a prodrug thymoxamine: dose-dependence of the metabolite ratio in healthy subjects. Eur. J. Drug Metab. Pharmacokinet., 16, 183–188.
Von Wasilewski A. (1971): Klinisch-pharmakologischer Nachweis der Wirkung von Essigsäure — [4-(2-dimethylaminoaethoxy)-5-isopropyl-2-methyl-phenyl]-ester. HCl (Moxisylyt). Arzneimittelforschung., 21, 1183–1189.
Gomeni R., Gomeni C. (1978): Comp. Biomed. Res. 11, 345.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aldana, I., González-Peñas, E., Fos, D. et al. Pharmacokinetics of thymoxamine in rabbits after ophthalmic and intravenous administration. Eur. J. Drug Metab. Pharmacokinet. 19, 79–83 (1994). https://doi.org/10.1007/BF03188827
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03188827